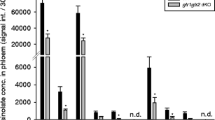
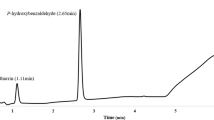
Abstract
In this study we examined the role of sorghum flavonoids in providing resistance against corn leaf aphid (CLA) Rhopalosiphum maidis. In sorghum, accumulation of these flavonoids is regulated by a MYB transcription factor, yellow seed1 (y1). Functional y1 alleles accumulate 3-deoxyflavonoids (3-DFs) and 3—deoxyanthocyanidins (3-DAs) whereas null y1 alleles fail to accumulate these compounds. We found that significantly higher numbers of alate CLA adults colonized null y1 plants as compared to functional y1 plants. Controlled cage experiments and pairwise choice assays demonstrated that apterous aphids preferred to feed and reproduce on null y1 plants. These near-isogenic sorghum lines do not differ in their epicuticular wax content and were also devoid of any leaf trichomes. Significantly higher mortality of CLA was observed on artificial aphid diet supplemented with flavonoids obtained from functional y1 plants as compared to null y1 plants or the relevant controls. Our results demonstrate that the proximate mechanism underlying the deleterious effects on aphids is y1-regulated flavonoids which are important defense compounds against CLA.







Similar content being viewed by others
References
Anjali M, Sridevi G, Prabhakar M et al (2017) Dynamic changes in carotenoid and flavonoid content and relative water content (RWC) by corn leaf aphid infestation on sorghum. J Pharmacogn Phytochem 6:1240–1245
Ashley JN, Hobbs BC, Raistrick H (1937) Studies in the biochemistry of micro-organisms: the crystalline colouring matters of Fusarium culmorum (WG Smith) Sacc. and related forms. Biochem J 31:385
Ateyyat M, Abu-Romman S, Abu-Darwish M, Ghabeish I (2012) Impact of flavonoids against woolly apple aphid, Eriosoma lanigerum (Hausmann) and its sole parasitoid, Aphelinus mali (Hald.). J Agric Sci 4:227
Bass C, Puinean AM, Zimmer CT et al (2014) The evolution of insecticide resistance in the peach potato aphid, Myzus persicae. Insect Biochem Mol Biol 51:41–51
Betsiashvili M, Ahern KR, Jander G (2014) Additive effects of two quantitative trait loci that confer Rhopalosiphum maidis (corn leaf aphid) resistance in maize inbred line Mo17. J Exp Bot 66:571–578
Boddu J, Jiang CH, Sangar V et al (2006) Comparative structural and functional characterization of sorghum and maize duplications containing orthologous Myb transcription regulators of 3-deoxyflavonoid biosynthesis. Plant Mol Biol 60:185–199
Boveris AD, Galatro A, Sambrotta L et al (2001) Antioxidant capacity of a 3-deoxyanthocyanidin from soybean. Phytochemistry 58:1097–1105. https://doi.org/10.1016/s0031-9422(01)00378-8
Campbell BC, Dreyer DL (1985) Host-plant resistance of sorghum: differential hydrolysis of sorghum pectic substances by polysaccharases of greenbug biotypes (Schizaphis graminum, homoptera: Aphididae). Arch Insect Biochem Physiol 2:203–215
Carvalho CHS, Boddu J, Zehr UB et al (2005) Genetic and molecular characterization of Candystripe1 transposition events in sorghum. Genetica 124:201–212
Cheng WN, Lei JX, Rooney WL et al (2013) High basal defense gene expression determines sorghum resistance to the whorl-feeding insect southwestern corn borer. Insect Sci 20:307–317. https://doi.org/10.1111/1744-7917
Chopra S, Brendel V, Zhang JB et al (1999) Molecular characterization of a mutable pigmentation phenotype and isolation of the first active transposable element from Sorghum bicolor. Proc Natl Acad Sci U S A 96:15330–15335
Costa-Arbulú C, Gianoli E, Gonzáles WL, Niemeyer HM (2001) Feeding by the aphid Sipha flava produces a reddish spot on leaves of Sorghum halepense: an induced defense. J Chem Ecol 27:273–283
Czerniewicz P, Sytykiewicz H, Durak R et al (2017) Role of phenolic compounds during antioxidative responses of winter triticale to aphid and beetle attack. Plant Physiol Biochem 118:529–540
Dadd RH, Mittler TE (1965) Studies on the artificial feeding of the aphid Myzus persicae (Sulzer)—III. Some major nutritional requirements. J Insect Physiol 11:717–743
Dayan FE, Rimando AM, Pan ZQ et al (2010) Sorgoleone. Phytochemistry 71:1032–1039. https://doi.org/10.1016/j.phytochem.2010.03.011
Dreyer DL, Reese JC, Jones KC (1981) Aphid feeding deterrents in sorghum. J Chem Ecol 7:273–284
Eigenbrode SD, Espelie KE (1995) Effects of plant Epicuticular lipids on insect herbivores. Annu Rev Entomol 40:171–194. https://doi.org/10.1146/annurev.en.40.010195.001131
Ejeta G, Knoll JE (2007) Marker-assisted selection in sorghum. In: Genomics-assisted crop improvement. Springer, Dordrecht, pp 187–205
van Emden HF, Harrington R (2007) Aphids as crop pests. Cabi
Goffreda JC, Mutschler MA, Tingey WM (1988) Feeding behavior of potato aphid affected by glandular trichomes of wild tomato. Entomol Exp Appl 48:101–107
Goławska S, Łukasik I, Goławski A et al (2010) Alfalfa (Medicago sativa L.) apigenin glycosides and their effect on the pea aphid (Acyrthosiphon pisum). Polish J Environ Stud 19:913–919
Goławska S, Łukasik I, Kapusta I, Janda B (2012) Do the contents of luteolin, tricin, and chrysoeriol glycosides in alfalfa (Medicago sativa L.) affect the behavior of pea aphid (Acyrthosiphon pisum)? Polish J Environ Stud 21:1613–1619
Grayer R, Harborne J, Kimmins F et al (1993) Phenolics in rice phloem sap as sucking deterrents to the brown planthopper, Nilaparvata lugens. In: International symposium on natural phenols in plant resistance. Acta Hortic 381:691–694
Grotewold E, Chamberlin M, Snook M et al (1998) Engineering secondary metabolism in maize cells by ectopic expression of transcription factors. Plant Cell 10:721–740
Heim D, Nicholson RL, Pascholati SF et al (1983) Etiolated maize mesocotyls: a tool for investigating disease interactions. Phytopathology 73:424–428
Hijaz F, Manthey JA, Van der Merwe D, Killiny N (2016) Nucleotides, micro-and macro-nutrients, limonoids, flavonoids, and hydroxycinnamates composition in the phloem sap of sweet orange. Plant Signal Behav 11:e1183084
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66. https://doi.org/10.1146/annurev.arplant.59.032607.092825
Ibraheem F, Gaffoor I, Chopra S (2010) Flavonoid phytoalexin dependent resistance to anthracnose leaf blight requires a functional yellow seed1 in Sorghum bicolor. Genetics 184:915–926
Ibraheem F, Gaffoor I, Tan Q, Shyu CR, Chopra S (2015) A sorghum MYB transcription factor induces 3-deoxyanthocyanidins and enhances resistance against leaf blights in maize. Molecules 20:2388–2404. https://doi.org/10.3390/molecules20022388
Kariyat RR, Smith JD, Stephenson AG et al (2017) Non-glandular trichomes of Solanum carolinense deter feeding by Manduca sexta caterpillars and cause damage to the gut peritrophic matrix. Proc R Soc B 284:20162323
Lattanzio V, Arpaia S, Cardinali A et al (2000) Role of endogenous flavonoids in resistance mechanism of Vigna to aphids. J Agric Food Chem 48:5316–5320
Lipsey B (2017) Management strategies for sugarcane aphid, Melanaphis sacchri (Zehntner), in grain sorghum. Mississippi State Univ
Lo SCC, Nicholson RL (1998) Reduction of light-induced anthocyanin accumulation in inoculated sorghum mesocotyls - implications for a compensatory role in the defense response. Plant Physiol 116:979–989
Lo SCC, De Verdier K, Nicholson RL (1999) Accumulation of 3-deoxyanthocyanidin phytoalexins and resistance to Colletotrichum sublineolum in sorghum. Physiol Mol Plant Pathol 55:263–273
Louis J, Leung Q, Pegadaraju V et al (2010) PAD4-dependent antibiosis contributes to the ssi2-conferred hyper-resistance to the green peach aphid. Mol Plant-Microbe Interact 23:618–627
Malathi P, Viswanathan R, Padmanaban P et al (2008) Differential accumulation of 3-deoxy anthocyanidin phytoalexins in sugarcane varieties varying in red rot resistance in response to Colletotrichum falcatum infection. Sugar Tech 10:154–157
Mizuno H, Kawahigashi H, Kawahara Y et al (2012) Global transcriptome analysis reveals distinct expression among duplicated genes during sorghum-interaction. BMC Plant Biol 12:121. https://doi.org/10.1186/1471-2229-12-121
Morkunas I, Woźniak A, Formela M et al (2016) Pea aphid infestation induces changes in flavonoids, antioxidative defence, soluble sugars and sugar transporter expression in leaves of pea seedlings. Protoplasma 253:1063–1079
Mote UN, Shahane AK (1993) Studies on varietal reaction of sorghum to Delphacid, aphid and leaf sugary exudation. Indian J Entomol 55:360–367
Nicholson RL, Kollipara SS, Vincent JR et al (1987) Phytoalexin synthesis by the sorghum mesocotyl in response to infection by pathogenic and nonpathogenic fungi. Proc Natl Acad Sci U S A 84:5520–5524
Pompon J, Quiring D, Giordanengo P, Pelletier Y (2010) Role of xylem consumption on osmoregulation in Macrosiphum euphorbiae (Thomas). J Insect Physiol 56:610–615
Pompon J, Quiring D, Goyer C et al (2011) A phloem-sap feeder mixes phloem and xylem sap to regulate osmotic potential. J Insect Physiol 57:1317–1322
Powell G, Tosh CR, Hardie J (2006) Host plant selection by aphids: behavioral, evolutionary, and applied perspectives. Annu Rev Entomol 51:309–330
Premachandra GS, Hahn DT, Joly RJ (1993) A simple method for determination of abaxial and adaxial epicuticular wax loads in intact leaves of Sorghum bicolor L. Can J Plant Sci 73:521–524
Sharma HC (1993) Host-plant resistance to insects in sorghum and its role in integrated pest management. Crop Prot 12:11–34. https://doi.org/10.1016/0261-2194(93)90015-B
Sharma HC, Bhagwat VR, Daware DG et al (2014) Identification of sorghum genotypes with resistance to the sugarcane aphid Melanaphis sacchari under natural and artificial infestation. Plant Breed 133:36–44
Smith GR, Borg Z, Lockhart BEL et al (2000) Sugarcane yellow leaf virus: a novel member of the Luteoviridae that probably arose by inter-species recombination. J Gen Virol 81:1865–1869
Snyder BA, Nicholson RL (1990) Synthesis of phytoalexins in sorghum as a site-specific response to fungal ingress. Science (80- ) 248:1637–1639
Toler RW (1985) Maize dwarf mosaic, the most important virus disease of sorghum. Plant Dis 69:1011–1015
Tsumuki H, Kanehisa K, Kawada K (1989) Leaf surface wax as a possible resistance factor of barley to cereal aphids. Appl Ent Zool 24:294–301. https://doi.org/10.1303/aez.24.295
Walling LL (2008) Avoiding effective defenses: strategies employed by phloem-feeding insects. Plant Physiol 146:859–866
Webster JA, Inayatullah C, Hamissou M, Mirkes KA (1994) Leaf pubescence effects in wheat on yellow sugarcane aphids and greenbugs (Homoptera: Aphididae). J Econ Entomol 87:231–240. https://doi.org/10.1093/jee/87.1.231
Winefield CS, Lewis DH, Swinny EE et al (2005) Investigation of the biosynthesis of 3-deoxyanthocyanins in Sinningia cardinalis. Physiol Plant 124:419–430. https://doi.org/10.1111/j.1399-3054.2005.00531.x
Young WR, Teetes GL (1977) Sorghum entomology. Annu Rev Entomol 22:193–218
Zummo N (2000) Rough leaf spot. In: Frederiksen RA, Odovody GN (eds) Compendium Sorghum Diseases. APS Press, St. Paul, p 15
Züst T, Agrawal AA (2016) Mechanisms and evolution of plant resistance to aphids. Nat plants 2:15206
Acknowledgements
This work was supported by a USDA/NIFA award 2011-67009-30017, AES awards PEN04430 and PEN04613 to SC, and start up research funds from UTRGV for RK, and an Undergraduate Research Award, Penn State College of Ag. to Cullen Dixon. We thank Dr. Hari C. Sharma for his expertise and suggestions on this study, Thomas Eubanks for assisting with electron microscopy, Scott DiLoreto for greenhouse management, Scott Harkcom for tending sorghum nurseries at Penn State Agronomy farm, Dinakaran Elango with sorghum seed multiplication, and Kameron Wittmeyer for critical reading of this manuscript. We also wish to express our gratitude for annonymous reviewers for their constructive criticisms to help finalize this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Fig. S1
Effect of Flavan-4-ols on corn leaf aphid (CLA). Aphids fed on artificial diet supplemented with flavonoid compounds extracted from the developing pericarps of either Y1-rr3 seeds (green) containing flavan-4-ols or y1-ww4 seeds (purple) were observed and compared to the control treatments of either diet only (blue), or diet supplemented with 0.5% ethanol (red). The number of live aphids feeding and walking, dead aphids and live and dead nymphs were quantified 24 (A), 48 (B), and 72 (C) hr. after start of the experiment. (PNG 53 kb)
Fig. S2
Time course of effect of 3-DAs on corn leaf aphid (CLA) survival. Adult survival (A) and offspring production (B) was assessed using an artificial diet assay. Aphid and nymphs were counted up till four days and each treatment group had four biological replicates. Differences in aphid numbers between treatments were analyzed by Two way ANOVA with time and treatment as factors. Means that do not share a letter are significantly different after post hoc Tukey test at P < 0.05. (PNG 23 kb)
Rights and permissions
About this article
Cite this article
Kariyat, R.R., Gaffoor, I., Sattar, S. et al. Sorghum 3-Deoxyanthocyanidin Flavonoids Confer Resistance against Corn Leaf Aphid. J Chem Ecol 45, 502–514 (2019). https://doi.org/10.1007/s10886-019-01062-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-019-01062-8