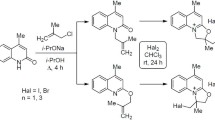
Abstract
The chemistry of 3-acetyl-4-hydroxyquinolin-2(1H)-ones has attracted increased attention in both synthetic organic and medicinal chemistry, as numerous advancements in the use of this family of important compounds seems to be of considerable value. This account summarizes the results from the literature focusing on synthesis of 3-acetyl-4-hydroxyquinolin-2(1H)-ones as well as their reactions are also discussed.
























Similar content being viewed by others
References
I.A. Bessonova, Chem. Nat. Compd. 36, 324 (2000)
K. Sharma, S. Khandelwal, R.M. Samarth, M. Kumar, J. Heterocycl. Chem. 53, 220 (2016)
E.A. Clarke, M.F. Grundon, J. Chem. Soc. 438 (1964)
M.F. Grundon, Tetrahedron 34, 143 (1978)
V. Vijayakumar, Int. J. Chemtech. Res. 9, 629 (2016)
B.T. Ngadjui, J.F. Ayafor, A.N. Bilon, B.L. Sondengam, J.D. Connolly, D.S. Rycroft, Tetrahedron 48, 8711 (1992)
I.V. Ukrainets, N.L. Bereznyakova, E.V. Mospanova, Chem. Heterocycl. Compd. 43, 856 (2007)
E. Lager, P. Andersson, J. Nilsson, I. Pettersson, E. Nielsen, J. Med. Chem. 49, 2526 (2006)
V. Cecchetti, C. Parolin, S. Moro, T. Pecere, E. Filipponi, A. Calistri, O. Tabarrini, B. Gatto, M. Palumbo, A. Fravolini, T. Giorgio Palu, J. Med. Chem. 43, 3799 (2000)
M.I. Crespo, J. Gràcia, C. Puig, A. Vega, J. Bou, J. Beleta, T. Doménech, H. Ryder, V. Segarra, J.M. Palacios, Med. Chem. Lett. 10, 2661 (2000)
C.M. Hall, H.G. Johnson, J.B. Wright, J. Med. Chem. 17, 685 (1974)
Y.F. Suen, L. Robins, B. Yang, A.S. Verkman, M.H. Nantz, M.J. Kurth, Med. Chem. Lett. 16, 537 (2006)
R.V. Davies, D.B. Yates, Prog. Med. Chem. 32, 115 (1995)
I.V. Ukrainets, S.G. Taran, O.V. Gorokhova, E.A. Taran, N.A. Jaradat, I.Y. Petukhova, Chem. Heterocycl. Compd. 36, 166 (2000)
I.V. Ukrainets, S.G. Taran, P.A. Bezuglyi, S.N. Kovalenko, A.V. Turov, N.A. Marusenko, Chem. Heterocycl. Compd. 29, 1044 (1993)
I.V. Ukrainets, A.A. Tkach, L.Y. Yang, Chem. Heterocycl. Compd. 44, 1347 (2008)
K. Arya, M. Agarwal, Med. Chem. Lett. 17, 86 (2007)
B.S. Jayashree, S. Thomas, Y. Nayak, Med. Chem. Res. 19, 193 (2010)
R. Watpade, A. Bholay, R. Toche, J. Heterocycl. Chem. 54, 3434 (2017)
J.A. Smiley, S.J. Benkovic, J. Am, Chem. Soc. 117, 3877 (1995)
E.J. O’loughlin, G.K. Sims, S.J. Traina, Biodegradation 10, 93 (1999)
S. Heeb, M.P. Fletcher, S.R. Chhabra, S.P. Diggle, P. Williams, M. Cámara, FEMS Microbiol. Rev. 35, 247 (2011)
S. Sarveswari, V. Vijayakumar, R. Siva, R. Priya, Appl. Biochem. Biotechnol. 175, 43 (2015)
M.D. Ferretti, A.T. Neto, A.F. Morel, T.S. Kaufman, E.L. Larghi, Eur. J. Med. Chem. 81, 253 (2014)
M.A. Ibrahim, H.M. Hassanin, Y.A. Alnamer, Synth. Commun. 44, 3470 (2014)
J.J. Kulagowski, R. Baker, N.R. Curtis, I.M. Mawer, A.M. Moseley, M.P. Ridgill, M. Rowley, I. Stansfield, P.D. Leeson, J. Med. Chem. 37, 1402 (1994)
N. Dodia, A. Shah, Indian J. Heterocycl. Chem. 9, 139 (1999)
M.B. Macedo, R. Kimmel, D. Urankar, M. Gazvoda, A. Peixoto, F. Cools, E. Torfs, L. Verschaeve, E.S. Lima, A. Lyčka, D. Milićević, Eur. J. Med. Chem. 138, 491 (2017)
C. Zwergel, B. Czepukojc, E. Evain-Bana, Z. Xu, G. Stazi, M. Mori, A. Patsilinakos, A. Mai, B. Botta, R. Ragno, D. Bagrel, Eur. J. Med. Chem. 134, 316 (2017)
Y.F. Chen, Y.C. Lin, S.L. Morris-Natschke, C.F. Wei, T.C. Shen, H.Y. Lin, M.H. Hsu, L.C. Chou, Y. Zhao, S.C. Kuo, K.H. Lee, Br. J. Pharmacol. 172, 1195 (2015)
D.A. Sabbaha, B. Hishmaha, K. Sweidana, S. Bardaweelb, M. AlDamenc, G.A. Sheikhaa, M.S. Mubarakc, Anticancer Agents Med. Chem. 18, 263 (2018)
D.A. Sabbah, N.A. Simms, W. Wang, Y. Dong, E.L. Ezell, M.G. Brattain, J.L. Vennerstrom, H.A. Zhong, Bioorg. Med. Chem. 20, 7175 (2012)
Y. Xia, Z.Y. Yang, P. Xia, T. Hackl, E. Hamel, A. Mauger, J.H. Wu, K.H. Lee, J. Med. Chem. 44, 3932 (2001)
H.M. Manzoor, M. Ahmad, F.A. Saddique, S. Aslam, S.G. Khan, A. Saddiqa, Afinidad 74, 228 (2017)
A. Rivkin, Y.R. Kim, M.T. Goulet, N. Bays, A.D. Hill, I. Kariv, S. Krauss, N. Ginanni, P.R. Strack, N.E. Kohl, C.C. Chung, Bioorg. Med. Chem. Lett. 16, 4620 (2006)
K.J. Hwang, Arch. Pharmacal Res. 23, 31 (2000)
A.M. MacLeod, S. Grimwood, C. Barton, L. Bristow, K.L. Saywell, G.R. Marshall, R.G. Ball, J. Med. Chem. 38, 2239 (1995)
M.M. Abdou, New Azo Disperse Dyes Derived from 3-(Hydroxyphenyl)-2-Pyrazolin-5-One, 1st (Lambert Academic Publishing, German Publisher LAP, Riga, 2013), p. 21
M.M. Abdou, Azo Disperse Dyes with 2-Pyrazolin-5-Ones for Dyeing Polyester Fabrics, 1st (Lambert Academic Publishing, German Publisher LAP, Riga, 2013), p. 30
M.M. Abdou, R.A. El-Saeed, K.M. Elattar, Z. Seferoğlu, J. Boukouvalas, Mol. Divers 20, 989 (2016)
M.M. Abdou, Arab. J. Chem. (2014). https://doi.org/10.1016/j.arabjc.2014.11.0213
M.M. Abdou, A. El Rasha, S. Bondock, Arab. J. Chem. (2015). https://doi.org/10.1016/j.arabjc.2015.06.029
M.M. Abdou, Arab. J. Chem. 10, S3955 (2017)
M.M. Abdou, Arab. J. Chem. 10, S3664 (2017)
M.M. Abdou, R.A. El-Saeed, S. Bondock, Arab. J. Chem. (2015). https://doi.org/10.1016/j.arabjc.2015.06.012
M.M. Abdou, R.A. El-Saeed, M.A. Abozeid, K.M. Elattar, E.G. Zaki, Y. Barakat, V. Ibrahim, M. Fathy, M. Amine, S. Bondock, Arab. J. Chem. (2015). https://doi.org/10.1016/j.arabjc.2015.11.004
M.M. Abdou, Arab. J. Chem. 10, S3324 (2017)
M.M. Abdou, S. Bondock, S.I. El-Desouky, M.A. Metwally, Am. J. Chem. 3, 59 (2013)
M.A. Metwally, S. Bondock, E.S.I. El-Desouky, M.M. Abdou, Coloration Technol. 129, 418 (2013)
M.M. Abdou, S. Bondock, S.I. El-Desouky, M.A. Metwally, Int. J. Mod. Org. Chem. 1, 165 (2012)
M.M. Abdou, S. Bondock, S.I. El-Desouky, M.A. Metwally, Int. J. Mod. Org. Chem. 1, 19 (2012)
M.A. Metwally, S. Bondock, S.I. El-Desouky, M.M. Abdou, J. Korean Chem. Soc. 56, 348 (2012)
M.M. Abdou, S. Bondock, S.I. El-Desouky, M.A. Metwally, Am. J. Chem. 2, 347 (2012)
M.A. Metwally, S. Bondock, S.I. El-Desouky, M.M. Abdou, J. Korean Chem. 56, 82 (2012)
W. Stadlbauers, G. Hojas, J. Heterocycl. Chem. 41, 681 (2004)
S. Sarveswari, T.K. Raja, R. Vijayaraghavan, T. Narasimhamurthy, Acta Cryst. E 63, o4600 (2007)
M.M. Hassan, E.S. Othman, M. Abass, Res. Chem. Intermed. 39, 1209 (2013)
A. Detsi, V. Bardakos, J. Markopoulos, O. Igglessi-Markopoulou, J. Chem. Soc. Perkin Trans. 1, 2909 (1996)
T. Kappe, R. Aigner, P. Hohengassner, W. Stadlbauer, Adv. Synth. Catal. 336, 596 (1994)
M.A. Ibrahim, H.M. Hassanin, M. Abass, S. Badran, Arkivoc 4, 424 (2013)
M. Abass, B.B. Mostafa, Bioorg. Med. Chem. 13, 6133 (2005)
M.M. Hassan, H.M. Hassanin, J. Heterocycl. Chem. 54, 3321 (2017)
K. Faber, T. Kappe, J. Heterocycl. Chem. 21, 1881 (1984)
I.V. Ukrainets, A.A. Tkach, L.Y. Yang, J. Heterocycl. Chem. 45, 169 (2009)
S.K. Phadtare, S.K. Kamat, G.T. Panse, Indian J. Chem., Sect B 22, 496 (1983)
T. Kappe, R. Aigner, P. Roschger, B. Schnell, W. Stadibauer, Tetrahedron 51, 12923 (1995)
M. Abass, H.A. Allimony, H. Hassan, Phosphorus Sulfur Silicon Rel. Elem. 188, 1799 (2013)
P. Roschger, W. Stadlbauer, Eur. J. Org. Chem. 1990, 821 (1990)
R.E. Bowman, A. Campbell, E.M. Tanner, J. Chem. Soc. 81, 444 (1959)
T. Razzaq, C.O. Kappe, Tetrahedron Lett. 48, 2513 (2007)
P. Roschger, W. Fiala, W. Stadlbauer, J. Heterocycl. Chem. 29, 225 (1992)
K. Proisl, S. Kafka, J. Kosmrlj, Curr. Org. Chem. 21, 1949 (2017)
S. Chimichi, M. Boccalini, M.M. Hassan, G. Viola, F. Dall’Acqua, M. Curini, Tetrahedron 62, 90 (2006)
S. Chimichi, M. Boccalini, A. Matteucci, Tetrahedron 63, 11656 (2007)
C. Mitsos, J. Petrou, O. Igglessi-Markopoulou, J. Markopoulos, J. Heterocycl. Chem. 36, 881 (1999)
G. Athanasellis, E. Gavrielatos, O. Igglessi-Markopoulou, J. Markopoulos, J. Heterocycl. Chem. 40, 645 (2003)
A.V. Manaev, I.N. Okhrimenko, K.A. Lyssenko, Russ. Chem. Bull. 57, 1734 (2008)
V.B. Kovalska, K.D. Volkova, A.V. Manaev, M.Y. Losytskyy, I.N. Okhrimenko, V.F. Traven, S.M. Yarmoluk, Dyes Pigm. 84, 159 (2010)
A.B. Tathe, N. Sekar, J. Fluoresc. 26, 471 (2016)
F.M.A.A. El-Taweel, S.Z.A. Sowellim, A.G.A. Elagamey, Bull. Chem. Soc. Jpn 68, 905 (1995)
M.M. Girges, M.A. Hanna, H.M. Hassan, E.B. Moawad, Czech Chem. Commun. 53, 3179 (1988)
F.M. El-Taweel, D.A. Ibrahim, M.A. Hanna, Boll. Chim. Farm. 140, 287 (2001)
G.V. Kalechits, V.K. Ol’khovik, I.I. Kalosha, E.D. Skakovskii, A.A. Pap, A.A. Zenyuk, Y.V. Matveenko, Russ. J. Gen. Chem. 71, 1257 (2001)
S. Sarveswari, T.K. Raja, Indian J. Heterocycl. Chem. 16, 171 (2006)
R.S. Bhupathi, B.R. Devi, P.K. Dubey, Asian J. Chem. 23, 4215 (2011)
M. Roussaki, B. Hall, S.C. Lima, A.C. da Silva, S. Wilkinson, A. Detsi, Med. Chem. Lett. 23, 6436 (2013)
M. Abass, M. Abdel-Megid, M. Hassan, Synth. Commun. 37, 329 (2007)
M.M. Hassan, O. Farouk, J. Heterocycl. Chem. 54, 3133 (2017)
S. Sarveswari, V. Vijayakumar, Arab. J. Chem. 9, S835 (2016)
R.S. Bhupathi, B.R. Devi, P.K. Dubey, Indian J. Chem., Sect B 51, 855 (2012)
M. Abdel-Megid, M. Abass, M. Hassan, J. Heterocycl. Chem. 44, 315 (2007)
F.M.A. El-Taweel, J. Heterocycl. Chem. 42, 943 (2005)
T. Kappe, R. Aigner, M. Jöbstl, P. Hohengassner, W. Stadlbauer, Heterocycl. Commun. 1, 341 (1995)
M.M. Hassan, S.M. Abdel-Kariem, T.E. Ali, Phosphorus Sulfur Silicon Rel. Elem. 192, 1290625 (2017)
B. Baruah, K. Dasu, B. Vaitilingam, A. Vanguri, S.R. Casturi, K.R. Yeleswarapu, Med. Chem. Lett. 14, 445 (2004)
T. Arasakumar, S. Mathusalini, K. Lakshmi, P.S. Mohan, A. Ata, C.H. Lin, Synth. Commun. 46, 232 (2016)
M. Sankaran, C. Kumarasamy, U. Chokkalingam, P.S. Mohan, Med. Chem. Lett. 20, 7147 (2010)
S. Latypov, A. Balandina, M. Boccalini, A. Matteucci, K. Usachev, S. Chimichi, Eur. J. Org. Chem. 2008, 4640 (2008)
J.H. Ye, K.Q. Ling, Y. Zhang, N. Li, J.H. Xu, J. Chem. Soc. Perkin Trans. 1, 2017 (1999)
D.K. Kumar, R. Rajkumar, S.P. Rajendran, Chem. Heterocycl. Compd. 52, 322 (2016)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdou, M.M., Seferoğlu, Z., Fathy, M. et al. Synthesis and chemical transformations of 3-acetyl-4-hydroxyquinolin-2(1H)-one and its N-substituted derivatives: bird’s eye view. Res Chem Intermed 45, 919–934 (2019). https://doi.org/10.1007/s11164-018-3652-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3652-1