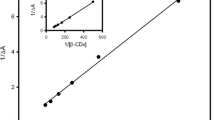
Abstract
The absorption and emission characteristics of donor−acceptor charge transfer system 4-amino-3-methyl benzoic acid methyl ester (AMBME), capable of dual emission, i.e., local emission (LE) and charge transfer (CT) emission, have been investigated inside the β-cyclodextrin (β-CD) nanocavity in the aqueous and non-aqueous dimethylsulphoxide (DMSO) medium. Large enhancement of both LE and CT band in aqueous β-CD medium is due to decrease in non-radiative processes in less polar and restricted environment. Whereas in non-aqueous DMSO medium the CT process is hindered as a result CT intensity decreases with enhancement of LE band. These spectral differences indicate that in aqueous medium the donor –NH2 group sticking in the hydrophilic region of β-CD cavity whereas in non-aqueous DMSO medium it exists in the hydrophobic part of the cavity. Spectral characteristics indicate that different stoichiometry of host–guest inclusion complexes are formed in aqueous and non-aqueous β-CD medium.






Similar content being viewed by others
References
Lakowicz, J.R.: Principle of Fluorescence Spectroscopy. New York, Plenum Press (1983)
Kalyanasundaram, K.: Photochemistry in Microheterogeneous Systems. Academic Press, New York (1987)
Bendr, M.L., Komiyama, M.: Cyclodextrin Chemistry. Springer, New York (1978)
Kalfter, J., Drake, J.M.: Molecular Dynamics in Restricted Geometries. Wiley, New York (1989)
Smith, V.K., Ndou, T.T., Warner, I.: J. Phys. Chem. 98, 8627 (1994)
Catena, G.C., Bright, F.V.: Anal. Chem. 61, 905 (1989)
Petkov, I., Petinova, A., Stoyanov, S.S., Metsov, S., Stoyanov, S.I.: J. Incl. Phenom. Macrocycl. Chem. 60, 329 (2007)
Dawoud, A.A., Al-Rawashdeh, N.: J. Incl. Phenom. Macrocycl. Chem. 60, 293 (2007)
Ozawa, R., Hayashita, T., Matsui, T., Nakayama, C., Yamauchi, A., Suzuki, I.: J. Incl. Phenom. Macrocycl. Chem. 60, 253 (2007)
Wood, D.J., Hruska, F.E., Saenger, W.: J. Am. Chem. Soc. 99, 1735 (1977)
Hamasaki, K., Ikeda, H., Nakamura, A., Ueno, A., Toda, F., Suzuki, I., Osa, T.: J. Am. Chem. Soc. 115, 5035 (1993)
Schneider, H.-J., Hacket, F., Rudiger, V.: Chem. Rev. 98, 1755 (1998)
Bratu, I., Gavira-Vallejo, J.M., Hernanz, A., Bogdan, M., Bora, G.: Biopolymers 73, 451 (2004)
Shayira Banu, H., Pitchumani, K., Srinivasan, C.: J. Photochem. Photobiol. A: Chem. 131, 101 (2000)
Chelli, S., Majdoub, M., Jouini, M., Aeiyach, S., Maurel, F., Chane-Ching, K.I., Lacaze, P.-C.: J. Phys. Org. Chem. 20, 30 (2007)
Adeagbo, W.A., Buss, V., Entel, P.: Multinatl. J. 77, 53 (2004)
Grabowski, Z.R., Rotkiewicz, K., Rettig, W.: Chem. Rev. 103, 3899 (2003)
Rotkiewicz, K., Grellmann, K.H., Grabowski, Z.R.: Chem. Phys. Lett. 19, 315 (1973)
Mataga, N., Yao, H., Okada, T., Rettig, W.: J. Phys. Chem. 93, 3383 (1989)
Kajimoto, O., Futakami, M., Kobayashi, T., Yamasaki, K.: J. Phys. Chem. 92, 1347 (1988)
Nag, A., Bhattachayya, K.: Chem. Phys. Lett. 151, 474 (1988)
Cox, G.S., Turro, J.J.: Am. Chem. Soc. 106, 422 (1984)
Al-Hasan, K.A., Klein, U.K.A., Suwaiyan, A.: Chem. Phys. Lett. 212, 581 (1993)
Kim, Y.H., Cho, D.W., Yoon, M., Kim, D.: J. Phys. Chem. 100, 15670 (1996)
Paja, S., Bangal, P.R., Chakravorti, S.: Chem. Phys. Lett. 329, 377 (2000)
Chakraborty, A., Kar, S., Nath, D.N., Guchhait, N.: J. Chem. Sci. 119, 195 (2007)
Street, K.W., Acree, W.E.: Appl. Spectrosc. 42, 1315 (1988)
Jiang, Y.-B.: J. Photochem. Photobiol. A: Chem. 88, 109 (1995)
Stalin, T., Shanthi, B., Rani, V., Rajendiran, N.: J. Incl. Phenom. Macrocycl. Chem. 55, 21 (2006)
Stalin, T., Rajendiran, N.: J. Photochem. Photobiol. A: Chem. 182, 137 (2006)
Stalin, T., Rajendiran, N.: Chem. Phys. 322, 311 (2006)
Das, S.K.: Chem. Phys. Lett. 361, 21 (2002)
Acknowledgements
NG gratefully acknowledges the financial support received from Department of Science and Technology, India (Project No. SP/S1/PC-1/2003). The authors thank Dr. P. K. Gupta of CAT, Indore for providing fluorescence lifetime measurement facility and Dr. Kaustuv Das, CAT, Indore for fluorescence lifetime measurement.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chakraborty, A., Guchhait, N. Inclusion complex of charge transfer probe 4-amino-3-methyl benzoic acid methyl ester (AMBME) with β-CD in aqueous and non-aqueous medium: medium dependent stoichiometry of the complex and orientation of probe molecule inside β-CD nanocavity. J Incl Phenom Macrocycl Chem 62, 91–97 (2008). https://doi.org/10.1007/s10847-008-9442-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-008-9442-4