Abstract
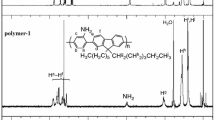
Phenazine-containing poly(phenylenevinylene) (P(PHN-PV)) was synthesized using Wittig-Horner polycondensation of the appropriately designed monomers viz. 5,10-dioctyl-5,10-dihydrophenazine-2,7-dicarbaldehyde and tetraethyl ((2,5-bis((2-ethylhexyl) oxy)-1,4 phenylene) bis(methylene)) bis(phosphonate). The design embraces the specific motivation of incorporating the nitrogen-containing heterocycle viz. .phenazine in poly(phenylenevinylene) backbone. P(PHN-PV) exhibited reversible redox properties. In the field emission measurements performed on the film of P(PHN-PV), the turn-on field was observed to be 1.93 V/μm for the current density of 10 μA/cm2. The maximum current density of ~4.87 mA/cm2 was achieved at the applied field of 3.84 V/μm. The emission current showed impressive stability for 6 h at a constant current of 1 μA (current density of about 20 μA/cm2). These results emphasize the role of phenazine heterocycle with lone pair of electrons on nitrogen in lowering the oxidation onset and in turn reduction of the turn-on voltage.









Similar content being viewed by others
References
Lin Z, Wang J (eds) (2014) Low-cost nanomaterials: Toward greener and more efficient energy applications. Springer - Verlag London, VI, 488. https://doi.org/10.1007/978-1-4471-6473-9
Torres T, Bottari G (eds) (2013) Organic nanomaterials: synthesis, characterization, and device applications. John Wiley & Sons, Inc., Hoboken
Burchell TD (1999) Carbon materials for advanced technologies. Elsevier Science, Oak Ridge, p 540
Jia C, Ma B, Xin N, Guo X (2015). Acc Chem Res 48:2565–2575
Pan J, Sheng Y, Zhang J, Huang P, Zhang X, Feng B (2015). ACS Appl Mater Interfaces 7:7878–7883
Yang C, Wei H, Guan L, Guo J, Wang Y, Yan X, Zhang X, Wei S, Guo Z (2015). J Mater Chem A 3:14929
Patil SS, Jha P, Aswal DK, Gupta SK, Yakhmi JV, Joag DS, More MA (2012). Polym Adv Technol 23:215–219
Wang C, Wang Z, Li M, Li H (2001). Chem Phys Lett 341:431–434
Patil SS, Harpale KV, Koiry SP, Patil KR, Aswal DK, More MA (2015). J Appl Polym Sci 132:5
Sameera I, Bhatia R, Ouyang J, Prasad V, Menon R (2013). Appl Phys Lett 102:033102
Ding H, Feng T, Chen Y, Sun Z (2012). Appl Surf Sci 25:5191–5194
Yan H, Zhang L, Shen J, Chen Z, Shi G, Zhang B (2006). Nanotechnology 17:3446–3450
Patil SS, Koiry SP, Veerender P, Aswal DK, Gupta SK, Joag DS, More MA (2012). RSC Adv Technol 2:5822–5827
Kim BH, Park DH, Joo J, Yu SG, Lee SH (2005). Synth Met 150:279–284
Musa I, Munindrasdasa DAI, Amaratunga GAJ, Eccleston W (1998). Nature 395:362
Sridhar S, Tiwary C, Vinod S, Taha-Tijerina JJ, Sridhar S, Kalaga K, Sirota B, Hart AHC, Ozden S, Sinha RK, Harsh VR, Choi W, Kordás K, Ajayan PM (2014). ACS Nano 8:7763–7770
Kim BH, Kim MS, Park KT, Lee JK, Park DH, Joo J, Yu SG, Lee SH (2003). Appl Phys Lett 83:539
Rujia Z, Zhang Z, Jiang L, Xu K, Tian Q, Xue S, Hu J, Bandob Y, Golberg D (2012). J Mater Chem 22:19196
Chen N, Qian X, Lin H, Liua H, Li Y (2012). J Mater Chem 22:11068
Viskadouros GM, Stylianakis MM, Kymakis E, Stratakis E (2014). ACS Appl Mater Interfaces 6:388–393
Alexandrou I, Kymakis E, Amaratunga GAJ (2002). Appl Phys Lett 80:1435
Jin YW, Jung JE, Park YJ, Choi JH, Jung DS, Lee HW, Park SH, Lee NS, Kim JM, Ko TY, Lee SJ, Hwang SY, You JH, Yoo JB, Park CY (2002). J Appl Phys 92:1065
Okano K, Koizumi S, Silva SP, Amaratunga GAJ (1996). Nature 381:140–141
Amaratunga GAJ, Silva SRP (1996). Appl Phys Lett 68:2529–2531
Bagher AM (2014). Sustainable Energy 2:85–90
Mihailetchi VD, Koster LJA, Blom PWM, Melzer C, de Boer B, van Duren JKJ, Janssen RAJ (2005). Adv Funct Mater 15:795–801
Suppiah S, Mohamad SM, Juhari N (2011) Fabrication of MEH-PPV based organic light emitting diode and transistor. IEEE Regional Symposium on Micro and Nanoelectronics (RSM), 28-30 Sept 2011. doi:https://doi.org/10.1109/RSM.2011.6088367
Lei T, Xia X, Wang JY, Liu CJ, Pei J (2014). J Am Chem Soc 136:2135–2141
Lei T, Dou JH, Cao XY, Wang JY, Pei J (2013). J Am Chem Soc 135:12168–12171
Todescato F, Capelli R, Dinelli F, Murgia M, Camaioni N, Yang M, Bozio R, Muccini M (2008). J Phys Chem B 112:10130–10136
Hiraoka S, Okamoto T, Kozaki M, Shiomi D, Sato K, Takui T, Okada K (2004). J Am Chem Soc 126:58–59
Terada E, Okamoto T, Kozaki M, Masaki ME, Shiomi D, Sato K, Takui T, Okada K (2005). J Organomet Chem 70:10073–10081
Thalladi VR, Smolka T, Gehrke A, Boese R, Sustmann R (2000). New J Chem 24:143–147
Price-Whelan A, Dietrich LEP, Newman DK (2006). Nat Chem Biol 2:71–78
Pierson IIILS, Pierson EA (2010). Appl Microbiol Biotechnol 86:1659–1670
Zheng Z, Dong Q, Gou L, Su JH, Huang J (2014). J Mater Chem C 2:9858–9865
Gu PY, Zhao Y, He JH, Zhang J, Wang C, Xu QF, Lu JM, Sun XW, Zhang QJ (2015). Org Chem 80:3030–3035
Lee J, Shizu K, Tanaka H, Nakanotani H, Yasuda T, Kaji H, Adachi C (2015). J Mater Chem C 3:2175–2181
Song HJ, Goh M, Choi KH, Lee S, Moon DK, Shin GJ (2015). J IndEng Chem 23:338–343
Lee DC, Brownell LV, Yan L, You W (2014). ACS Appl Mater Interfaces 6:15767–15773
Li G, Lu Z, Li C, Bo Z (2015). Polym Chem 6:1613–1618
Lu X, Lan T, Qin Z, Wang ZS, Zhou G (2014). ACS Appl Mater Interfaces 6:19308–19317
Richard CA, Pan Z, Hsu HY, Cekli S, Schanze KS, Reynolds JR (2014). ACS Appl Mater Interfaces 6:5221–5227
Yang L, Li X, Yang J, Qu Y, Hua J (2013). ACS Appl Mater Interfaces 5:1317–1326
Wang B, Wasielewski MR (1997). J Am Chem Soc 119:12–21
Liu B, Yu WL, Pei J, Liu SY, Lai YH, Huang W (2001). Macromolecules 34:7932–7940
Iwasa Y, Koda T, Koshihara S, Tokura Y, Iwasawa N, Saito G (1989). Phys Rev B: Condens Matter Mater Phys 39:10441–10444
Okamoto T, Terada E, Kozaki M, Uchida M, Kikukawa S, Okada K (2003). Org Lett 5:373–376
Koike R, Katayose Y, Ohta A, Motoyoshiya J, Nishii Y, Aoyam H (2005). Tetrahedron 61:11020–11026
Narasimha K, Jayakannan M (2014). Appl Mater Interfaces 6:19385–19396
Fowler RH, Nordheim L (1928). R Soc Lond A 119:173
Acknowledgements
The authors would like to thank Dr. Satishchandra B. Ogale (Centre of Excellence in Solar Energy, Physical and Materials Chemistry Division, CSIR-National Chemical Laboratory (CSIR-NCL), Pune, India) for valuable support. The authors also thank Dr. Dipti Dhakaras for conductivity measurements, Dr. Meenal Deo for help with Mott Schottky measurements, Mukta Tathavadekar for spray coating and Dr. Manoj Mane for DFT calculations. Shraddha Chhatre gratefully acknowledges the CSIR India, for the fellowship.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
ESM 1
(DOC 6250 kb)
Rights and permissions
About this article
Cite this article
Chhatre, S., Ichake, A., Harpale, K. et al. Phenazine-containing poly(phenylenevinylene): a new polymer with impressive field emission properties. J Polym Res 25, 61 (2018). https://doi.org/10.1007/s10965-017-1428-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-017-1428-8