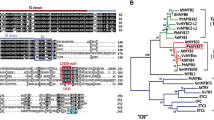
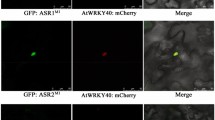
Abstract
Key message
A Kelch repeat F-box containing protein, SMALL AND GLOSSY LEAVES1 (SAGL1) regulates phenylpropanoid biosynthesis as a post-translational regulator for PAL1 (phenylalanine ammonia-lyase) and an indirect transcriptional regulator for ANTHOCYANIDIN SYNTHASE.
Abstract
Phenylpropanoid biosynthesis in plants produces diverse aromatic metabolites with important biological functions. Phenylalanine ammonia-lyase (PAL) catalyzes the first step in phenylpropanoid biosynthesis by converting l-phenylalanine to trans-cinnamic acid. Here, we report that SMALL AND GLOSSY LEAVES1 (SAGL1), a Kelch repeat F-box protein, interacts with PAL1 protein for proteasome-mediated degradation to regulate phenylpropanoid biosynthesis in Arabidopsis. Mutations in SAGL1 caused high accumulation of anthocyanins and lignin derived from the phenylpropanoid biosynthesis pathway. We found that PAL enzyme activity increased in SAGL1-defective mutants, sagl1, but decreased in SAGL1-overexpressing Arabidopsis (SAGL1OE) without changes in the transcript levels of PAL genes, suggesting protein-level regulation by SAGL1. Indeed, the levels of PAL1-GFP fusion protein were reduced when both SAGL1 and PAL1-GFP were transiently co-expressed in leaves of Nicotiana benthamiana. In addition, bimolecular fluorescence complementation analysis suggested an interaction between SAGL1 and PAL1. We also found that the transcript levels of ANTHOCYANIDIN SYNTHASE (ANS) increased in the sagl1 mutants but decreased in SAGL1OE. Our results suggest that SAGL1 regulates phenylpropanoid biosynthesis post-translationally at PAL1 and transcriptionally at ANS.









Similar content being viewed by others
References
Adams J, Kelso R, Cooley L (2000) The kelch repeat superfamily of proteins: propellers of cell function. Trends Cell Biol 10:17–24
Appelhagen I, Jahns O, Bartelniewoehner L, Sagasser M, Weisshaar B, Stracke R (2011) Leucoanthocyanidin dioxygenase in Arabidopsis thaliana: characterization of mutant alleles and regulation by MYB-BHLH-TTG1 transcription factor complexes. Gene 484:61–68. https://doi.org/10.1016/j.gene.2011.05.031
Blount JW, Korth KL, Masoud SA, Rasmussen S, Lamb C, Dixon RA (2000) Altering expression of cinnamic acid 4-hydroxylase in transgenic plants provides evidence for a feedback loop at the entry point into the phenylpropanoid pathway. Plant Physiol 122:107–116
Boerjan W, Ralph J, Baucher M (2003) Lignin biosynthesis Annu Rev Plant Biol 54:519–546. https://doi.org/10.1146/annurev.arplant.54.031902.134938
Borah P, Khurana JP (2018) The OsFBK1 E3 ligase subunit affects anther and root secondary cell wall thickenings by mediating turnover of a cinnamoyl-CoA reductase. Plant Physiol 176:2148–2165. https://doi.org/10.1104/pp.17.01733
Chang A, Lim MH, Lee SW, Robb EJ, Nazar RN (2008) Tomato phenylalanine ammonia-lyase gene family, highly redundant but strongly underutilized. J Biol Chem 283:33591–33601. https://doi.org/10.1074/jbc.M804428200
Cochrane FC, Davin LB, Lewis NG (2004) The Arabidopsis phenylalanine ammonia lyase gene family: kinetic characterization of the four PAL isoforms Phytochemistry 65:1557–1564. https://doi.org/10.1016/j.phytochem.2004.05.006
Dixon RA, Paiva NL (1995) Stress-lnduced phenylpropanoid metabolism. Plant Cell 7:1085–1097
Dixon RA, Xie DY, Sharma SB (2005) Proanthocyanidins–a final frontier in flavonoid research? New Phytol 165:9–28. https://doi.org/10.1111/j.1469-8137.2004.01217.x
Fraser CM, Chapple C (2011) The phenylpropanoid pathway in Arabidopsis. Arabidopsis Book 9:e0152. https://doi.org/10.1199/tab.0152
Gabetta B, Fuzzati N, Griffini A, Lolla E, Pace R, Ruffilli T, Peterlongo F (2000) Characterization of proanthocyanidins from grape seeds. Fitoterapia 71:162–175
Gagne JM, Downes BP, Shiu SH, Durski AM, Vierstra RD (2002) The F-box subunit of the SCF E3 complex is encoded by a diverse superfamily of genes in Arabidopsis. Proc Natl Acad Sci USA 99:11519–11524. https://doi.org/10.1073/pnas.162339999
Given NK, Venis MA, Grierson D (1988) Purification and properties of phenylalanine ammonia-lyase from strawberry fruit and its synthesis during ripening. J Plant Physiol 133:31–37. https://doi.org/10.1016/s0176-1617(88)80080-4
Herrmann KM (1995) The shikimate pathway: early steps in the biosynthesis of aromatic. Compounds Plant Cell 7:907–919. https://doi.org/10.1105/tpc.7.7.907
Hershko A, Ciechanover A (1998) The ubiquitin system. Annu Rev Biochem 67:425–479. https://doi.org/10.1146/annurev.biochem.67.1.425
Ho MS, Tsai PI, Chien CT (2006) F-box proteins: the key to protein degradation. J Biomed Sci 13:181–191. https://doi.org/10.1007/s11373-005-9058-2
Huang J et al (2010) Functional analysis of the Arabidopsis PAL gene family in plant growth, development, and response to environmental stress. Plant Physiol 153:1526–1538. https://doi.org/10.1104/pp.110.157370
Joos HJ, Hahlbrock K (1992) Phenylalanine ammonia-lyase in potato (Solanum tuberosum L.) genomic complexity, structural comparison of two selected genes and modes of expression. Eur J Biochem 204:621–629
Kao YY, Harding SA, Tsai CJ (2002) Differential expression of two distinct phenylalanine ammonia-lyase genes in condensed tannin-accumulating and lignifying cells of quaking aspen. Plant Physiol 130:796–807. https://doi.org/10.1104/pp.006262
Kim WY et al (2007) ZEITLUPE is a circadian photoreceptor stabilized by GIGANTEA in blue light. Nature 449:356–360. https://doi.org/10.1038/nature06132
Kim E-J, Lee S-H, Park C-H, Kim T-W (2018a) Functional role of BSL1 subcellular localization in brassinosteroid signaling. J Plant Biol 61:40–49. https://doi.org/10.1007/s12374-017-0363-x
Kim H, Yu S, Jung SH, Lee, Bh, Suh MC (2018b) The Arabidopsis SAGL1 E3 Ligase and ECERIFERUM3 module regulates cuticular wax biosynthesis in response to humidity. (in press)
Kumar A, Ellis BE (2001) The phenylalanine ammonia-lyase gene family in raspberry. Structure, expression, and evolution. Plant Physiol 127:230–239
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Kwon Y et al (2012) OsREL2, a rice TOPLESS homolog functions in axillary meristem development in rice inflorescence. Plant Biotechnol Rep 6:213–224. https://doi.org/10.1007/s11816-011-0214-z
Lewis NG, Yamamoto E (1990) Lignin: occurrence, biogenesis and biodegradation. Annu Rev Plant Biol 41:455–496. https://doi.org/10.1146/annurev.pp.41.060190.002323
Leyva A, Jarillo JA, Salinas J, Martinez-Zapater JM (1995) Low temperature lnduces the accumulation of phenylalanine ammonia-lyase and chalcone synthase mRNAs of Arabidopsis thaliana in a light-dependent. Manner Plant Physiol 108:39–46
Majee M et al (2018) KELCH F-BOX protein positively influences Arabidopsis seed germination by targeting PHYTOCHROME-INTERACTING FACTOR1 P Natl. Acad Sci USA 115:E4120–E4129. https://doi.org/10.1073/pnas.1711919115
Martin K, Kopperud K, Chakrabarty R, Banerjee R, Brooks R, Goodin MM (2009) Transient expression in Nicotiana benthamiana fluorescent marker lines provides enhanced definition of protein localization, movement and interactions in planta. Plant J 59:150–162. https://doi.org/10.1111/j.1365-313X.2009.03850.x
Mele G, Ori N, Sato Y, Hake S (2003) The knotted1-like homeobox gene BREVIPEDICELLUS regulates cell differentiation by modulating metabolic pathways. Genes Dev 17:2088–2093. https://doi.org/10.1101/gad.1120003
Moura JC, Bonine CA, de Oliveira Fernandes Viana J, Dornelas MC, Mazzafera P (2010) Abiotic and biotic stresses and changes in the lignin content and composition in plants. J Integr Plant Biol 52:360–376. https://doi.org/10.1111/j.1744-7909.2010.00892.x
Nakagawa T et al (2007) Improved gateway binary vectors: high-performance vectors for creation of fusion constructs in transgenic analysis of plants. Biosci Biotechnol Biochem 71:2095–2100. https://doi.org/10.1271/bbb.70216
Nelson DC, Lasswell J, Rogg LE, Cohen MA, Bartel B (2000) FKF1, a clock-controlled gene that regulates the transition to flowering in Arabidopsis. Cell 101:331–340
Olsen KM, Lea US, Slimestad R, Verheul M, Lillo C (2008) Differential expression of four Arabidopsis PAL genes; PAL1 and PAL2 have functional specialization in abiotic environmental-triggered flavonoid synthesis. J Plant Physiol 165:1491–1499. https://doi.org/10.1016/j.jplph.2007.11.005
Petrussa E, Braidot E, Zancani M, Peresson C, Bertolini A, Patui S, Vianello A (2013) Plant flavonoids–biosynthesis, transport and involvement in stress responses. Int J Mol Sci 14:14950–14973. https://doi.org/10.3390/ijms140714950
Pickart CM (2004) Back to the future with ubiquitin. Cell 116:181–190
Prag S, Adams JC (2003) Molecular phylogeny of the kelch-repeat superfamily reveals an expansion of BTB/kelch proteins in animals. BMC Bioinform 4:42. https://doi.org/10.1186/1471-2105-4-42
Rohde A et al (2004) Molecular phenotyping of the pal1 and pal2 mutants of Arabidopsis thaliana reveals far-reaching consequences on phenylpropanoid, amino acid, and carbohydrate metabolism. Plant Cell 16:2749–2771. https://doi.org/10.1105/tpc.104.023705
Santos-Buelga C, Scalbert A (2000) Proanthocyanidins and tannin-like compounds—nature, occurrence, dietary intake and effects on nutrition and health. J Sci Food Agric 80:1094–1117
Sawa M, Nusinow DA, Kay SA, Imaizumi T (2007) FKF1 and GIGANTEA complex formation is required for day-length measurement in Arabidopsis. Science 318:261–265
Sharma D, Tiwari M, Pandey A, Bhatia C, Sharma A, Trivedi PK (2016) MicroRNA858 is a potential regulator of phenylpropanoid pathway and plant development. Plant Physiol 171:944–959. https://doi.org/10.1104/pp.15.01831
Sun Y, Zhou X, Ma H (2007) Genome-wide analysis of Kelch repeatcontaining F-box family. J Integr Plant Biol 49:940–952. https://doi.org/10.1111/j.1672-9072.2007.00498.x
Thompson J, Higgins D, Gibson T (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Vierstra RD (2009) The ubiquitin-26S proteasome system at the nexus of plant biology. Nat Rev Mol Cell Biol 10:385–397. https://doi.org/10.1038/nrm2688
Vogt T (2010) Phenylpropanoid biosynthesis. Mol Plant 3:2–20. https://doi.org/10.1093/mp/ssp106
Wanner LA, Li G, Ware D, Somssich IE, Davis KR (1995) The phenylalanine ammonia-lyase gene family in Arabidopsis thaliana. Plant Mol Biol 27:327–338. https://doi.org/10.1007/bf00020187
Winkel-Shirley B (2001) Flavonoid biosynthesis. a colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 126:485–493
Xu G, Ma H, Nei M, Kong H (2009) Evolution of F-box genes in plants: different modes of sequence divergence and their relationships with functional diversification. Proc Natl Acad Sci USA 106:835–840. https://doi.org/10.1073/pnas.0812043106
Xue F, Cooley L (1993) Kelch encodes a component of intercellular bridges in drosophila. Egg Chambers Cell 72:681–693
Yasuhara M et al (2004) Identification of ASK and clock-associated proteins as molecular partners of LKP2 (LOV kelch protein 2) in Arabidopsis. J Exp Bot 55:2015–2027. https://doi.org/10.1093/jxb/erh226
Zhang X, Gou M, Liu CJ (2013) Arabidopsis Kelch repeat F-box proteins regulate phenylpropanoid biosynthesis via controlling the turnover of phenylalanine ammonia-lyase. Plant Cell 25:4994–5010. https://doi.org/10.1105/tpc.113.119644
Zhang X, Gou M, Guo C, Yang H, Liu CJ (2015) Down-regulation of Kelch domain-containing F-box protein in Arabidopsis enhances the production of (poly)phenols and tolerance to ultraviolet radiation. Plant Physiol 167:337–350. https://doi.org/10.1104/pp.114.249136
Zhang X, Abrahan C, Colquhoun TA, Liu CJ (2017) A proteolytic regulator controlling chalcone synthase stability and flavonoid biosynthesis in Arabidopsis. Plant Cell 29:1157–1174. https://doi.org/10.1105/tpc.16.00855
Zhou J, Lee C, Zhong R, Ye ZH (2009) MYB58 and MYB63 are transcriptional activators of the lignin biosynthetic pathway during secondary cell wall formation in Arabidopsis. Plant Cell 21:248–266. https://doi.org/10.1105/tpc.108.063321
Zhu JY et al (2017) The F-box protein KIB1 mediates brassinosteroid-induced inactivation and degradation of GSK3-like kinases in Arabidopsis. Mol Cell 66:648–657 e644. https://doi.org/10.1016/j.molcel.2017.05.012
Acknowledgements
This research was supported by Basic Science Research Program (NRF-2016R1D1A1B03933195) and Global Research Laboratory Program (2017K1A1A2013146) through the National Research Foundation of Korea (NRF) funded by the Ministry of Education and the Ministry of Science and ICT (MSIT), South Korea.
Author information
Authors and Affiliations
Contributions
BhL and MCS conceived and designed the experiments. SiY and HK conducted experiments. SiY, HK, DJY, MCS and BhL discussed and interpreted the results. SiY and BhL wrote the paper with the help of HK, DJY and MCS.
Corresponding author
Ethics declarations
Conflict interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yu, Si., Kim, H., Yun, DJ. et al. Post-translational and transcriptional regulation of phenylpropanoid biosynthesis pathway by Kelch repeat F-box protein SAGL1. Plant Mol Biol 99, 135–148 (2019). https://doi.org/10.1007/s11103-018-0808-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-018-0808-8