Abstract
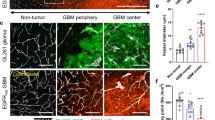
Glioblastoma multiforme (GBM) is among the most highly vascularized of solid tumors, contributing to the infiltrative nature of the disease, and conferring poor outcome. Due to the critical dependency of GBM on growth of new endothelial vasculature, we evaluated the preclinical activity of a novel adenoviral gene therapy that targets the endothelium within newly formed blood vessels for apoptosis. VB-111, currently in phase II clinical trials, consists of a non-replicating Adenovirus 5 (El deleted) carrying a proapoptotic human Fas-chimera (transgene) under the control of a modified murine promoter (PPE-1-3×) which specifically targets endothelial cells within the tumor vasculature. Here we report that a single intravenous dose of 2.5 × 1011 or 1 × 1011 VPs was sufficient to extend survival in nude rats bearing U87MG-luc2 or nude mice bearing U251-luc, respectively. Bioluminescence imaging of nude rats showed that VB-111 effectively inhibited tumor growth within four weeks of treatment. This was confirmed in a select group of animals by MRI. In our mouse model we observed that 3 of 10 nude mice treated with VB-111 completely lost U251 luciferase signal and were considered long term survivors. To assess the antiangiogenic effects of VB-111, we evaluated the tumor-associated microvaculature by CD31, a common marker of neovascularization, and found a significant decrease in the microvessel density by IHC. We further assessed the neovasculature by confocal microscopy and found that VB-111 inhibits vascular density in two separate mouse models bearing U251-RFP xenografts. Collectively, this study supports the clinical development of VB-111 as a treatment for GBM.





Similar content being viewed by others
References
Wen PY, Kesari S (2008) Malignant gliomas in adults. N Engl J Med 359:492–507. doi:10.1056/NEJMra0708126
Jain RK, di Tomaso E, Duda DG, Loeffler JS, Sorensen AG, Batchelor TT (2007) Angiogenesis in brain tumours. Nat Rev Neurosci 8:610–622. doi:10.1038/nrn2175
Carbone C, Campisi A, Musumeci T, Raciti G, Bonfanti R, Puglisi G (2014) FA-loaded lipid drug delivery systems: preparation, characterization and biological studies. Eur J Pharm Sci 52:12–20. doi:10.1016/j.ejps.2013.10.003
Salameh TS, Banks WA (2014) Delivery of therapeutic peptides and proteins to the CNS. Adv Pharmacol 71:277–299. doi:10.1016/bs.apha.2014.06.004
Garrido W, Rocha JD, Jaramillo C, Fernandez K, Oyarzun C, San Martin R, Quezada C (2014) Chemoresistance in high-grade gliomas: relevance of adenosine signalling in stem-like cells of glioblastoma multiforme. Curr Drug Targets 15:931–942
Tentori L, Ricci-Vitiani L, Muzi A, Ciccarone F, Pelacchi F, Calabrese R, Runci D, Pallini R, Caiafa P, Graziani G (2014) Pharmacological inhibition of poly(ADP-ribose) polymerase-1 modulates resistance of human glioblastoma stem cells to temozolomide. BMC Cancer 14:151. doi:10.1186/1471-2407-14-151
Frampton JE, Keating GM (2008) Bevacizumab: in first-line treatment of advanced and/or metastatic renal cell carcinoma. BioDrugs 22:113–120
Gubbi A, Kendrick JE, Finkler NJ (2014) The role of bevacizumab in recurrent, platinum-sensitive ovarian cancer. Expert Rev Anticancer Ther 14:1105–1113. doi:10.1586/14737140.2014.956095
Keating GM (2014) Bevacizumab: a review of its use in advanced cancer. Drugs 74:1891–1925. doi:10.1007/s40265-014-0302-9
Maillet M, Dreanic J, Dhooge M, Mir O, Brezault C, Goldwasser F, Chaussade S, Coriat R (2014) The predictive and prognostic value of the Glasgow Prognostic Score in metastatic colorectal carcinoma patients receiving bevacizumab. Anti-cancer Drugs 25:1215–1219. doi:10.1097/CAD.0000000000000129
Odia Y, Shih JH, Kreisl TN, Fine HA (2014) Bevacizumab-related toxicities in the National Cancer Institute malignant glioma trial cohort. J Neuro Oncol 120:431–440. doi:10.1007/s11060-014-1571-6
Zinner RG, Obasaju CK, Spigel DR, Weaver RW, Beck JT, Waterhouse DM, Modiano MR, Hrinczenko B, Nikolinakos PG, Liu J, Koustenis AG, Winfree KB, Melemed SA, Guba SC, Ortuzar WI, Desaiah D, Treat JA, Govindan R, Ross HJ (2014) PRONOUNCE: randomized, open-Label, phase III study of first-line pemetrexed + carboplatin followed by maintenance pemetrexed versus paclitaxel + carboplatin + bevacizumab followed by maintenance bevacizumab in patients ith advanced nonsquamous non-small-cell lung cancer. J Thorac Oncol. doi:10.1097/JTO.0000000000000366
Cea V, Sala C, Verpelli C (2012) Antiangiogenic therapy for glioma. J Signal Transduct 2012:483040. doi:10.1155/2012/483040
Hou LC, Veeravagu A, Hsu AR, Tse VC (2006) Recurrent glioblastoma multiforme: a review of natural history and management options. Neurosurg Focus 20:E5
Greenberger S, Shaish A, Varda-Bloom N, Levanon K, Breitbart E, Goldberg I, Barshack I, Hodish I, Yaacov N, Bangio L, Goncharov T, Wallach D, Harats D (2004) Transcription-controlled gene therapy against tumor angiogenesis. J Clin Investig 113:1017–1024. doi:10.1172/JCI20007
Harats D, Kurihara H, Belloni P, Oakley H, Ziober A, Ackley D, Cain G, Kurihara Y, Lawn R, Sigal E (1995) Targeting gene expression to the vascular wall in transgenic mice using the murine preproendothelin-1 promoter. J Clin Investig 95:1335–1344. doi:10.1172/JCI117784
Varda-Bloom N, Shaish A, Gonen A, Levanon K, Greenbereger S, Ferber S, Levkovitz H, Castel D, Goldberg I, Afek A, Kopolovitc Y, Harats D (2001) Tissue-specific gene therapy directed to tumor angiogenesis. Gene Ther 8:819–827. doi:10.1038/sj.gt.3301472
Brenner AJ, Cohen YC, Breitbart E, Bangio L, Sarantopoulos J, Giles FJ, Borden EC, Harats D, Triozzi PL (2013) Phase I dose-escalation study of VB-111, an antiangiogenic virotherapy, in patients with advanced solid tumors. Clin Cancer Res 19:3996–4007. doi:10.1158/1078-0432.CCR-12-2079
Phillips WT, Goins B, Bao A, Vargas D, Guttierez JE, Trevino A, Miller JR, Henry J, Zuniga R, Vecil G, Brenner AJ (2012) Rhenium-186 liposomes as convection-enhanced nanoparticle brachytherapy for treatment of glioblastoma. Neuro Oncol 14:416–425. doi:10.1093/neuonc/nos060
de Bouard S, Herlin P, Christensen JG, Lemoisson E, Gauduchon P, Raymond E, Guillamo JS (2007) Antiangiogenic and anti-invasive effects of sunitinib on experimental human glioblastoma. Neuro Oncol 9:412–423. doi:10.1215/15228517-2007-024
Furnari FB, Fenton T, Bachoo RM, Mukasa A, Stommel JM, Stegh A, Hahn WC, Ligon KL, Louis DN, Brennan C, Chin L, DePinho RA, Cavenee WK (2007) Malignant astrocytic glioma: genetics, biology, and paths to treatment. Genes Dev 21:2683–2710. doi:10.1101/gad.1596707
Soda Y, Myskiw C, Rommel A, Verma IM (2013) Mechanisms of neovascularization and resistance to anti-angiogenic therapies in glioblastoma multiforme. J Mol Med 91:439–448. doi:10.1007/s00109-013-1019-z
Baker GJ, Yadav VN, Motsch S, Koschmann C, Calinescu AA, Mineharu Y, Camelo-Piragua SI, Orringer D, Bannykh S, Nichols WS, deCarvalho AC, Mikkelsen T, Castro MG, Lowenstein PR (2014) Mechanisms of glioma formation: iterative perivascular glioma growth and invasion leads to tumor progression, VEGF-independent vascularization, and resistance to antiangiogenic therapy. Neoplasia 16:543–561. doi:10.1016/j.neo.2014.06.003
Nandhu MS, Hu B, Cole SE, Erdreich-Epstein A, Rodriguez-Gil DJ, Viapiano MS (2014) Novel paracrine modulation of Notch-DLL4 signaling by fibulin-3 promotes angiogenesis in high-grade gliomas. Cancer Res 74:5435–5448. doi:10.1158/0008-5472.CAN-14-0685
Conrad C, Miller CR, Ji Y, Gomez-Manzano C, Bharara S, McMurray JS, Lang FF, Wong F, Sawaya R, Yung WK, Fueyo J (2005) Delta24-hyCD adenovirus suppresses glioma growth in vivo by combining oncolysis and chemosensitization. Cancer Gene Ther 12:284–294. doi:10.1038/sj.cgt.7700750
Jiang H, Gomez-Manzano C, Alemany R, Medrano D, Alonso M, Bekele BN, Lin E, Conrad CC, Yung WK, Fueyo J (2005) Comparative effect of oncolytic adenoviruses with E1A–55 kDa or E1B–55 kDa deletions in malignant gliomas. Neoplasia 7:48–56. doi:10.1593/neo.04391
Samoto K, Ehtesham M, Perng GC, Hashizume K, Wechsler SL, Nesburn AB, Black KL, Yu JS (2002) A herpes simplex virus type 1 mutant with gamma 34.5 and LAT deletions effectively oncolyses human U87 glioblastomas in nude mice. Neurosurgery 50:599–605; discussion 605–596
Gomez-Manzano C, Holash J, Fueyo J, Xu J, Conrad CA, Aldape KD, de Groot JF, Bekele BN, Yung WK (2008) VEGF Trap induces antiglioma effect at different stages of disease. Neuro Oncol 10:940–945. doi:10.1215/15228517-2008-061
Muldoon LL, Gahramanov S, Li X, Marshall DJ, Kraemer DF, Neuwelt EA (2011) Dynamic magnetic resonance imaging assessment of vascular targeting agent effects in rat intracerebral tumor models. Neuro Oncol 13:51–60. doi:10.1093/neuonc/noq150
Provenzale JM (2007) Imaging of angiogenesis: clinical techniques and novel imaging methods. AJR 188:11–23. doi:10.2214/AJR.06.0280
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. B. received a commercial research Grant from and is a consultant/advisory board member of Vascular Biogenics. Y.C.C., L.B., N. Y. and E. B. are employed by and have ownership interest in VBL Therapeutics.
Funding
The study was supported by in part by the UTHSCSA Cancer Therapy and Research Center through the National Institutes of Health National Cancer Institute P30 award CA054174, as well as by a service agreement with VBL Therapeutics.
Rights and permissions
About this article
Cite this article
Gruslova, A., Cavazos, D.A., Miller, J.R. et al. VB-111: a novel anti-vascular therapeutic for glioblastoma multiforme. J Neurooncol 124, 365–372 (2015). https://doi.org/10.1007/s11060-015-1853-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-1853-7