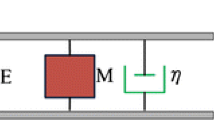
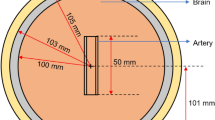
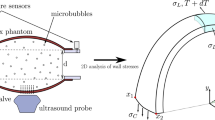
Abstract
Peripheral arterial occlusive disease is a serious cardiovascular disorder. The arterial occlusion leads to turbulent flow and arterial sound generation on the inner vessel wall. Stenosis-induced vibro-acoustic waves propagate through the surrounding soft tissues and reach the skin surface. In this study, the feasibility of noninvasive acoustic detection of the peripheral arterial stenosis is investigated using the vibration responses by means of experimental and computational models. Latex rubber tube is used to model the artery, and it is surrounded by a tissue mimicking phantom made of bovine gelatin. Vibration responses on phantom surface are measured using laser Doppler vibrometer, and computational results are obtained performing modal analysis. Experimental findings and computational results showed well agreement in terms of spectral content and vibration amplitudes. The effects of various stenosis severities, flow rates, and phantom thicknesses on the vibration responses are investigated from diagnostic perspective. Stenosis severities greater than 70% resulted in a considerable increase in vibration amplitudes. The structural mode shapes of the tissue phantom are dominant between 0 and 100 Hz, suppressing the signals generated by the stenosis. The optimum range of frequency for acoustic stenosis detection is concluded to be between 200 and 500 Hz, particularly around 300 Hz.

.













Similar content being viewed by others
References
Acikgoz S, Ozer MB, Royston TJ, Mansy HA, Sandler RH (2008) Experimental and computational models for simulating sound propagation within the lungs. J Vib Acoust 130(2):021010
Akay YM, Akay M, Welkowitz W, Semmlow JL, Kostis JB (1993) Noninvasive acoustical detection of coronary artery disease: a comparative study of signal processing methods. IEEE Trans Biomed Eng 40(6):571–578
Azimpour F, Caldwell E, Tawfik P, Duval S, Wilson RF (2016) Audible coronary artery stenosis. Am J Med 129(5):515–521
Banks HT, Hu S, Kenz ZR, Kruse C, Shaw S, Whiteman J, Brewin MP, Greenwald SE, Birch MJ (2014) Model validation for a noninvasive arterial stenosis detection problem. Math Biosci Eng 11(3):427–448
Bathe KJ, Wilson EL (1973) Solution methods for eigenvalue problems in structural mechanics. Int J Numer Meth Bio 6(2):213–226
Biswas M, Kuppili V, Saba L, Edla DR, Suri HS, Sharma A, Cuadrado-Godia E, Laird JR, Nicolaides A, Suri JS (2019) Deep learning fully convolution network for lumen characterization in diabetic patients using carotid ultrasound: a tool for stroke risk. Med Biol Eng Comput 57(2):543–564
Borisyuk AO (2002) Experimental study of noise produced by steady flow through a simulated vascular stenosis. J Sound Vib 256(3):475–498
Borisyuk AO (2002) Modeling of noise generation by a vascular stenosis. Int J Fluid Mech Res 29(1):65–86
Borisyuk AO (2003) Experimental study of wall pressure fluctuations in a pipe behind a stenosis. Int J Fluid Mech Res 30(3):264–278
Borisyuk AO (2003) Model study of noise field in the human chest due to turbulent flow in a larger blood vessel. J Fluid Struct 17(8):1095–1110
Borisyuk AO (2010) Experimental study of wall pressure fluctuations in rigid and elastic pipes behind an axisymmetric narrowing. J Fluid Struct 26(4):658–674
Cassanova RA, Giddens DP (1978) Disorder distal to modeled stenoses in steady and pulsatile flow. J Biomech 11:441–453
Chami HA, Keyes MJ, Vita JA, Mitchell GF, Larson MG, Fan S, Vasan RS, O’Connor GT, Benjamin EJ, Gottlieb DJ (2009) Brachial artery diameter, blood flow and flow-mediated dilation in sleep-disordered breathing. Vasc Med 14(4):351–360
Chang Y, Kim N, Stenfelt S (2016) The development of a whole-head human finite-element model for simulation of the transmission of bone-conducted sound. J Acoust Soc Am 140(3):1635–1651
Chhai P, Rhee K (2018) Effect of distal thickening and stiffening of plaque cap on arterial wall mechanics. Med Biol Eng Comput 56(11):2003–2013
Clark C (1976) The fluid mechanics of aortic stenosis—I. theory and steady flow experiments. J Biomech 9(8):521–528
Clark C (1977) Turbulent wall pressure measurements in a model of aortic stenosis. J Biomech 10(8):461–472
Clough RW, Penzien J (2003) Dynamics of structures. Computers & Structures, California
Fredberg JJ (1977) Origin and character of vascular murmurs: model studies. J Acoust Soc Am 61(4):1077–1085
Gayathri K, Shailendhra K (2014) Pulsatile blood flow in large arteries: comparative study of Burton’s and McDonald’s models. Appl Math Mech 35(5):575–590
Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Borden WB, Bravata DM, Dai S, Ford ES, Fox CS, Franco S, Fullerton HJ, Gillespie C, Hailpern SM, Heit JA, Howard VJ, Huffman MD, Kissela BM, Kittner SJ, Lackland DT, Lichtman JH, Lisabeth LD, Magid D, Marcus GM, Marelli A, Matchar DB, McGuire DK, Mohler ER, Moy CS, Mussolino ME, Nichol G, Paynter NP, Schreiner PJ, Sorlie PD, Stein J, Turan TN, Virani SS, Wong ND, Woo D, Turner MB (2013) AHA statistical update. Circulation 127:e62–e245
Guala A, Camporeale C, Ridolfi L, Mesin L (2017) Non-invasive aortic systolic pressure and pulse wave velocity estimation in a primary care setting: an in silico study. Med Eng Phys 42:91–98
Heidenreich PA, Trogdon JG, Khavjou OA, Butler J, Dracup K, Ezekowitz MD, Finkelstein EA, Hong Y, Johnston SC, Khera A, Lloyd-Jones DM, Nelson SA, Nichol G, Orenstein D, Wilson PWF, Woo YJ (2011) Forecasting the future of cardiovascular disease in the United States. Circulation 123(8):933–944
Holzapfel GA, Gasser TC, Stadler M (2002) A structural model for the viscoelastic behavior of arterial walls: continuum formulation and finite element analysis. Eur J Mech A-Solid 21(3):441–463
Holzapfel GA, Sommer G, Gasser CT, Regitnig P (2005) Determination of layer-specific mechanical properties of human coronary arteries with nonatherosclerotic intimal thickening and related constitutive modeling. Am J Physiol Heart Circ Physiol 289(5):H2048–H2058
Hurty WC, Rubinstein MF (1964) Dynamics of structures. Prentice-Hall, Upper Saddle River
Kirkeeide RL, Young DF, Cholvin NR (1977) Wall vibrations induced by flow through simulated stenoses in models and arteries. J Biomech 10(7):431–437
Krejza J, Arkuszewski M, Kasner SE, Weigele J, Ustymowicz A, Hurst RW, Cucchiara BL (2006) Messe SR (2006) carotid artery diameter in men and women and the relation to body and neck size. Stroke 37:1103–1105. https://doi.org/10.1161/01.STR.0000206440.48756.f7
Lees RS, Dewey CF (1970) Phonoangiography: a new noninvasive diagnostic method for studying arterial disease. Proc Natl Acad Sci U S A 67(2):935–942
Olson LG, Bathe KJ (1985) Analysis of fluid-structure interactions. A direct symmetric coupled formulation based on the fluid velocity potential. Comput Struct 21(1–2):21–32
Ozer MB, Acikgoz S, Royston TJ, Mansy HA, Sandler RH (2007) Boundary element model for simulating sound propagation and source localization within the lungs. J Acoust Soc Am 122(1):657–671
Rezvani-Sharif A., Tafazzoli-Shadpour M, Avolio A. (2018) Progressive changes of elastic moduli of arterial wall and atherosclerotic plaque components during plaque development in human coronary arteries. Med Biol Eng Comput 1–10
Royston TJ, Mansy HA, Sandler RH (1999) Excitation and propagation of surface waves on a viscoelastic half-space with application to medical diagnosis. J Acoust Soc Am 106(6):3678–3686
Royston TJ, Yazicioglu Y, Loth F (2003) Surface response of a viscoelastic medium to subsurface acoustic sources with application to medical diagnosis. J Acoust Soc Am 113(2):1109–1121
Salman HE, Sert C, Yazicioglu Y (2013) Computational analysis of high frequency fluid–structure interactions in constricted flow. Comput Struct 122:145–154
Salman HE, Yazicioglu Y (2017) Flow-induced vibration analysis of constricted artery models with surrounding soft tissue. J Acoust Soc Am 142(4):1913–1925
Sandgren T, Sonesson B, Ahlgren ÅR, Länne T (1999) The diameter of the common femoral artery in healthy human: influence of sex, age, and body size. J Vasc Surg 29(3):503–510
Sazonov I, Khir AW, Hacham WS, Boileau E, Carson JM, van Loon R, Ferguson C, Nithiarasu P (2017) A novel method for non-invasively detecting the severity and location of aortic aneurysms. Biomech Model Mechanobiol 16(4):1225–1242
Sussman T, Bathe KJ (1987) A finite element formulation for nonlinear incompressible elastic and inelastic analysis. Comput Struct 26(1–2):357–409
Sussman T, Sundqvist J (2003) Fluid–structure interaction analysis with a subsonic potential-based fluid formulation. Comput Struct 81(8):949–962
Thomas JL, Winther S, Wilson R, Bøttcher M (2017) A novel approach to diagnosing coronary artery disease: acoustic detection of coronary turbulence. Int J Card Imaging 33(1):129–136
Tobin RJ, Chang ID (1976) Wall pressure spectra scaling downstream of stenoses in steady tube flow. J Biomech 9(10):633–640
Van Loocke M, Lyons CG, Simms CK (2008) Viscoelastic properties of passive skeletal muscle in compression: stress-relaxation behaviour and constitutive modelling. J Biomech 41(7):1555–1566
Wang JZ, Tie BING, Welkowitz W, Semmlow JL, Kostis JB (1990) Modeling sound generation in stenosed coronary arteries. IEEE Trans Biomed Eng 37(11):1087–1094
Weng SF, Reps J, Kai J, Garibaldi JM, Qureshi N (2017) Can machine-learning improve cardiovascular risk prediction using routine clinical data? PLoS One 12(4):e0174944
Winther S, Nissen L, Schmidt SE, Westra JS, Rasmussen LD, Knudsen LL, Madsen LH, Johansen JK, Larsen BS, Struijk JJ, Frost L, Holm NR, Christiansen EH, Botker HE, Bøttcher M (2018) Diagnostic performance of an acoustic-based system for coronary artery disease risk stratification. Heart 104(11):928–935
Yazicioglu Y, Royston TJ, Spohnholtz T, Martin B, Loth F, Bassiouny HS (2005) Acoustic radiation from a fluid-filled, subsurface vascular tube with internal turbulent flow due to a constriction. J Acoust Soc Am 118(2):1193–1209
Young DF (1979) Fluid mechanics of arterial stenosis. J Biomech Eng 101(3):157–175
Acknowledgments
The authors would like to thank Prof. Oguz Uzol for the use of the facilities of the Center for Wind Energy (METUWIND) at Middle East Technical University.
Funding
This work is internally supported by Middle East Technical University (METU) Scientific Research Project (BAP) funding with grant number BAP-03-02-2015-006. The authors acknowledge the financial support from the Turkish Scientific and Technical Research Council (TUBITAK) under 2211/C program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Salman, H.E., Yazicioglu, Y. Experimental and numerical investigation on soft tissue dynamic response due to turbulence-induced arterial vibration. Med Biol Eng Comput 57, 1737–1752 (2019). https://doi.org/10.1007/s11517-019-01995-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11517-019-01995-y