Abstract
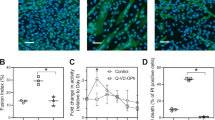
Phosphorylase b kinase (PhK) is a key enzyme involved in the conversion of glycogen to glucose in skeletal muscle and ultimately an increase in intracellular ATP. Since apoptosis is an ATP-dependent event, we investigated the regulation of skeletal muscle PhK during apoptosis. Incubation of PhK with purified caspase-3 in vitro resulted in the highly selective cleavage of the regulatory α subunit and resulted in a 2-fold increase in PhK activity. Edman protein sequencing of a stable 72 kD amino-terminal fragment and a 66 kD carboxy-terminal fragment revealed a specific caspase-3 cleavage site within the α subunit at residue 646 (DWMD↓G). Treatment of differentiated C2C12 mouse muscle myoblasts with the inducers of apoptosis staurosporine, TPEN, doxorubicin, or UV irradiation resulted in the disappearance of the α subunit of PhK as determined by immunoblotting, as well as a concurrent increase in caspase-3 activity. Moreover, induction of apoptosis by TPEN resulted in increased phosphorylase activity and sustained ATP levels throughout a 7 h time course. However, induction of apoptosis with staurosporine, also a potent PhK inhibitor, led to a rapid loss in phosphorylase activity and intracellular ATP, suggesting that PhK inhibition by staurosporine impairs the ability of apoptotic muscle cells to generate ATP. Thus, these studies indicate that PhK may be a substrate for caspase regulation during apoptosis and suggest that activation of this enzyme may be important for the generation of ATP during programmed cell death.
Similar content being viewed by others
Abbreviations
- ddFSK:
-
1,9-dideoxyforskolin
- PhK:
-
Phosphorylase b kinase
- STS:
-
Staurosporine
- TPEN:
-
N, N, N′, N′,-tetrakis(2-pyridylmethyl)ethylenediamine
- TUNEL:
-
Terminal deoxynucleotidyl transferase mediated dUTP nick end labeling
References
Saraste A, Pulkki K: Morphologic and biochemical hallmarks of apoptosis. Cardiovasc Res 45: 528–537, 2000
Sandri M, Carraro U: Apoptosis of skeletal muscles during development and disease. Int J Biochem Cell Biol 31: 1373–1390, 1999
Casciola-Rosen L, Nicholson DW, Chong T, Rowan KR, Thornberry NA, Miller DK, Rosen A: Apopain/CPP32 cleaves proteins that are essential for cellular repair: A fundamental principle of apoptotic death. J Exp Med 183: 1957–1964, 1996
Cohen GM: Caspases: The executioners of apoptosis. Biochem J 326: 1–16, 1997
Krebs EG, Graves JD: Interactions between protein kinases and proteases in cellular signaling and regulation. Adv Enzyme Regul 40: 441–470, 2000
Allen DL, Linderman JK, Roy RR, Bigbee AJ, Grindeland RE, Mukku V, Edgerton VR: Apoptosis: A mechanism contributing to remodeling of skeletal muscle in response to hindlimb unweighting. Am J Physiol 273: C579–C587, 1997
Hasselgren PO, Fischer JE: Muscle cachexia: Current concepts of intracellular mechanisms and molecular regulation. Ann Surg 233: 9–17, 2001
Sandri M, Carraro U, Podhorska-Okolov M, Rizzi C, Arslan P, Monti D, Franceschi C: Apoptosis, DNA damage and ubiquitin expression in normal and mdx muscle fibers after exercise. FEBS Lett 373: 291–295, 1995
Sandri M, Minetti C, Pedemonte M, Carraro U: Apoptotic myonuclei in human Duchenne muscular dystrophy. Lab Invest 78: 1005–1016, 1998
Tews DS, Goebel HH: DNA-fragmentation and expression of apoptosis-related proteins in muscular dystrophies. Neuropathol Appl Neurobiol 23: 331–338, 1997
Yasuhara S, Perez ME, Kanakubo E, Yasuhara Y, Shin YS, Kaneki M, Fujita T, Martyn JA: Skeletal muscle apoptosis after burns is associated with activation of proapoptotic signals. Am J Physiol Endocrinol Metab 279: E1114–E1121, 2000
McArdle A, Maglara A, Appleton P, Watson AJ, Grierson I, Jackson MJ: Apoptosis in multinucleated skeletal muscle myotubes. Lab Invest 79: 1069–1076, 1999
Wang J, Walsh K: Resistance to apoptosis conferred by Cdk inhibitors during myocyte differentiation. Science 273: 359–361, 1996
Wang J, Guo K, Wills KN, Walsh K: Rb functions to inhibit apoptosis during myocyte differentiation. Cancer Res 57: 351–354, 1997
Eguchi Y, Shimizu S, Tsujimoto Y: Intracellular ATP levels determine cell death fate by apoptosis or necrosis. Cancer Res 57: 1835–1840, 1997
Lieberthal W, Menza SA, Levine JS: Graded ATP depletion can cause necrosis or apoptosis of cultured mouse proximal tubular cells. Am J Physiol 274: F315–F327, 1998
Lelli JL, Jr., Becks LL, Dabrowska MI, Hinshaw DB: ATP converts necrosis to apoptosis in oxidant-injured endothelial cells. Free Radic Biol Med 25: 694–702, 1998
Leist M, Single B, Naumann H, Fava E, Simon B, Kuhnle S, Nicotera P: Inhibition of mitochondrial ATP generation by nitric oxide switches apoptosis to necrosis. Exp Cell Res 249: 396–403, 1999
Latta M, Kunstle G, Leist M, Wendel A: Metabolic depletion of ATP by fructose inversely controls CD95- and tumor necrosis factor receptor 1-mediated hepatic apoptosis. J Exp Med 191: 1975–1985, 2000
Grusch M, Polgar D, Gfatter S, Leuhuber K, Huettenbrenner S, Leisser C, Fuhrmann G, Kassie F, Steinkellner H, Smid K, Peters GJ, Jayaram HN, Klepal W, Szekeres T, Knasmuller S, Krupitza G: Maintenance of ATP favours apoptosis over necrosis triggered by benzamide riboside. Cell Death Differ 9: 169–178, 2002
Gramaglia D, Gentile A, Battaglia M, Ranzato L, Petronilli V, Fassetta M, Bernardi P, Rasola A: Apoptosis to necrosis switching downstream of apoptosome formation requires inhibition of both glycolysis and oxidative phosphorylation in a BCL-X(L)- and PKB/AKT-independent fashion. Cell Death Differ 11: 342–353, 2004
Leist M, Single B, Castoldi AF, Kuhnle S, Nicotera P: Intracellular adenosine triphosphate (ATP) concentration: A switch in the decision between apoptosis and necrosis. J Exp Med 185: 1481–1486, 1997
Brushia RJ, Walsh DA: Phosphorylase kinase: The complexity of its regulation is reflected in the complexity of its structure. Front Biosci 4: D618–D641, 1999
Cohen P, Burchell A, Foulkes JG, Cohen PT: Identification of the Ca2+-dependent modulator protein as the fourth subunit of rabbit skeletal muscle phosphorylase kinase. FEBS Lett 92: 287–293, 1978
Huston RB, Krebs EG: Activation of skeletal muscle phosphorylase kinase by Ca2+. II. Identification of the kinase activating factor as a proteolytic enzyme. Biochemistry 7: 2116–2122, 1968
Walsh DA, Perkins JP, Krebs EG: An adenosine 3′,5′-monophosphate-dependant protein kinase from rabbit skeletal muscle. J Biol Chem 243: 3763–3765, 1968
Cohen P: The subunit structure of rabbit-skeletal-muscle phosphorylase kinase, and the molecular basis of its activation reactions. Eur J Biochem 34: 1–14, 1973
Cohen P, Watson DC, Dixon GH: The hormonal control of activity of skeletal muscle phosphorylase kinase. Amino-acid sequences at the two sites of action of adenosine-3′:5′-monophosphate-dependent protein kinase. Eur J Biochem 51: 79–92, 1975
Wang JH, Stull JT, Huang TS, Krebs EG: A study on the autoactivation of rabbit muscle phosphorylase kinase. J Biol Chem 251: 4521–4527, 1976
Cheng A, Fitzgerald TJ, Carlson GM: Adenosine 5′-diphosphate as an allosteric effector of phosphorylase kinase from rabbit skeletal muscle. J Biol Chem 260: 2535–2542, 1985
Traxler KW, Norcum MT, Hainfeld JF, Carlson GM: Direct visualization of the calmodulin subunit of phosphorylase kinase via electron microscopy following subunit exchange. J Struct Biol 135: 231–238, 2001
King MM, Carlson GM: Synergistic activation by Ca2+ and Mg2+ as the primary cause for hysteresis in the phosphorylase kinase reactions. J Biol Chem 256: 11058–11064, 1981
Damer CK, Partridge J, Pearson WR, Haystead TA: Rapid identification of protein phosphatase 1-binding proteins by mixed peptide sequencing and data base searching. Characterization of a novel holoenzymic form of protein phosphatase 1. J Biol Chem 273: 24396–24405, 1998
Wilkinson DA, Marion TN, Tillman DM, Norcum MT, Hainfeld JF, Seyer JM, Carlson GM: An epitope proximal to the carboxyl terminus of the alpha-subunit is located near the lobe tips of the phosphorylase kinase hexadecamer. J Mol Biol 235: 974–982, 1994
Gilboe DP, Larson KL, Nuttall FQ: Radioactive method for the assay of glycogen phosphorylases. Anal Biochem 47: 20–27, 1972
Pogolotti AL, Jr., Santi DV: High-pressure liquid chromatography–ultraviolet analysis of intracellular nucleotides. Anal Biochem 126: 335–345, 1982
Graves DJ, Hayakawa T, Horvitz RA, Beckman E, Krebs EG: Studies on the subunit structure of trypsin-activated phosphorylase kinase. Biochemistry 12: 580–585, 1973
Nakatani T, Tawaramoto M, Opare Kennedy D, Kojima A, Matsui-Yuasa I: Apoptosis induced by chelation of intracellular zinc is associated with depletion of cellular reduced glutathione level in rat hepatocytes. Chem Biol Interact 125: 151–163, 2000
Perry DK, Smyth MJ, Stennicke HR, Salvesen GS, Duriez P, Poirier GG, Hannun YA: Zinc is a potent inhibitor of the apoptotic protease, caspase-3. A novel target for zinc in the inhibition of apoptosis. J Biol Chem 272: 18530–18533, 1997
Elliott LH, Wilkinson SE, Sedgwick AD, Hill CH, Lawton G, Davis PD, Nixon JS: K252a is a potent and selective inhibitor of phosphorylase kinase. Biochem Biophys Res Commun 171: 148–154, 1990
Rudel T, Bokoch GM: Membrane and morphological changes in apoptotic cells regulated by caspase-mediated activation of PAK2. Science 276: 1571–1574, 1997
Walter BN, Huang Z, Jakobi R, Tuazon PT, Alnemri ES, Litwack G, Traugh JA: Cleavage and activation of p21-activated protein kinase gamma-PAK by CPP32 (caspase 3). Effects of autophosphorylation on activity. J Biol Chem 273: 28733–28739, 1998
Cardone MH, Salvesen GS, Widmann C, Johnson G, Frisch SM: The regulation of anoikis: MEKK-1 activation requires cleavage by caspases. Cell 90: 315–323, 1997
Krebs EG, Love DS, Bratvold GE, Trayser KA, Meyer WL, Fischer EH: Purification and properties of rabbit skeletal muscle phosphorylase B kinase. Biochemistry 28: 1022–1033, 1964
Trempe MR, Carlson GM: Phosphorylase kinase conformers. Detection by proteases. J Biol Chem 262: 4333–4340, 1987
Zander NF, Meyer HE, Hoffmann-Posorske E, Crabb JW, Heilmeyer LM, Jr., Kilimann MW: cDNA cloning and complete primary structure of skeletal muscle phosphorylase kinase (alpha subunit). Proc Natl Acad Sci USA 85: 2929–2933, 1988
Andreeva IE, Rice NA, Carlson GM: The regulatory alpha subunit of phosphorylase kinase may directly participate in the binding of glycogen phosphorylase. Biochemistry (Mosc) 67: 1197–1202, 2002
Rice NA, Nadeau OW, Yang Q, Carlson GM: The calmodulin-binding domain of the catalytic gamma subunit of phosphorylase kinase interacts with its inhibitory alpha subunit: Evidence for a Ca2+ sensitive network of quaternary interactions. J Biol Chem 277: 14681–14687, 2002
Ayers NA, Wilkinson DA, Fitzgerald TJ, Carlson GM: Self-association of the alpha subunit of phosphorylase kinase as determined by two-hybrid screening. J Biol Chem 274: 35583–35590, 1999
Ferrari D, Stepczynska A, Los M, Wesselborg S, Schulze-Osthoff K: Differential regulation and ATP requirement for caspase-8 and caspase-3 activation during CD95- and anticancer drug-induced apoptosis. J Exp Med 188: 979–984, 1998
Feldenberg LR, Thevananther S, del Rio M, de Leon M, Devarajan P: Partial ATP depletion induces Fas- and caspase-mediated apoptosis in MDCK cells. Am J Physiol 276: F837–F846, 1999
Yaoita Y, Nakajima K: Induction of apoptosis and CPP32 expression by thyroid hormone in a myoblastic cell line derived from tadpole tail. J Biol Chem 272: 5122–5127, 1997
Fernando P, Kelly JF, Balazsi K, Slack RS, Megeney LA: Caspase 3 activity is required for skeletal muscle differentiation. Proc Natl Acad Sci USA 99: 11025–11030, 2002
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hilder, T.L., Carlson, G.M., Haystead, T.A.J. et al. Caspase-3 dependent cleavage and activation of skeletal muscle phosphorylase b kinase. Mol Cell Biochem 275, 233–242 (2005). https://doi.org/10.1007/s11010-005-2411-y
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-2411-y