Purpose
To design an in vitro apparatus that could simulate the in vivo range of surface shear stresses relevant for the human stomach under fed conditions.
Methods
Computer simulations were combined with in vitro experiments to quantify tablet erosion rate vs. surface shear stress. From two separate computer models, of tablets in the fed stomach and of tablets in vitro, we first estimated the intragastric range of surface stress and Reynolds number (Re), and then designed a dissolution apparatus and parameter space to replicate the in vivo conditions. The in vitro tablet erosion was determined by a new rotating beaker apparatus that provided predictable surface shear on tablets. Tablet mass erosion rates were measured for two different extended-release tablets at a range of in vivo relevant surface shear stresses obtained by varying viscosity of test media and rotation rate of the beaker.
Results
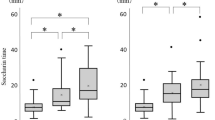
Mass erosion rate and surface shear were found to be highly correlated. Erosion rate increased with surface shear more rapidly at “low” stresses (<35 dyne/cm2) independent of tablet material. At higher surface stress, erosion was strongly material dependent.
Conclusions
Shear force effects on drug release from matrix tablets relevant for fed state are for the first time possible to predict by in vitro dissolution testing.










Similar content being viewed by others
Abbreviations
- ER:
-
extended-release
- GI:
-
gastrointestinal
- MRI:
-
magnetic resonance imaging
- Re :
-
Reynolds number
References
A. Lindahl A. L. Ungell L. Knutson H. Lennernas (1997) ArticleTitleCharacterization of fluids from the stomach and proximal jejunum in men and women Pharm. Res. 14 497–502 Occurrence Handle10.1023/A:1012107801889 Occurrence Handle9144738
J. B. Dressman G. L. Amidon C. Reppas V. P. Shah (1998) ArticleTitleDissolution testing as a prognostic tool for oral drug absorption: immediate release dosage forms Pharm. Res. 15 11–22 Occurrence Handle10.1023/A:1011984216775 Occurrence Handle9487541
B. Abrahamsson K. Roos J. Sjogren (1999) ArticleTitleInvestigation of prandial effects on hydrophilic matrix tablets Drug Dev. Ind. Pharm. 25 765–771 Occurrence Handle10.1081/DDC-100102236 Occurrence Handle10349562
D. A. Alderman (1984) ArticleTitleReview of cellulose ethers in hydrophilic matrices for oral controlled-release dosage forms Int. J. Pharm. Technol. Prod. Manuf. 5 1–9
B. Abrahamsson M. Alpsten B. Bake U. E. Jonsson M. Eriksson-Lepkowska A. Larsson (1998) ArticleTitleDrug absorption from nifedipine hydrophilic matrix extended-release (ER) tablet-comparison with an osmotic pump tablet and effect of food J. Control. Release 52 301–310 Occurrence Handle10.1016/S0168-3659(97)00267-8 Occurrence Handle9743450
M. Kamba Y. Seta A. Kusai M. Ikeda K. Nishimura (2000) ArticleTitleA unique dosage form to evaluate the mechanical destructive force in the gastrointestinal tract Int. J. Pharm. 208 61–70 Occurrence Handle10.1016/S0378-5173(00)00552-4 Occurrence Handle11064212
L. E. Dahlinder C. Graffner J. Sjogren (1973) ArticleTitleStrength of the insoluble residues of plastic matrix slow release tablets (Duretter) in vitro and in vivo Acta Pharm. Suec. 10 323–332 Occurrence Handle4759357
M. Shameem N. Katori N. Aoyagi S. Kojima (1995) ArticleTitleOral solid controlled release dosage forms: role of GI-mechanical destructive forces and colonic release in drug absorption under fasted and fed conditions in humans Pharm. Res. 12 1049–1054 Occurrence Handle10.1023/A:1016270701021 Occurrence Handle7494801
B. Abrahamsson M. Alpsten B. Bake A. Larsson J. Sjogren (1998) ArticleTitleIn vitro and in vivo erosion of two different hydrophilic gel matrix tablets Eur. J. Pharm. Biopharm. 46 69–75 Occurrence Handle10.1016/S0939-6411(98)00002-2 Occurrence Handle9700024
A. Pal, K. Indireshkumar, W. Schwizer, B. Abrahamsson, M. Fried and J. G. Brasseur, Gastric flow and mixing studied using computer simulation. Proc. R. Soc. Lond. B Biol. Sci. 271 (2004).
A. Pal B. Abrahamsson W. Schwizer G. S. Hebbard J. G. Brasseur (2003) ArticleTitle Application of a virtual stomach to evaluate gastric mixing and breakdown of solid food Gastroenterology 124 A673–A674
R. L. Panton (1996) Incompressible Flow John Wiley & Sons New York
K. Indireshkumar J. G. Brasseur H. Faas G. S. Hebbard P. Kunz J. Dent C. Feinle M. J. Li P. Boesiger M. Fried W. Schwizer (2000) ArticleTitleRelative contributions of “pressure pump” and “peristaltic pump” to gastric emptying Am. J. Physiol. Gasterointest. Liver Physiol. 278 G604–G616
N. Pallotta M. Cicala C. Frandina E. Corazziari (1998) ArticleTitleAntro-pyloric contractile patterns and transpyloric flow after meal ingestion in humans Am. J. Gastroenterol. 93 2513–2522 Occurrence Handle10.1111/j.1572-0241.1998.00598.x Occurrence Handle9860417
J. L. Baxter J. Kukura F. J. Muzzio (2005) ArticleTitleHydrodynamics-induced variability in the USP apparatus II dissolution test Int. J. Pharm. 292 17–28 Occurrence Handle10.1016/j.ijpharm.2004.08.003 Occurrence Handle15725550
J. Kukura J. L. Baxter F. J. Muzzio (2004) ArticleTitleShear distribution and variability in the USP Apparatus 2 under turbulent conditions Int. J. Pharm. 279 9–17 Occurrence Handle10.1016/j.ijpharm.2004.03.033 Occurrence Handle15234789
K. Wingstrand B. Abrahamsson B. Edgar (1990) ArticleTitleBioavailability from felodipine extended-release tablets with different dissolution properties Int. J. Pharm. 60 151–156 Occurrence Handle10.1016/0378-5173(90)90301-J
B. Abrahamsson M. Alpsten M. Hugosson U. E. Jonsson M. Sundgren A. Svenheden J. Tolli (1993) ArticleTitleAbsorption, gastrointestinal transit, and tablet erosion of felodipine extended-release (ER) tablets Pharm. Res. 10 709–714 Occurrence Handle10.1023/A:1018959732744 Occurrence Handle8321836
L. Karlson S. Nilsson K. Thuresson (1999) ArticleTitleRheology of an aqueous solution of an end-capped poly(ethylene glycol) polymer at high concentration Colloid Polym. Sci. 277 798–804 Occurrence Handle10.1007/s003960050454
J. D. Sartor C. E. Abbott (1975) ArticleTitlePrediction and measurement of the accelerated motion of water drops in the air J. Appl. Meteorol. 14 232–239 Occurrence Handle10.1175/1520-0450(1975)014<0232:PAMOTA>2.0.CO;2
F. M. White (2003) Fluid Mechanics McGraw-Hill Boston
Acknowledgments
This work was supported by AstraZeneca. We are very grateful to Ms. Lena Wejkum for her contributions to the experiments on tablet Y.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Development of the “Tablet Erosion Model”
To develop the tablet erosion model we applied a systematic procedure that exists in the engineering literature for developing empirical mathematical relationships between an “independent variable,” here the mass erosion rate \(\ifmmode\expandafter\dot\else\expandafter\.\fi{m}\)and the “dependent variables” that affect mass erosion rate (shear stress, relative velocity, fluid viscosity, etc.). This procedure is a combination of “similarity theory” and “dimensional analysis.” We can only briefly summarize the method here. The reader should refer to standard textbooks in fluid dynamics (21) for further details.
Two different flows around two different tablets are “similar” if the flow patterns around the tablets are the same. This requires, among other things, that the geometry and orientation of the two tablets in the flow be the same. The tablet size, which we define with D, need not be the same. Neither does the velocity U of the flow relative to the tablet, the fluid viscosity μ, nor the fluid density ρ have to be the same to produce the same flow pattern (“similarity”). Using a process called “dimensional analysis” (21), however, it can be shown that the flow patterns will be the same only if a specific combination of μ, ρ, U, and D, given by the “Reynolds number” Re = (ρUD/μ), is the same between the two flows.
We applied “similarity theory” with “dimensional analysis” to generalize the development of empirical mathematical relationships from data. We first argued that, for fixed orientation and tablet shape, average surface shear stress τ avg depends only on relative flow velocity U, tablet size D, and fluid properties viscosity μ and density ρ. Dimensional analysis leads to the result that an appropriately nondimensionalized τ avg depends only on Reynolds number. In other words, given sufficient data, we develop a mathematical equation to fit the symbolic form,
Dimensional analysis leads to μU/D being the appropriate nondimensionalization for τ avg , and the symbol “f” implies a mathematical relationship between τ avg /(μU/D) and Re = ρUD/μ that is to be determined from experimental data. Inthis work we generated this data from computer simulations that replicate the in vitro flow. The important point from Eq. (A1) is that to determine an empirical mathematical relationship between τ avg , and the variables U, μ, D, and ρ, we need not vary each of the four dependent variables (U, μ, D, ρ) separately. In this study we vary only μ and U (for fixed D and ρ) to collect data from which we empirically estimate the mathematical relationship (f) between the two nondimensional variables τ avg /(μU/D) and Re = ρUD/μ. Once done, multiplication of right-hand side of the empirical mathematical expression for f by μU/D produces a mathematical relationship between dimensional τ avg and U, μ, D, and ρ.
We applied the same procedure to develop the form of the mathematical relationship between an appropriately nondimensionalized mass erosion rate by arguing that, for fixed tablet shape and orientation, \(\ifmmode\expandafter\dot\else\expandafter\.\fi{m}\)depends only on average surface shear stress τ avg , tablet size D, relative tablet velocity U, a material surface parameter τ u (see Materials and Methods), and fluid properties μ and ρ. The dimensional analysis procedure leads to the following symbolic relationship:
Here dimensional analysis lead to the result that mass erosion rate, nondimensionalized by τ avg D2/U, can be written as a mathematical expression “h” that contains \(\frac{{\tau _{{avg}} }} {{{\mu U} \mathord{\left/ {\vphantom {{\mu U} D}} \right. \kern-\nulldelimiterspace} D}}\) Re and τ u /τ avg . In this work we determined this mathematical relationship by combining experimental and computer simulation data.
However, we can reduce Eq. (A2) to a simpler form. Assuming that (A1) is uniquely invertible, we replace Re in (A2) with a function that depends only on \(\frac{{\tau _{{avg}} }} {{{\mu U} \mathord{\left/ {\vphantom {{\mu U} D}} \right. \kern-\nulldelimiterspace} D}}\), producing the following reduced form:
Here “g” implies a mathematical relationship between \({\ifmmode\expandafter\dot\else\expandafter\.\fi{m}} \mathord{\left/ {\vphantom {{\ifmmode\expandafter\dot\else\expandafter\.\fi{m}} {{\left( {{\tau _{{avg}} D^{2} } \mathord{\left/ {\vphantom {{\tau _{{avg}} D^{2} } U}} \right. \kern-\nulldelimiterspace} U} \right)}}}} \right. \kern-\nulldelimiterspace} {{\left( {{\tau _{{avg}} D^{2} } \mathord{\left/ {\vphantom {{\tau _{{avg}} D^{2} } U}} \right. \kern-\nulldelimiterspace} U} \right)}}\), τ avg /(μU/D), and τ u /τ avg that we determine through data obtained by combining in vitro experimental measurements of \(\ifmmode\expandafter\dot\else\expandafter\.\fi{m}\)with computer simulation “measurement” of τ avg , for tablets in flows with different μ and U, but with fixed orientation, size D and material property τ u . From the mathematical form of g, the tablet erosion model is obtained by multiplying g by τ avg D2/U.
Rights and permissions
About this article
Cite this article
Abrahamsson, B., Pal, A., Sjöberg, M. et al. A Novel in Vitro and Numerical Analysis of Shear-Induced Drug Release from Extended-Release Tablets in the Fed Stomach. Pharm Res 22, 1215–1226 (2005). https://doi.org/10.1007/s11095-005-5272-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-005-5272-x