Abstract
Purpose
This paper is concerned with the solid-state characterization of dehydrated calcium salts as well as the effect of dehydration on the physical properties of these salts.
Methods
The salts were analyzed by X-ray powder diffraction (XRPD), single crystal X-ray and polarized light microscopy (PLM).
Results
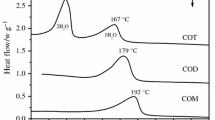
Our research was able to identify three general behaviors of the desolvation of calcium salts of carboxylic acids. Upon desolvation, ethanolate and pentahydrate calcium indomethacin (CaIndo and CaInd2, respectively) stayed crystalline, fenoprofen calcium (CaFEN), calcium ketoprofen (CaKTN), calcium salicylate (CaSAL), calcium mefenamate (CaMEF) and calcium tolfenamate (CaTOLF) became mesophases, while calcium diflunisal became partially crystalline. On the other hand, the solubility studies of CaFEN, CaKTN and CaSAL showed that all dehydrated calcium salts had higher solubility than their crystalline counterparts and amorphous CaKTN had higher solubility than mesomorphous CaKTN.
Conclusions
Several factors influence the desolvation behavior of calcium salts. We believe the flexibility of the benzene rings in CaKTN, CaFEN, CaMEF and CaTOLF was important for these products to become mesomorphous when they loose their crystalline water; meanwhile, CaDIF where the two benzene rings are coplanar remained crystalline when heated. Additionally, the existence of water channels and the hydrogen bonding networks in the crystals is hypothesized to play an important role in the desolvation behavior of these materials.





Similar content being viewed by others
References
A. Terakita and S. R. Byrn. Structure and physical stability of hydrates and thermotropic mesophase of calcium benzoate. J. Pharm. Sci. 95:1162–1172 (2006).
The United States Pharmacopoeia, 28th revision, United States Pharmacopeial Convention, Rockville, U.S.A., 2005, p. 2513.
G. R. Desiraju. In search of clarity. Nature 423:485 (2003).
J. Patterson, A. Bary, and T. Rades. Physical stability and solubility of the thermotropic mesophase of fenoprofen calcium as pure drug and in a tablet formulation. Int. J. Pharm. 247:147–157 (2002).
J. S. G. Cox, G. D. Woodard, and W. McCrone. Solid-state chemistry of cromolyn sodium (disodium cromoglycate). J. Pharm. Sci. 60:1485 (1971).
E. Vadas, P. Toma, and G. Zografi. Solid-state phase transition initiated by water vapor sorption of crystalline L-660,711, a leukotriene D4 receptor antagonist. Pharm. Res. 8:148–155 (1991).
T. Rades, Y. Padmadisastra, and C. Muller-Goymann. Thermal behavior and solubility of fenoprofen calcium. Pharmazie 51:846–851 (1996).
D. Lechuga-Ballesteros, A. Abdul-Fattah, C. Stevenson, and D. B. Bennett. Properties and stability of a liquid crystal form of Cyclosporin—the first reported naturally occurring peptide that exists as a thermotropic liquid crystal. J. Pharm. Sci. 92:1821–1831 (2003).
D. Lechhuga-Ballesteros, A. Abdul-Fattah, Y. Liang, D. P. Miller, C. L. Stevenson, J. Zhang, and D. B. Bennett. Liquid crystallinity improves the physical stability of spray dried powders for inhalation. Respir. Drug Delivery IX 569–571 (2004).
C. J. Strachan, T. Rades, D. A. Newnham, K. C. Gordon, M. Pepper, and P. F. Taday. Using terahertz pulsed spectroscopy to study crystallinity of pharmaceutical materials. Chem. Phys. Lett. 390:20–24 (2004).
C. L. Stevenson, M. M. Tan, and D. Lechuga-Ballesteros. Secondary structure of cyclosporine in spray-dried liquid crystal by FTIR. J. Pharm. Sci. 92:1832–1843 (2003).
H. Bunjes, and T. Rades. Thermotropic liquid crystalline drugs. J. Pharm. Pharmacol. 57:807–816 (2005).
C. L. Stevenson, D. B. Bennett, and D. Lechuga-Ballesteros. Pharmaceutical liquid crystals: the relevance of partially ordered systems. J. Pharm. Sci. 94:1861–1880 (2005).
M. Pavlov, P. E. M. Siegbahn, and M. Sandstrom. Hydration of beryllium, magnesium, calcium, and zinc ions using density functional theory. J. Phys. Chem. 102:219–228 (1998).
R. Debuyst, F. Dejehet, and M. Dekandelaer. Determination des structures crystalline et moleculaire des salicylates de calcium et de strontium dehydrates. Etude par resonance paramagnetique electronique du cuivre (II) incorpore a ces composes de coordination huit. J. Chim. Phys. 76:1117–1124 (1979).
Acknowledgments
The authors acknowledge the financial support from the Purdue–Michigan Solid State Program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Atassi, F., Byrn, S.R. General Trends in the Desolvation Behavior of Calcium Salts. Pharm Res 23, 2405–2412 (2006). https://doi.org/10.1007/s11095-006-9015-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-006-9015-4