Abstract
Purpose
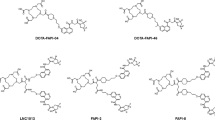
The cystine transporter, system xC-, plays a crucial role in sustaining redox homeostasis and is reported to be overexpressed in several cancer subtypes. 5-[18F]Fluoroaminosuberic acid ([18F]FASu) is a novel positron emission tomography (PET) tracer, which exhibits specific uptake via system xC-. [18F]FASu synthesis by the commonly used Kryptofix 2.2.2/K2CO3-facilitated fluorination method results in four diastereomers, as a result of 2 chiral centers at positions 2- and 5- of the tracer. We recently reported the synthesis of the optically pure 2S-[18F]FASu from chiral precursors. Our preliminary results indicated preferential uptake of the 2S-isomer by tumor cells compared to 2R-[18F]FASu. Few studies have investigated the biodistribution of chiral 18F-labeled amino acids. The aim of this study was to evaluate the imaging utility and biodistribution of the 5-position diastereomers as well as the racemic (2S,5R/S-) mixture in three different tumor models.
Procedures
In vitro tracer uptake experiments and Western blotting were performed in breast cancer (MDA-MB-231), glioblastoma (U-87), and prostate (PC-3) cancer cell lines. PET imaging and biodistribution studies were conducted in xenograft-bearing immunocompromised Rag2M female mice.
Results
All three tracer conformations allowed for the visualization of tumor xenografts at 1 h (for U-87 and PC-3 tumors) or 2 h (in the case of MDA-MB-231 xenografts) post-injection, with the racemate (2S,5R/S-) displaying similar image contrast as compared to the 5- position diastereomers and the 2S,5S-[18F]FASu conformation exhibiting relatively higher contrast for imaging U-87 and PC-3 xenografts. Tumor uptake of the isomers was blocked by an excess of the non-radioactive standard, aminosuberic acid (ASu), confirming target specificity. All three isomers were excreted via the renal pathway. Biodistribution analyses showed that PC-3 tumors had the highest tracer uptake, and the accumulation (%ID/g) of the 2S,5R/S-, 2S,5S-, and 2S,5R- isomers was 9.19 ± 1.14, 8.00 ± 1.41, and 7.16 ± 2.13 at 1 h post-injection, respectively. This gave corresponding tumor-to-muscle ratios of 33.68 ± 9.52, 31.42 ± 4.54, and 25.33 ± 4.97, respectively.
Conclusion
Our data suggest that pure 2S-[18F]FASu can be used to noninvasively image system xC- in a variety of cancers, either as the racemic mixture (2S,5R/S-) or optically pure form. Furthermore, this work shows potential utility of [18F]FASu for detection of glioblastoma and prostate cancer.






Similar content being viewed by others
References
Nakanishi T, Tamai I (2011) Solute carrier transporters as targets for drug delivery and pharmacological intervention for chemotherapy. J Pharm Sci 100:3731–3750
Timmerman LA, Holton T, Yuneva M et al (2013) Glutamine sensitivity analysis identifies the xCT antiporter as a common triple negative breast tumor therapeutic target. Cancer Cell 24:450–465
Ganapathy V, Thangaraju M, Prasad RD (2009) Nutrient transporters in cancer: Relevance to Warburg hypothesis and beyond. Pharmacol Ther 121:29–40
Wang Q, Holst J (2015) L-type amino acid transport and cancer: targeting the mTORC1 pathway to inhibit neoplasia. Am J Cancer Res 5:1281–1294
Altan B, Kaira K, Watanabe A et al (2018) Relationship between LAT1 expression and resistance to chemotherapy in pancreatic ductal adenocarcinoma. Cancer Chemother Pharmacol 81:141–153
Toyoda M, Kaira K, Ohshima Y et al (2014) Prognostic significance of amino-acid transporter expression (LAT1, ASCT2, and xCT) in surgically resected tongue cancer. Br J Cancer 110:2506–2513
Piroth MD, Pinkawa M, Richard H et al (2011) Prognostic value of early [ 18F]fluoroethyltyrosine positron emission tomography after radiochemotherapy in glioblastoma multiforme. Int J Radiat Oncol Biol Phys 80:176–184
Yanagisawa N, Ichinoe M, Mikami T, Nakada N, Hana K, Koizumi W, Endou H, Okayasu I (2012) High expression of L-type amino acid transporter 1 (LAT1) predicts poor prognosis in pancreatic ductal adenocarcinomas. J Clin Pathol 65:1019–1023
McConathy J, Goodman M (2008) Non-natural amino acids for tumor imaging using positron emission tomography and single photon emission computed tomography. Cancer Metastasis Rev 27:555–557
Huang C, McConathy J (2013) Radiolabeled amino acids for oncologic imaging. J Nucl Med 54:1007–1010
Qu W, Zha Z, Ploessl K et al (2011) Synthesis of optically pure 4-fluoro-glutamines as potential metabolic imaging agents for tumors. J Am Chem Soc 133:1122–1133
Liu F, Xu X, Zhu H, Zhang Y, Yang J, Zhang L, Li N, Zhu L, Kung HF, Yang Z (2018) PET imaging of 18F-(2S,4R)4-fluoroglutamine accumulation in breast cancer: from xenografts to patients. Mol Pharm 15:3448–3455
Urakami T, Sakai K, Asai T et al (2009) Evaluation of O-[18F]fluoromethyl-d-tyrosine as a radiotracer for tumor imaging with positron emission tomography. Nucl Med Biol 36:295–303
Webster JM, Morton CA, Johnson BF et al (2014) Functional Imaging of oxidative stress with a novel PET imaging agent, 18F-5-fluoro-L-aminosuberic acid. J Nucl Med 55:657–664
Yang H, Jenni S, Čolović M et al (2017) 18F-5-Fluoroaminosuberic acid as a potential tracer to gauge oxidative stress in breast cancer models. J Nucl Med 58:367–373
Čolović M, Yang H, Merkens H et al (2019) Non-invasive use of positron emission tomography to monitor diethyl maleate and radiation-induced changes in system xC− activity in breast cancer. Mol Imaging Biol. https://doi.org/10.1007/s11307-019-01331-8
Conrad M, Sato H (2012) The oxidative stress-inducible cystine/glutamate antiporter, system xc-: cystine supplier and beyond. Amino Acids 42:231–246
Bridges RJ, Natale NR, Patel SA (2012) System xc- cystine/glutamate antiporter: an update on molecular pharmacology and roles within the CNS. Br J Pharmacol 165:20–34
Ishii T, Sato H, Miura K, Sagara J, Bannai S (1992) Induction of cystine transport activity by stress. Ann N Y Acad Sci 663:497–498
Sasaki H, Sato H, Kuriyama-Matsumura K et al (2002) Electrophile response element-mediated induction of the cystine/glutamate exchange transporter gene expression. J Biol Chem 277:44765–44771
Yang H, Tam B, Čolović M et al (2017) Addressing chirality in the structure and synthesis of [18F]5-fluoroaminosuberic acid ([18F]FASu). Chem Eur J 23:11100–11107
Čolović M, Rousseau E, Zhang Z et al (2018) Synthesis and evaluation of an 18F-labeled boramino acid analog of aminosuberic acid for PET imaging of the antiporter system xC−. Bioorg Med Chem Lett 28:3579–3584
Heiss P, Mayer S, Herz M et al (1999) Investigation of transport mechanism and uptake kinetics of O-(2-[18F]fluoroethyl)-L-tyrosine in vitro and in vivo. J Nucl Med 40:1367–1373
Langen KJ, Hamacher K, Bauer D et al (2005) Preferred stereoselective transport of the D-isomer of cis-4-[18F]fluoro-proline at the blood-brain barrier. J Cereb Blood Flow Metab 25:607–616
Tsukada T, Sato K, Fukumoto D, Kakiuchi T (2006) Evaluation of D-isomers of O-18F-fluoromethyl, O-18F-fluoroethyl and O-18F-fluoropropyl tyrosine as tumour imaging agents in mice. Eur J Nucl Med Mol Imaging 33:1017–1024
Kersemans V, Cornelissen B, Kersemans K et al (2006) 123/125I-labelled 2-iodo-L-phenylalanine and 2-iodo-D-phenylalanine: comparative uptake in various tumour types and biodistribution in mice. Eur J Nucl Med Mol Imaging 33:919–927
Bauwens M, Keyaerts M, Lahoutte T, Kersemans K, Caveliers V, Bossuyt A, Mertens J (2007) Intra-individual comparison of the human biodistribution and dosimetry of the D and L isomers of 2-[123I]iodo-phenylalanine. Nucl Med Commun 28:823–828
Martarello L, McConathy J, Camp VM et al (2002) Synthesis of syn- and anti-1-amino-3-[18F]fluoromethyl-cyclobutane-1-carboxylic acid (FMACBC), potential PET ligands for tumor detection. J Med Chem 45:2250–2259
Zhang Z, Liu S, Tang Z et al (2018) Radiosynthesis and preliminary biological evaluation of the 2-[18F]fluoropropionic acid enantiomers for tumor PET imaging. J Radioanal Nucl Chem 316:153–159
Acknowledgments
We thank TRIUMF TR13 cyclotron operators: David Prevost, Linda Graham, and Samuel Varah for their technical assistance and Dr. Aron Roxin for his constructive feedback which helped finalize this manuscript.
Funding
This study is financially supported by the CIHR (201403COP, 329895). TRIUMF receives federal funding via a contribution agreement with the National Research Council of Canada. M.Č. is supported by the NSERC CREATE IsoSiM fellowship, grant no. 448110, competition year 2014. This work was supported in part by the BC Leading Edge Endowment Fund.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All in vivo experiments were conducted in accordance with the guidelines established by the Canadian Council on Animal Care and were approved by the Animal Ethics Committee of the University of British Columbia.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 360 kb)
Rights and permissions
About this article
Cite this article
Čolović, M., Yang, H., Merkens, H. et al. The Effect of Chirality on the Application of 5-[18F]Fluoro-Aminosuberic Acid ([18F]FASu) for Oxidative Stress Imaging. Mol Imaging Biol 22, 873–882 (2020). https://doi.org/10.1007/s11307-019-01450-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-019-01450-2