Abstract
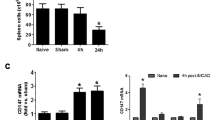
The delayed immune response to stroke is responsible for the increased neural injury that continues to occur after the initial ischemic event. This delayed immune response has been linked to the spleen, as splenectomy prior to middle cerebral artery occlusion (MCAO) is neuroprotective. Interferon gamma (IFNγ) is linked to the splenic response, which enhances neural injury following MCAO. IFNγ activates the expression of the inflammatory chemokine interferon-inducible protein 10 (IP-10). This study was designed to determine the role of IFNγ signaling in the inflammatory response following MCAO. Expression of IP-10 increased in the brain and the spleen following MCAO. Splenectomy inhibited the increase of IP-10 in the brain post-MCAO, while recombinant IFNγ administration to splenectomized rats returned IP-10 levels in the brain to levels found in rats after MCAO only. Systemic administration of an IFNγ neutralizing antibody to MCAO-treated rats reduced infarct volume and IP-10 levels in the brain. T cell infiltration was reduced in the MCAO-damaged brains of IFNγ antibody-treated animals relative to those that received isotype control antibodies. Additionally, inhibiting IFNγ signaling with splenectomy or an IFNγ neutralizing antibody blocked the induction of IP-10 expression and decreased neurodegeneration following MCAO. Targeting this pro-inflammatory pathway following stroke could be a promising stroke therapeutic.








Similar content being viewed by others
References
Ajmo CT Jr, Vernon DO, Collier L, Pennypacker KR, Cuevas J (2006) Sigma receptor activation reduces infarct size at 24 h after permanent middle cerebral artery occlusion in rats. Curr Neurovasc Res 3(2):89–98
Ajmo CT Jr, Vernon DO, Collier L, Hall AA, Garbuzova-Davis S, Willing A, Pennypacker KR (2008) The spleen contributes to stroke-induced neurodegeneration. J Neurosci Res 86:2227–2234
Boehm U, Klamp T, Groot M, Howard JC (1997) Cellular responses to interferon-gamma. Annu Rev Immunol 15:749–795
Das M, Leonardo CC, Rangooni S, Mohapatra SS, Mohapatra S, Pennypacker KR (2011) Lateral fluid percussion injury of the brain induces CCL20 inflammatory chemokine expression in rats. J Neuroinflammation 8:148
Duckworth EA, Butler TL, De Mesquita D, Collier SN, Collier L, Pennypacker KR (2005) Temporary focal ischemia in the mouse: technical aspects and patterns of Fluoro-Jade evident neurodegeneration. Brain Res 1042(1):29–36
Groom JR, Luster AD (2011) CXCR3 ligands: redundant, collaborative and antagonistic functions. Immunol Cell Biol 89(2):207–215
Hall AA, Guyer AG, Leonardo CC, Ajmo CT Jr, Collier LA, Willing AE, Pennypacker KR (2009) Human umbilical cord blood cells directly suppress ischemic oligodendrocyte cell death. J Neurosci Res 87(2):333–341
Hurn PD, Subramanian S, Parker SM, Afentoulis ME, Kaler LJ, Vandenbark AA, Offner H (2007) T- and B-cell-deficient mice with experimental stroke have reduced lesion size and inflammation. J Cereb Blood Flow Metab 27(11):1798–1805
Jiang H, Meng F, Li W, Tong L, Qiao H, Sun X (2007) Splenectomy ameliorates acute multiple organ damage induced by liver warm ischemia reperfusion in rats. Surgery 141(1):32–40
Jin R, Zhu X, Liu L, Nanda A, Granger DN, Li G (2013) Simvastatin attenuates stroke-induced splenic atrophy and lung susceptibility to spontaneous bacterial infection in mice. Stroke 44(4):1135–1143
Lee ST, Chu K, Jung KH, Kim SJ, Kim DH, Kang KM, Hong NH, Kim JH, Ban JJ, Park HK, Kim SU, Park CG, Lee SK, Kim M, Roh JK (2008) Anti-inflammatory mechanism of intravascular neural stem cell transplantation in haemorrhagic stroke. Brain 131(Pt 3):616–629
Leonardo CC, Pennypacker KR (2011) The splenic response to ischemic stroke: what have we learned from rodent models? Transl Stroke Res. doi:10.1007/s12975-0111-0075-3, e pub April
Leuschner F, Panizzi P, Chico-Calero I, Lee WW, Ueno T, Cortez-Retamozo V, Waterman P, Gorbatov R, Marinelli B, Iwamoto Y, Chudnovskiy A, Figueiredo JL, Sosnovik DE, Pittet MJ, Swirski FK, Weissleder R, Nahrendorf M (2010) Angiotensin-converting enzyme inhibition prevents the release of monocytes from their splenic reservoir in mice with myocardial infarction. Circ Res 107(11):1364–1373
Li M, Li F, Luo C, Shan Y, Zhang L, Qian Z, Zhu G, Lin J, Feng H (2011) Immediate splenectomy decreases mortality and improves cognitive function of rats after severe traumatic brain injury. J Trauma 71(1):141–147
Liesz A, Suri-Payer E, Veltkamp C, Doerr H, Sommer C, Rivest S, Giese T, Veltkamp R (2009) Regulatory T cells are key cerebroprotective immunomodulators in acute experimental stroke. Nat Med 15(2):192–199
Liesz A, Zhou W, Mracsko E, Karcher S, Bauer H, Schwarting S, Sun L, Bruder D, Stegemann S, Cerwenka A, Sommer C, Dalpke AH, Veltkamp R (2011) Inhibition of lymphocyte trafficking shields the brain against deleterious neuroinflammation after stroke. Brain 134(Pt 3):704–720
Loetscher P, Pellegrino A, Gong JH, Mattioli I, Loetscher M, Bardi G, Baggiolini M, Clark-Lewis I (2001) The ligands of CXC chemokine receptor 3, I-TAC, Mig, and IP10, are natural antagonists for CCR3. J Biol Chem 276(5):2986–2991
Longa E, Weinstein P, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91
Luster AD (2002) The role of chemokines in linking innate and adaptive immunity. Curr Opin Immunol 14(1):129–135
Offner H, Subramanian S, Parker SM, Afentoulis ME, Vandenbark AA, Hurn PD (2006) Experimental stroke induces massive, rapid activation of the peripheral immune system. J Cereb Blood Flow Metab 26(5):654–665
Okuaki Y, Miyazaki H, Zeniya M, Ishikawa T, Ohkawa Y, Tsuno S, Sakaguchi M, Hara M, Takahashi H, Toda G (1996) Splenectomy-reduced hepatic injury induced by ischemia/reperfusion in the rat. Liver 16(3):188–194
Ostrowski R, Schulte R, Nie Y, Ling T, Lee T, Manaenko A, Gridley D, Zhang J (2012) Acute splenic irradiation reduces brain injury in the rat focal ischemic stroke model. Transl Stroke Res 3:473–481. doi:10.1007/s12975-012-0206-5
Savas MC, Ozguner M, Ozguner IF, Delibas N (2003) Splenectomy attenuates intestinal ischemia-reperfusion-induced acute lung injury. J Pediatr Surg 38(10):1465–1470
Schmued LC, Albertson C, Slikker W Jr (1997) Fluoro-Jade: a novel fluorochrome for the sensitive and reliable histochemical localization of neuronal degeneration. Brain Res 751(1):37–46
Seifert HA, Hall AA, Chapman CB, Collier LA, Willing AE, Pennypacker KR (2012a) A transient decrease in spleen size following stroke corresponds to splenocyte release into systemic circulation. J NeuroImmune Pharm 7(4):1017–1024. doi:10.1007/s11481-012-9406-8
Seifert HA, Leonardo CC, Hall AA, Rowe DD, Collier LA, Benkovic SA, Willing AE, Pennypacker KR (2012b) The spleen contributes to stroke induced neurodegeneration through interferon gamma signaling. Metab Brain Dis 27(2):131–141
Walker PA, Shah SK, Jimenez F, Gerber MH, Xue H, Cutrone R, Hamilton JA, Mays RW, Deans R, Pati S, Dash PK, Cox CS Jr (2010) Intravenous multipotent adult progenitor cell therapy for traumatic brain injury: preserving the blood brain barrier via an interaction with splenocytes. Exp Neurol 225(2):341–352
Yilmaz G, Arumugam TV, Stokes KY, Granger DN (2006) Role of T lymphocytes and interferon-gamma in ischemic stroke. Circulation 113(17):2105–2112
Acknowledgments
The authors would like to thank the Lisa Muma Weitz Laboratory for Advanced Microscopy & Cell Imaging at the University of South Florida for assistance with obtaining the confocal images. This research was funded by National Institutes of Health – Neurological Disease and Stroke 1-R01-NS052839.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seifert, H.A., Collier, L.A., Chapman, C.B. et al. Pro-Inflammatory Interferon Gamma Signaling is Directly Associated with Stroke Induced Neurodegeneration. J Neuroimmune Pharmacol 9, 679–689 (2014). https://doi.org/10.1007/s11481-014-9560-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-014-9560-2