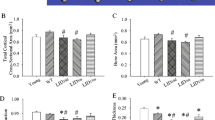
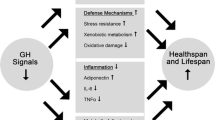
Abstract
Neuromusculoskeletal (physical) frailty is an aging-attributable biomedical issue of extremely high import, from both public health and individual perspectives. Yet, it is rarely studied in nonhuman research subjects and very rarely studied in animals with extended longevity. In an effort to address this relatively neglected area, we have conducted a longitudinal investigation of the neuromusculoskeletal healthspan in mice with two senescence-slowing interventions: growth hormone (GH) resistance, produced by GH receptor “knockout” (GHR-KO), and caloric restriction (CR). We report marked improvements in the retention of strength, balance, and motor coordination by the longevity-conferring GHR/BP gene disruption, CR regimen, or a combination of the two. Specifically, GHR-KO mice exhibit superior grip strength, balance, and motor coordination at middle age, and CR mice display superior grip strength at middle age. The advantageous effects established by middle-age are more pronounced in old-age, and these robust alterations are, generally, not gender-specific. Thus, we show that genetic and/or dietary interventions that engender longevity are also beneficial for the retention of neuromusculoskeletal health and functionality. The translational knowledge to be gained from subsequent molecular or histological investigations of these models of preserved functionality and decelerated senescence is potentially relevant to the efforts to reduce the specter of fear, falls, fracture, and frailty in the elderly.




Similar content being viewed by others
References
Abbatecola AM, Paolisso G (2008) Is there a relationship between insulin resistance and frailty syndrome? Curr Pharm Des 14(4):405–410
Ahmed N, Mandel R, Fain MJ (2007) Frailty: an emerging geriatric syndrome. Am J Med 120(9):748–753
Arum O, Bonkowski MS, Rocha JS, Bartke A (2009) The growth hormone receptor gene-disrupted mouse fails to respond to an intermittent fasting diet. Aging Cell 8(6):756–760
Avila-Funes JA, Amieva H, Barberger-Gateau P, Le Goff M, Raoux N, Ritchie K, Carrière I, Tavernier B, Tzourio C, Gutiérrez-Robledo LM, Dartigues JF (2009) Cognitive impairment improves the predictive validity of the phenotype of frailty for adverse health outcomes: the three-city study. J Am Geriatr Soc 57(3):453–461
Bartke A (2005) Insulin resistance and cognitive aging in long-lived and short-lived mice. J Gerontol A Biol Sci Med Sci 60(1):133–134
Berryman DE, List EO, Palmer AJ, Chung MY, Wright-Piekarski J, Lubbers E, O'Connor P, Okada S, Kopchick JJ (2010) Two-year body composition analyses of long-lived GHR null mice. J Gerontol A Biol Sci Med Sci 65(1):31–40
Bohannon RW (2008) Hand-grip dynamometry predicts future outcomes in aging adults. J Geriatr Phys Ther 31(1):3–10
Bonkowski MS, Pamenter RW, Rocha JS, Masternak MM, Panici JA, Bartke A (2006a) Long-lived growth hormone receptor knockout mice show a delay in age-related changes of body composition and bone characteristics. J Gerontol A Biol Sci Med Sci 61(6):562–567
Bonkowski MS, Rocha JS, Masternak MM, Al Regaiey KA, Bartke A (2006b) Targeted disruption of growth hormone receptor interferes with the beneficial actions of calorie restriction. Proc Natl Acad Sci USA 103(20):7901–7905
Coschigano KT, Clemmons D, Bellush LL, Kopchick JJ (2000) Assessment of growth parameters and life span of GHR/BP gene-disrupted mice. Endocrinology 141(7):2608–2613
Coschigano KT, Holland AN, Riders ME, List EO, Flyvbjerg A, Kopchick JJ (2003) Deletion, but not antagonism, of the mouse growth hormone receptor results in severely decreased body weights, insulin, and insulin-like growth factor I levels and increased life span. Endocrinology 144(9):3799–3810
Crawley JN (2007) What’s wrong with my mouse? Behavioral phenotyping of transgenic and knockout mice, 2nd edn. Wiley, Hoboken
De Laet CE, Pols HA (2000) Fractures in the elderly: epidemiology and demography. Baillieres Best Pract Res Clin Endocrinol Metab 14(2):171–179
Dennison E, Cooper C (2000) Epidemiology of osteoporotic fractures. Horm Res 54(Suppl 1):58–63
Ferrucci L, Cavazzini C, Corsi A, Bartali B, Russo CR, Lauretani F, Ferrucci L, Cavazzini C, Corsi AM, Bartali B, Russo CR, Lauretani F, Bandinelli S, Bandinelli S, Guralnik JM (2002) Biomarkers of frailty in older persons. J Endocrinol Invest 25(10 Suppl):10–15
Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, McBurnie MA (2001) Cardiovascular Health Study Collaborative Research Group. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 56(3):M146–M156
Glantz SA (2002) How to summarize data. In: Primer of biostatistics, 5th edn. McGraw-Hill, New York
Graber TG, Ferguson-Stegall L, Kim JH, Thompson LV (2013) C57BL/6 neuromuscular healthspan scoring system. J Gerontol A Biol Sci Med Sci, in press
Herndon LA, Schmeissner PJ, Dudaronek JM, Brown PA, Listner KM, Sakano Y, Paupard MC, Hall DH, Driscoll M (2002) Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans. Nature 419(6909):808–814
Ikeno Y, Hubbard GB, Lee S, Cortez LA, Lew CM, Webb CR, Berryman DE, List EO, Kopchick JJ, Bartke A (2009) Reduced incidence and delayed occurrence of fatal neoplastic diseases in growth hormone receptor/binding protein knockout mice. J Gerontol A Biol Sci Med Sci 64(5):522–529
Kanapuru B, Ershler WB (2009) Inflammation, coagulation, and the pathway to frailty. Am J Med 122(7):605–613
Kandel ER, Schwartz JH, Jessell TM (1991) Aging of the brain: dementia. In: Principles of neural science, 3rd edn. Appleton & Lange, Norwalk, p 974–983
Kenyon CJ (2010) The genetics of ageing. Nature 464(7288):504–512, Erratum in: Nature. 2010 Sep 30; 467(7315):622
Kinney BA, Coschigano KT, Kopchick JJ, Steger RW, Bartke A (2001) Evidence that age-induced decline in memory retention is delayed in growth hormone resistant GH-R-KO (Laron) mice. Physiol Behav 72(5):653–660
Kinney-Forshee BA, Kinney NE, Steger RW, Bartke A (2004) Could a deficiency in growth hormone signaling be beneficial to the aging brain? Physiol Behav 80(5):589–594
Kinsella K, Wan H (2009) U.S. Census Bureau, International Population Reports, P95/09-1, An aging world: 2008. U.S. Government Printing Office, Washington, DC. (http://www.census.gov/prod/2009pubs/p95-09-1.pdf). Accessed June 2009
Kirkwood TB, Finch CE (2002) Ageing: the old worm turns more slowly. Nature 419(6909):794–795
Ko FC (2011) The clinical care of frail, older adults. Clin Geriatr Med 27(1):89–100
Landi F, Abbatecola AM, Provinciali M, Corsonello A, Bustacchini S, Manigrasso L, Cherubini A, Bernabei R, Lattanzio F (2010) Moving against frailty: does physical activity matter? Biogerontology 11(5):537–545
Lang PO, Michel JP, Zekry D (2009) Frailty syndrome: a transitional state in a dynamic process. Gerontology 55(5):539–549
Levers MJ, Estabrooks CA, Ross Kerr JC (2006) Factors contributing to frailty: literature review. J Adv Nurs 56(3):282–291
Liu CK, Fielding RA (2011) Exercise as an intervention for frailty. Clin Geriatr Med 27(1):101–110
Lupu F, Terwilliger JD, Lee K, Segre GV, Efstratiadis A (2001) Roles of growth hormone and insulin-like growth factor 1 in mouse postnatal growth. Dev Biol 229(1):141–162
Marin RV, Pedrosa MA, Moreira-Pfrimer LD, Matsudo SM, Lazaretti-Castro M (2010) Association between lean mass and handgrip strength with bone mineral density in physically active postmenopausal women. J Clin Densitom 13(1):96–101
Marzetti E, Leeuwenburgh C (2006) Skeletal muscle apoptosis, sarcopenia and frailty at old age. Exp Gerontol 41(12):1234–1238
McCay CM, Crowell MF, Maynard LA (1935) The effect of retarded growth upon the length of life span and upon the ultimate body size. Nutrition 5(3):155–171, discussion 172
Mercken EM, Carboneau BA, Krzysik-Walker SM, de Cabo R (2011) Of mice and men: the benefits of caloric restriction, exercise, and mimetics. Ageing Res Rev 11(3):390–398
Miller RA (2009) “Dividends” from research on aging—can biogerontologists, at long last, find something useful to do? J Gerontol A Biol Sci Med Sci 64(2):157–160
Miller RA, Burke D, Nadon N (1999) Announcement: four-way cross mouse stocks: a new, genetically heterogeneous resource for aging research. J Gerontol A Biol Sci Med Sci 54(8):B358–B360
Moreschi C (1909) Ztschr. f. Immunitätsforsch. u. exper. Therap 2:651
Olshansky SJ, Perry D, Miller RA, Butler RN (2007) Pursuing the longevity dividend: scientific goals for an aging world. Ann N Y Acad Sci 1114:11–13
Parks RJ, Fares E, Macdonald JK, Ernst MC, Sinal CJ, Rockwood K, Howlett SE (2012) A procedure for creating a frailty index based on deficit accumulation in aging mice. J Gerontol A Biol Sci Med Sci 67(3):217–227
Rantanen T (2003) Muscle strength, disability and mortality. Scand J Med Sci Sports 13(1):3–8
Roth LW, Polotsky AJ (2012) Can we live longer by eating less? A review of caloric restriction and longevity. Maturitas 71(4):315–319
Rous P (1914) The influence of diet on transplanted and spontaneous mouse tumors. J Exp Med 20(5):433–451
Strawbridge WJ, Wallhagen MI, Cohen RD (2002) Successful aging and well-being: self-rated compared with Rowe and Kahn. Gerontologist 42(6):727–733
Thorner MO (2009) Statement by the Growth Hormone Research Society on the GH/IGF-I axis in extending health span. J Gerontol A Biol Sci Med Sci 64(10):1039–1044
Topinková E (2008) Aging, disability and frailty. Ann Nutr Metab 52(Suppl 1):6–11
Walston J, Hadley EC, Ferrucci L, Guralnik JM, Newman AB, Studenski SA, Ershler WB, Harris T, Fried LP (2006) Research agenda for frailty in older adults: toward a better understanding of physiology and etiology: summary from the American Geriatrics Society/National Institute on Aging Research Conference on Frailty in Older Adults. J Am Geriatr Soc 54(6):991–1001
Warner HR, Sierra F (2009) The longevity dividend: why invest in basic aging research? Can J Aging 28(4):391–394, French 395-8
Weiss CO (2011) Frailty and chronic diseases in older adults. Clin Geriatr Med 27(1):39–52
Zhou Y, Xu BC, Maheshwari HG, He L, Reed M, Lozykowski M, Okada S, Cataldo L, Coschigamo K, Wagner TE, Baumann G, Kopchick JJ (1997) A mammalian model for Laron syndrome produced by targeted disruption of the mouse growth hormone receptor/binding protein gene (the Laron mouse). Proc Natl Acad Sci USA 94(24):13215–13220
Acknowledgments
We thank Lisa Cox for the technical assistance, Steve Sandstrom for the copyediting, and Ravneet Boparai, Ph.D., for the photographic assistance. This work was supported by the National Institute on Aging Grants AG19899, U19 AG023122, and 3R01AG019899-07S1, as well as a Senior Scholar Award in Aging from The Ellison Medical Foundation, and The Glenn Foundation for Medical Research; this work was vitally supported by a grant from the Center for Alzheimer’s Disease and Related Disorders at The Southern Illinois University, and the authors would especially like to thank Thomas A. Ala, M.D., and Robert G. Struble, Ph.D.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Table 1
(DOC 51 kb)
Supplemental Table 2
(DOC 42 kb)
About this article
Cite this article
Arum, O., Rickman, D.J., Kopchick, J.J. et al. The slow-aging growth hormone receptor/binding protein gene-disrupted (GHR-KO) mouse is protected from aging-resultant neuromusculoskeletal frailty. AGE 36, 117–127 (2014). https://doi.org/10.1007/s11357-013-9551-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-013-9551-x