Abstract
Purpose
A significant positive correlation has been observed between ketone body availability and their uptake by tumor cells. Our objective was to evaluate [11C]acetoacetate as a potential tracer of ketone body utilization by breast and prostate tumors and to compare it with [11C]acetate.
Methods
Biodistribution studies were performed with [11C]acetoacetate and [11C]acetate in mice bearing breast or prostate tumors. The percentage of the injected dose accumulated per gram of tissue was determined. These results were complemented by dynamic positron emission tomography (PET) imaging of the radiotracer uptake and dosimetry calculations.
Results
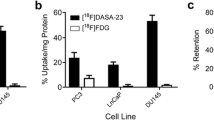
[11C]Acetoacetate uptake was optimal between 5 and 30 min, with maximal uptake of 2.72, 2.42, 2.54, and 2.19% injected dose (%ID)/g for MC7-L1, MC4-L2, PC3, and LN-CaP tumors respectively. Tumor retention for [11C]acetoacetate tended to be higher than [11C]acetate, but this did not reach statistical significance. [11C]Acetate uptake was reached within 15 min with optimal uptake of 1.25, 2.30, and 0.96%ID/g for MC7-L1, MC4-L2, and PC-3 tumors, respectively.
Conclusions
We observed a moderate uptake of [11C]acetoacetate in breast and prostate tumors with low background activity due to rapid elimination of this tracer. Further studies are warranted to determine if this tracer can detect slow-growing breast and prostate cancers in the clinical setting.




Similar content being viewed by others
References
Gambhir SS, Czernin J, Schwimmer J, Silverman DH, Coleman RE, Phelps ME (2001) A tabulated summary of the FDG PET literature. J Nucl Med 42(5 Suppl):1S–93S
Quon A, Gambhir SS (2005) FDG-PET and beyond: molecular breast cancer imaging. J Clin Oncol 23(8):1664–1673
Oyama N, Akino H, Kanamaru H et al (2002) 11C-acetate PET imaging of prostate cancer. J Nucl Med 43:181–186
Kumar R, Alavi A (2004) Fluorodeoxyglucose-PET in the management of breast cancer. Radiol Clin North Am 42:1113–1122
Kang DE, White RL, Zuger JH, Sasser HC, Teigland CM (2004) Clinical use of fluorodeoxyglucose F18 positron emission tomography for detection of renal cell carcinoma. J Urol 171:1806–1809
Oyama N, Miller TR, Dehdashti F, Siegel BA et al (2003) 11C-acetate PET imaging of prostate cancer: detection of recurrent disease at PSA relapse. J Nucl Med 44:549–555
Fricke E, Machtens S, Hofmann M et al (2003) Positron emission tomography with 11C-acetate and 18F-FDG in prostate cancer patients. Eur J Nucl Med Mol Imaging 30:607–611
Seltzer MA, Jahan SA, Sparks R et al (2004) Radiation dose estimates in humans for 11C-acetate whole-body PET. J Nucl Med 45:1233–1236
Tremblay S, Ouellet R, Rodrigue S, Langlois R, Benard F, Cunnane SC (2007) Automated Synthesis of 11 C-Acetoacetic Acid, a Key Alternate Brain Fuel to Glucose. Appl Radiat Isotopes 65(8):934–940
Voet D, Voet JG (1998) Métabolisme des lipides. In: Voet D, Voet JG (eds) Biochimie. De Boeck Université s.a., Paris, pp 662–726
Sauer LA, Dauchy RT (1983) Ketone body, glucose, lactic acid, and amino acid utilization by tumors in vivo in fasted rats. Cancer Res. 43:3497–3503
Kallinowskil F, Davel S, Vaupell P, Baessler KH, Wagner K (1985) Glucose, lactate, and ketone body utilization by human mammary carcinomas in vivo. Adv Exp Med Biol 43:763–773
Fenselau A, Wallis K, Morris HP (1976) Subcellular localization of acetoacetate coenzyme A transferase in rat hepatomas. Cancer Res 36:4429–4433
Lanari C, Luthyl I, Lamb CA et al (2001) Five novel hormone-responsive cell lines derived from murine mammary ductal carcinomas: in vivo and in vitro effects of estrogens and progestins. Cancer Res 61:293–302
Pike VW, Eakins MN, Allan RM et al (1982) Preparation of [1-11C]acetate: an agent for the study of myocardial metabolism by positron emission tomography. Int J Appl Radiat Isot 33(7):505–512
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Lecomte R, Cadorette J, Rodrigue S et al (1996) Initial results from the Sherbrooke avalanche photodiode positron tomograph. IEEE Trans Nucl Sci 43:1952–1957
Stabin MG (1996) MIRDOSE: Personal computer software for internal dose assessment in nuclear medicine. J Nucl Med 37(3):538–546
Stabin MG, Richard BS, Crowe E (2005) OLINDA/EXM: The second-generation personal computer software for internal dose assessment in nuclear medicine. J Nucl Med 46(6):1023–1027
Yoshimoto M, Waki A, Yonekura Y et al (2001) Characterization of acetate metabolism in tumor cells in relation to cell proliferation: acetate metabolism in tumor cells. Nucl Med Biol 28:117–122
Acknowledgments
This project received financial support from the Canadian Foundation for Innovation, the Research Center on Aging, the Canada Research Chairs programs, and a Fonds de la recherche du Québec scholarship. The pilot PET imaging studies were supported by CIHR grant MOP-15348.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Authier, S., Tremblay, S., Dumulon, V. et al. [11C] Acetoacetate Utilization by Breast and Prostate Tumors: a PET and Biodistribution Study in Mice. Mol Imaging Biol 10, 217–223 (2008). https://doi.org/10.1007/s11307-008-0143-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-008-0143-6