Abstract
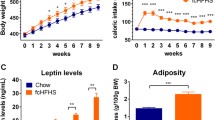
Leptin and cholecystokinin (CCK) are two hormones involved in body weight regulation and feeding behavior. Leptin, an adiposity-derived signal, and CCK, a gut-satiety signal, interact in a synergistic manner and their concomitant treatment causes more weight loss than any hormone administered alone. The synergistic interaction between CCK and leptin has been widely characterized in lean rats, but there are a few studies performed in obese rats. Herein, in the same individual, we compared the sensitivity to CCK, leptin and the combined treatment, leptin + CCK, on body weight and food intake in two experiments one performed in juvenile-lean rats, and in a second experiment comparing adult Chow-fed versus adult-high fat diet (HFD) rats. We found that only the combined treatment had a long-term effect on food intake and caused a substantial and synergistic body weight loss, in juvenile rats. However, the same combined treatment failed to induce weight loss in both Chow-fed and HFD rats. We thus conclude that the sensitivity to leptin + CCK was age dependent, and at the doses used ineffective in adult and obese rats.



Similar content being viewed by others
References
Allman M, Wallace M, Gaskin L, Rivera CA (2009) Leptin induces an inflammatory phenotype in lean Wistar rats. Mediators Inflamm 2009(ID 738620):1–8. doi:10.1155/2009/738620
Aponte Y, Atasoy D, Sternson SM (2011) AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nat Neurosci 14(3):351–355
Barrachina MD, Martinez V, Wang L, Wei JY, Tache Y (1997) Synergistic interaction between leptin and cholecystokinin to reduce short-term food intake in lean mice. Proc Natl Acad Sci USA 94(19):10455–10460
Cakir B, Kasimay O, Devseren E, Yegen BC (2007) Leptin inhibits gastric emptying in rats: role of CCK receptors and vagal afferent fibers. Physiol Res 56(3):315–322
Campfield LA, Smith FJ, Guisez Y, Devos R, Burn P (1995) Recombinant mouse OB protein: evidence for a peripheral signal linking adiposity and central neural networks. Science 269(5223):546–549
Cannon CP, Kumar A (2009) Treatment of overweight and obesity: lifestyle, pharmacologic, and surgical options. Clin Cornerstone 9(4):55–68; discussion 69–71
Cano V, Ezquerra L, Ramos MP, Ruiz-Gayo M (2003) Regulation of leptin distribution between plasma and cerebrospinal fluid by cholecystokinin receptors. Br J Pharmacol 140(4):647–652
Chandler PC, Wauford PK, Oswald KD, Maldonado CR, Hagan MM (2004) Change in CCK-8 response after diet-induced obesity and MC3/4-receptor blockade. Peptides 25(2):299–306
Covasa M (2010) Deficits in gastrointestinal responses controlling food intake and body weight. Am J Physiol Regul Integr Comp Physiol 299(6):R1423–R1439
Covasa M, Ritter RC (1998) Rats maintained on high-fat diets exhibit reduced satiety in response to CCK and bombesin. Peptides 19(8):1407–1415
de Lartigue GLG, Dimaline R, Varro A, Raybould H, Dockray GJ (2010) EGR1 is a target for cooperative interactions between cholecystokinin and leptin, and inhibition by ghrelin, in vagal afferent neurons. Endocrinology 151(8):3589–3599
de Saint Hilaire-Kafi Z, Depoortere H, Nicolaïdis S (1989) Does cholecystokinin induce physiological satiety and sleep? Brain Res 488(1–2):304–310
Dourish CT, Rycroft W, Iversen SD (1989) Postponement of satiety by blockade of brain cholecystokinin (CCK-B) receptors. Science 245(4925):1509–1511
Duncan EA, Davita G, Woods SC (2005) Changes in the satiating effect of cholecystokinin over repeated trials. Physiol Behav 85(4):387–393. doi:10.1016/j.physbeh.2005.04.023
Emond M, Schwartz GJ, Ladenheim EE, Moran TH (1999) Central leptin modulates behavioral and neural responsivity to CCK. Am J Physiol 276(5 Pt 2):R1545–R1549
Ervin GN, Mosher J, Birkemo LS, Johnson MF (1995) Multiple, small doses of cholecystokinin octapeptide are more efficacious at inducing taste aversion conditioning than single, large doses. Peptides 16(3):539–545
Feltrin KL, Little T, Meyer JH, Horowitz M, Smout AJ, Wishart J, Pilichiewicz AN, Rades T, Chapman IM, Feinle-Bisset C (2004) Effects of intraduodenal fatty acids on appetite, antropyloroduodenal motility, and plasma CCK and GLP-1 in humans vary with their chain length. Am J Physiol Regul Integr Comp Physiol 287(3):R524–R533
French SJ, Murray B, Rumsey RD, Fadzlin R, Read NW (1995) Adaptation to high-fat diets: effects on eating behaviour and plasma cholecystokinin. Br J Nutr 73(2):179–189
Friedman JM, Halaas JL (1998) Leptin and the regulation of body weight in mammals. Nature 395(6704):763–770
Gibbs J, Young RC, Smith GP (1973) Cholecystokinin decreases food intake in rats. J Comp Physiol Psychol 84(3):488–495
Goodison T, Siegel S (1995) Learning and tolerance to the intake suppressive effect of cholecystokinin in rats. Behav Neurosci 109(1):62–70
Guilmeau S, Buyse M, Tsocas A, Laigneau JP, Bado A (2003) Duodenal leptin stimulates cholecystokinin secretion: evidence of a positive leptin-cholecystokinin feedback loop. Diabetes 52(7):1664–1672
Guilmeau S, Buyse M, Bado A (2004) Gastric leptin: a new manager of gastrointestinal function. Curr Opin Pharmacol 4(6):561–566. doi:10.1016/j.coph.2004.06.008
Halatchev IG, Cone RD (2005) Peripheral administration of PYY(3-36) produces conditioned taste aversion in mice. Cell Metab 1(3):159–168. doi:10.1016/j.cmet.2005.02.003
Harris RB, Mitchell TD, Hebert S (2003) Leptin-induced changes in body composition in high fat-fed mice. Exp Biol Med (Maywood) 228(1):24–32
Horvath TL, Sarman B, Garcia-Caceres C, Enriori PJ, Sotonyi P, Shanabrough M, Borok E, Argente J, Chowen JA, Perez-Tilve D, Pfluger PT, Bronneke HS, Levin BE, Diano S, Cowley MA, Tschop MH (2010) Synaptic input organization of the melanocortin system predicts diet-induced hypothalamic reactive gliosis and obesity. Proc Natl Acad Sci USA 107(33):14875–14880. doi:10.1073/pnas.1004282107
Jakicic JM (2009) The effect of physical activity on body weight. Obesity (Silver Spring) 17(Suppl 3):S34–S38
Jaworek J, Bonior J, Konturek SJ, Bilski J, Szlachcic A, Pawlik WW (2003) Role of leptin in the control of postprandial pancreatic enzyme secretion. J Physiol Pharmacol 54(4):591–602
Kinzig KP, Honors MA, Hargrave SL, Davenport BM, Strader AD, Wendt D (2010) Sensitivity to the anorectic effects of leptin is retained in rats maintained on a ketogenic diet despite increased adiposity. Neuroendocrinology 92(2):100–111
Kissane NA, Pratt JS (2011) Medical and surgical treatment of obesity. Best Pract Res Clin Anaesthesiol 25(1):11–25
Konturek PC, Jaworek J, Maniatoglou A, Bonior J, Meixner H, Konturek SJ, Hahn EG (2002) Leptin modulates the inflammatory response in acute pancreatitis. Digestion 65(3):149–160
Kulkosky PJ (1985) Conditioned food aversions and satiety signals. Ann N Y Acad Sci 443:330–347
Levin BE, Dunn-Meynell AA (2002) Reduced central leptin sensitivity in rats with diet-induced obesity. Am J Physiol Regul Integr Comp Physiol 283(4):R941–R948. doi:10.1152/ajpregu.00245.2002
Levin BE, Dunn-Meynell AA (2006) Differential effects of exercise on body weight gain and adiposity in obesity-prone and -resistant rats. Int J Obes (Lond) 30(4):722–727. doi:10.1038/sj.ijo.0803192
Little TJ, Russo A, Meyer JH, Horowitz M, Smyth DR, Bellon M, Wishart JM, Jones KL, Feinle-Bisset C (2007) Free fatty acids have more potent effects on gastric emptying, gut hormones, and appetite than triacylglycerides. Gastroenterology 133(4):1124–1131
Maggio CA, Haraczkiewicz E, Vasselli JR (1988) Diet composition alters the satiety effect of cholecystokinin in lean and obese Zucker rats. Physiol Behav 43(4):485–491
Matson CA, Ritter RC (1999) Long-term CCK-leptin synergy suggests a role for CCK in the regulation of body weight. Am J Physiol 276(4 Pt 2):R1038–R1045
Matson CA, Wiater MF, Kuijper JL, Weigle DS (1997) Synergy between leptin and cholecystokinin (CCK) to control daily caloric intake. Peptides 18(8):1275–1278
Matson CA, Reid DF, Cannon TA, Ritter RC (2000) Cholecystokinin and leptin act synergistically to reduce body weight. Am J Physiol Regul Integr Comp Physiol 278(4):R882–R890
Matson CA, Reid DF, Ritter RC (2002) Daily CCK injection enhances reduction of body weight by chronic intracerebroventricular leptin infusion. Am J Physiol Regul Integr Comp Physiol 282(5):R1368–R1373
Moran TH, Ladenheim EE (2011) Adiposity signaling and meal size control. Physiol Behav 103(1):21–24. doi:10.1016/j.physbeh.2010.11.013
Mussa BM, Sartor DM, Verberne AJ (2008) Activation of cholecystokinin (CCK 1) and serotonin (5-HT 3) receptors increases the discharge of pancreatic vagal afferents. Eur J Pharmacol 601(1–3):198–206. doi:10.1016/j.ejphar.2008.11.007
National Task Force on the Prevention and Treatment of Obesity (2000) Overweight, obesity, and health risk. Arch Intern Med 160(7):898–904
Ozcan L, Ergin AS, Lu A, Chung J, Sarkar S, Nie D, Myers MG Jr, Ozcan U (2009) Endoplasmic reticulum stress plays a central role in development of leptin resistance. Cell Metab 9(1):35–51
Patel JD, Ebenezer IS (2008) The effect of intraperitoneal administration of leptin on short-term food intake in rats. Eur J Pharmacol 580(1–2):143–152
Paulino G, Barbier de la Serre C, Knotts TA, Oort PJ, Newman JW, Adams SH, Raybould HE (2009) Increased expression of receptors for orexigenic factors in nodose ganglion of diet-induced obese rats. Am J Physiol Endocrinol Metab 296(4):E898–E903. doi:10.1152/ajpendo.90796.2008
Peters JH, Karpiel AB, Ritter RC, Simasko SM (2004) Cooperative activation of cultured vagal afferent neurons by leptin and cholecystokinin. Endocrinology 145(8):3652–3657
Rossner S, Hammarstrand M, Hemmingsson E, Neovius M, Johansson K (2008) Long-term weight loss and weight-loss maintenance strategies. Obes Rev 9(6):624–630
Roth JD, Roland BL, Cole RL, Trevaskis JL, Weyer C, Koda JE, Anderson CM, Parkes DG, Baron AD (2008) Leptin responsiveness restored by amylin agonism in diet-induced obesity: evidence from nonclinical and clinical studies. Proc Natl Acad Sci USA 105(20):7257–7262. doi:10.1073/pnas.0706473105
Scarpace PJ, Zhang Y (2009) Leptin resistance: a predisposing factor for diet-induced obesity. Am J Physiol Regul Integr Comp Physiol 296(3):R493–R500. doi:10.1152/ajpregu.90669.2008
Schwartz MW, Seeley RJ, Campfield LA, Burn P, Baskin DG (1996) Identification of targets of leptin action in rat hypothalamus. J Clin Investig 98(5):1101–1106
Sobhani I, Bado A, Vissuzaine C, Buyse M, Kermorgant S, Laigneau JP, Attoub S, Lehy T, Henin D, Mignon M, Lewin MJ (2000) Leptin secretion and leptin receptor in the human stomach. Gut 47(2):178–183
Spannagel AW, Nakano I, Tawil T, Chey WY, Liddle RA, Green GM (1996) Adaptation to fat markedly increases pancreatic secretory response to intraduodenal fat in rats. Am J Physiol 270(1 Pt 1):G128–G135
Swartz TD, Duca FA, Covasa M (2010) Differential feeding behavior and neuronal responses to CCK in obesity-prone and -resistant rats. Brain Res 1308:79–86. doi:10.1016/j.brainres.2009.10.045
Torregrossa AM, Smith GP (2003) Two effects of high-fat diets on the satiating potency of cholecystokinin-8. Physiol Behav 78(1):19–25
Trevaskis JL, Turek VF, Griffin PS, Wittmer C, Parkes DG, Roth JD (2010) Multi-hormonal weight loss combinations in diet-induced obese rats: therapeutic potential of cholecystokinin? Physiol Behav 100(2):187–195. doi:10.1016/j.physbeh.2010.02.023
Wang Y, Beydoun MA (2007) The obesity epidemic in the United States—gender, age, socioeconomic, racial/ethnic, and geographic characteristics: a systematic review and meta-regression analysis. Epidemiol Rev 29:6–28
Wang YH, Tache Y, Sheibel AB, Go VL, Wei JY (1997) Two types of leptin-responsive gastric vagal afferent terminals: an in vitro single-unit study in rats. Am J Physiol 273(2 Pt 2):R833–R837
Weller A, Smith GP, Gibbs J (1990) Endogenous cholecystokinin reduces feeding in young rats. Science 247(4950):1589–1591
West DB, Fey D, Woods SC (1984) Cholecystokinin persistently suppresses meal size but not food intake in free-feeding rats. Am J Physiol 246(5 Pt 2):R776–R787
West DB, Greenwood MR, Marshall KA, Woods SC (1987) Lithium chloride, cholecystokinin and meal patterns: evidence that cholecystokinin suppresses meal size in rats without causing malaise. Appetite 8(3):221–227
Wu XM, Liao YW, Ji KQ, Li GF, Zang B (2011) The trophic effect of cholecystokinin on the pancreas declines in rats on total parenteral nutrition. J Anim Physiol Anim Nutr (Berl). doi:10.1111/j.1439-0396.2011.01140.x
Acknowledgments
We thank to Garcia Osornio E. for technical help, Daniel Garcia for help with Matlab programming and to Eric E. Thomson for discussion on this manuscript. This work was supported by CONACYT Grants 78879, Salud 2010-02-151001, ICYTDF-PICDS08-59 and Productos Medix 000652 to R.G.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Londoño, M., Tellez, L.A. & Gutierrez, R. Physiological Low Doses of Leptin and Cholecystokinin Induces Body Weight-Loss in Juvenile and Lean, but not in Adult-Obese Rats. Int J Pept Res Ther 18, 77–88 (2012). https://doi.org/10.1007/s10989-011-9281-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-011-9281-9