Abstract
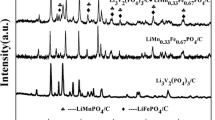
The compound Li9Cr3(P2O7)3(PO4)2 has been successfully synthesized using sol–gel method. X-ray diffraction Rietveld refinement analysis indicates that single phase Li9Cr3(P2O7)3(PO4)2 can be obtained under air condition and high purity nitrogen atmosphere. Scanning electron microscopy indicates that nanowires with lengths ranging from several to tens micrometers and diameters varying from 100nm to 500nm can be obtained in the Li9Cr3(P2O7)3(PO4)2 compound heated under air condition. The electrochemical properties of Li9Cr3(P2O7)3(PO4)2 sintered under N2 as cathode material is reported for the first time. The XRD patterns of the electrodes before and after 30 cycles indicate that the Li9Cr3(P2O7)3(PO4)2 keeps its original monodiphosphate structure.



Similar content being viewed by others
References
Padhi AK, Nanjundaswamy KS, Goodenough JB (1997) J Electrochem Soc 144:1188
Vadivel Murugan A, Muraliganth T, Manthiram A (2009) J Electrochem Soc 156:A79
Zhang Y, Sun CS, Zhou Z (2009) Electrochem Commun 11:1183
Zhu X, Liu Y, Geng L, Chen L, Liu H, Cao M (2008) Solid State Ionics 179:1679
Fu P, Zhao Y, An X, Dong Y, Hou X (2007) Electrochim Acta 52:5281
Chen Q, Wang J, Tang Z, He W, Shao H, Zhang J (2007) Electrochim Acta 52:5251
Wu C, Wu F, Chen L, Huang X (2002) Solid State Ionics 152–153:335
Sigala C, Verbaere A, Mansot JL, Guyomard D, Piffard Y, Tournoux M (1997) J Solid State Chem 132:372
Poisson S, d’Yvoire F, Guyen-Huy-Dung N, Bretey E, Berthet P (1998) J Solid State Chem 138:32
Capitelli F, Dridi N, Arbib EH, Valentini V, Mattei G (2007) Z Kristallogr 222:521
Acknowledgments
Financial support from an NSFC Grant (No. 50972046) funded by the NSFC Committee of China and the Fundamental Research Funds for the Central University SCUT (No. 2009ZM0022) is gratefully appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, J., Zhao, Y. & Kuang, Q. Preparation of Li9Cr3(P2O7)3(PO4)2 as cathode material for lithium ion batteries through sol–gel method. J Sol-Gel Sci Technol 59, 521–524 (2011). https://doi.org/10.1007/s10971-011-2521-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-011-2521-9