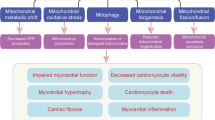
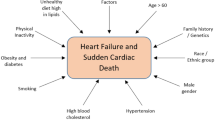
Abstract
The development of a diabetic cardiomyopathy is a multifactorial process, and evidence is accumulating that defects in intracellular free calcium concentration [Ca2+]i or its homeostasis are related to impaired mechanical performance of the diabetic heart leading to a reduction in contractile dysfunction. Defects in ryanodine receptor, reduced activity of the sarcoplasmic reticulum calcium pump (SERCA) and, along with reduced activity of the sodium-calcium exchanger (NCX) and alterations in myofilament, collectively cause a calcium imbalance within the diabetic cardiomyocytes. This in turn is characterized by cytosolic calcium overloading or elevated diastolic calcium leading to heart failure. Numerous studies have been performed to identify the cellular, subcellular, and molecular derangements in diabetes-induced cardiomyopathy (DCM), but the precise mechanism(s) is still unknown. This review focuses on the mechanism behind DCM, the onset of contractile dysfunction, and the associated changes with special emphasis on hyperglycemia, mitochondrial dysfunction in the diabetic heart. Further, management strategies, including treatment and emerging therapeutic modalities, are discussed.



Similar content being viewed by others
References
Jacobsen IB, Henriksen JE, Hother-Nielsen O, Vach W, Beck-Nielsen H (2009) Evidence-based insulin treatment in type 1 diabetes mellitus. Diabetes Res Clin Pract 86:1–10
American Diabetes Association (2009) Diagnosis and classification of diabetes mellitus. Diabetes Care 32(Suppl 1):S62–S67
Pereira L, Matthes J, Schuster I, Valdivia HH, Herzig S, Richard S, Gomez AM (2006) Mechanisms of [Ca2+]i transient decrease in cardiomyopathy of db/db type 2 diabetic mice. Diabetes 55:608–615
Schafer SA, Machicao F, Fritsche A, Häring HU, Kantartzis K (2011) New type 2 diabetes risk genes provide new insights in insulin secretion mechanisms. Diabetes Res Clin Pract 93(Suppl 1):S9–24
An D, Rodrigues B (2006) Role of changes in cardiac metabolism in development of diabetic cardiomyopathy. Am J Physiol Heart Circ Physiol 291:H1489–H1506
Long A, Jack SD (2011) The comorbidities of diabetes and hypertension: mechanisms and approach to target organ protection. J Clin Hypertens (Greenwich) 13(4):244–251. https://doi.org/10.1111/j.1751-7176.2011.00434.x
NCD Risk Factor Collaboration (2016) Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4·4 million participants. Lancet 387:1513–1530
Anand SS, Yusuf S (2011) Stemming the global tsunami of cardiovascular disease. Lancet 377:529–532
Singh RM, Cummings E, Pantos C, Singh J (2017) Protein kinase C and cardiac dysfunction: a review. Heart Fail Rev 22:843–859. https://doi.org/10.1007/s10741-017-9634-3
Pappachan JM, Varughese GI, Sriraman R, Arunagirinathan G (2013) Diabetic cardiomyopathy: pathophysiology, diagnostic evaluation and management. World J Diabetes 4:177–189
Kannel WB, McGee DL (1979) Diabetes and cardiovascular disease: the Framingham study. JAMA 241:2035–2038
Lee WS, Kim J (2017) Diabetic cardiomyopathy: where we are and where we are going. Korean J Intern Med 32:404–421
Isfort M, Stevens SCW, Schaffer S, Jong CJ, Wold LE (2014) Metabolic dysfunction in diabetic cardiomyopathy. Heart Fail Rev 19(1):9377–9388
Aneja A, Tang WH, Bansilal S, Garcia MJ, Farkouh ME (2008) Diabetic cardiomyopathy: insights into pathogenesis, diagnostic challenges, and therapeutic options. Am J Med 121:748–757
Gilca GM, Stefanescu G, Badulescu O, Tanase DM, Bararu I, Ciocoiu M (2017) Diabetic cardiomyopathy: current approach and potential diagnostic and therapeutic targets. J Diabetes Res. https://doi.org/10.1155/2017/1310265
Wang ZV, Hill JA (2015) Diabetic cardiomyopathy: catabolism-driving metabolism. Circulation 131:771–773
Boudina S, Abel ED (2007) Diabetic cardiomyopathy revisited. Circulation 115:3213–3223
Belke DD, Larsen TS, Gibbs EM, Severson DL (2000) Altered metabolism causes cardiac dysfunction in perfused hearts from diabetic (db/db) mice. Am J Physiol Endocrinol Metab 279:E1104–E1113
Bhimji S, Godin DV, McNeill JH (1986) Myocardial ultrastructural changes in alloxan-induced diabetes in rabbits. Acta Anat (Basel) 125:195–200
Forsterman U, Munzel T (2006) Endothelial nitric oxide synthase in vascular disease: from marvel to menace. Circulation 113:1708–1714
Jia G, DeMarco VG, Sowers JR (2016) Insulin resistance and hyperinsulinaemia in diabetic cardiomyopathy. Nat Rev Endocrinol 12:144–153
Nishikawa T, Edelstein D, Du XL, Yamagishi S, Matsumura T, Kaneda Y, Yorek MA, Beebe D, Oates PJ, Hammes HP, Giardino I, Brownlee M (2000) Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 404:787–790
Choi SW, Benzie IF, Ma SW, Strain JJ, Hannigan BM (2008) Acute hyperglycaemia and oxidative stress: direct cause and effect? Free Radic Biol Med 44:1217–1231
Borradaile NM, Han X, Harp JD, Gale SE, Ory DS, Schaffer JE (2006) Disruption of endoplasmic reticulum structure and integrity in lipotoxic cell death. J Lipid Res 47:2726–2737
Lebeche D, Davidoff AJ, Hajjar RJ (2008) Interplay between impaired calcium regulation and insulin signaling abnormalities in diabetic cardiomyopathy. Nat Clin Pract Cardiovasc Med 5:715–724
Minamino T, Komuro I, Kitakaze M (2010) Endoplasmic reticulum stress as a therapeutic target in cardiovascular disease. Circ Res 107(9):1071–1082
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414:813–820
Nishikawa T, Edelstein D, Brownlee M (2000) The missing link: a single unifying mechanism for diabetic complications. Kidney Int Suppl 77:S26–S30
Robertson RP (2004) Chronic oxidative stress as a central mechanism for glucose toxicity in pancreatic islet beta cells in diabetes. J Biol Chem 279:42351–42354
Choi YS, Kim S, Pak YK (2001) Mitochondrial transcription factor A (mtTFA) and diabetes. Diabetes Res Clin Pract 54(Suppl 2):S3–S9
Kanazawa A, Nishio Y, Kashiwagi A, Inagaki H, Kikkawa R, Horiike K (2002) Reduced activity of mtTFA decreases the transcription in mitochondria isolated from diabetic rat heart. Am J Physiol Endocrinol Metab 282:E778–E785
Bishop SP, Altschuld RA (1970) Increased glycolytic metabolism in cardiac hypertrophy and congestive failure. Am J Phys 218(1):153–159
Herrero P, Peterson LR, McGill JB, Matthew S, Lesniak D, Dence C, Gropler RJ (2006) Increased myocardial fatty acid metabolism in patients with type 1 diabetes mellitus. J Am Coll Cardiol 47(3):598–604
Young ME, Guthrie PH, Razeghi P, Leighton B, Abbasi S, Patil S, Youker KA, Taegtmeyer H (2002) Impaired long-chain fatty acid oxidation and contractile dysfunction in the obese Zucker rat heart. Diabetes 51(8):2587–2595
Finck BN, Lehman JJ, Leone TC, Welch MJ, Bennett MJ, Kovacs A, Han X, Gross RW, Kozak R, Lopaschuk GD, Kelly DP (2002) The cardiac phenotype induced by PPARalpha overexpression mimics that caused by diabetes mellitus. J Clin Invest 109(1):121–130
Awan MM, Saggerson ED (1993) Malonyl-CoA metabolism in cardiac myocytes and its relevance to the control of fatty acid oxidation. Biochem J 295(Pt 1):61–66
Stanley WC, Lopaschuk GD, McCormack JG (1997) Regulation of energy substrate metabolism in the diabetic heart. Cardiovasc Res 34(1):25–33
Huang B, Wu P, Popov KM, Harris RA (2003) Starvation and diabetes reduce the amount of pyruvate dehydrogenase phosphatase in rat heart and kidney. Diabetes 52(6):1371–1376
Randle PJ, Garland PB, Hales CN, Newsholme EA, Denton RM, Pogson CI (1966) Interactions of metabolism and the physiological role of insulin. Recent Prog Horm Res 22:1–48
Neely JR, Morgan HE (1974) Relationship between carbohydrate and lipid metabolism and the energy balance of heart muscle. Annu Rev Physiol 36:413–459
Sambandam N, Lopaschuk GD (2003) AMP-activated protein kinase (AMPK) control of fatty acid and glucose metabolism in the ischemic heart. Prog Lipid Res 42:238–256
Schaffer SW, Seyed-Mozaffari M, Cutcliff CR, Wilson GL (1986) Postreceptor myocardial metabolic defect in a rat model of non-insulin-dependent diabetes mellitus. Diabetes 35:593–597
Bowman RH (1966) Effects of diabetes, fatty acids, and ketone bodies on tricarboxylic acid cycle metabolism in the perfused rat heart. J Biol Chem 241:3041–3048
Kuo TH, Moore KH, Giacomelli F, Wiener J (1983) Defective oxidative metabolism of heart mitochondria from genetically diabetic mice. Diabetes 32:781–787
Pierce GN, Dhalla NS (1985) Heart mitochondrial function in chronic experimental diabetes in rats. Can J Cardiol 1:48–54
Tomita M, Mukae S, Geshi E, Umetsu K, Nakatani M, Katagirl T (1996) Mitochondrial respiratory impairment in streptozotocin-induced diabetic rat heart. Jpn Circ J 60:673–682
Boudina S, Sena S, O’Neill BT, Tathireddy P, Young ME, Abel ED (2005) Reduced mitochondrial oxidative capacity and increased mitochondrial uncoupling impair myocardial energetics in obesity. Circulation 112:2686–2695
Suarez J, Hu Y, Makino A, Fricosky E, Wang H, Dillmann WH (2008) Alterations in mitochondrial function and cytosolic calcium induced by hyperglycemia are restored by mitochondrial transcription factor A in cardiomyocytes. Am J Physiol Cell Physiol 295:C1561–C1568
Rogers TB, Gaa ST, Massey C, Dosemeci A (1990) Protein kinase C inhibits Ca21 accumulation in cardiac sarcoplasmic reticulum. J Biol Chem 265:4302–4308
Noland TA Jr, Kuo JF (1991) Protein kinase C phosphorylation of cardiac troponin I or troponin T inhibits Ca2+-stimulated actomyosin MgATPase activity. J Biol Chem 266:4974–4978
Malhotra A, Reich D, Nakouzi A, Sanghi V, Geenen DL, Buttrick PM (1997) Experimental diabetes is associated with functional activation of protein kinase C and phosphorylation of troponin I in the heart, which are prevented by angiotensin II receptor blockade. Circ Res 81:1027–1033
Liu X, Takeda N, Dhalla NS (1996) Troponin I phosphorylation in heart homogenate from diabetic rat. Biochim Biophys Acta 1316:78–84
Okumura K, Akiyama N, Hashimoto H, Ogawa K, Satake T (1988) Alteration of 1,2,-diacylglycerol content in myocardium from diabetic rats. Diabetes 37:1168–1172
Liu X, Wang J, Nobuakira T, Binaglia L, Panagia V, Dhalla NS (1999) Changes in cardiac protein kinase C activities and isozymes in streptozotocin-induced diabetes. Am. J. Physiol. 277 (Endocrinol. Metab. 40): E798–E804
Isfort M, Stevens SCW, Schaffer S, Jong CJ, Wold LE (2014) Metabolic dysfunction in diabetic cardiomyopathy. Heart Fail Rev 19(1). https://doi.org/10.1007/s10741-013-9377-8
Lopaschuk GD, Ussher JR, Folmes CDL, Jaswal JS, Stanley WC (2010) Myocardial fatty acid metabolism in health and disease. Physiol Rev 90:207–258
Mortuza R, Chakrabarti S (2014) Glucose-induced cell signalling in the pathogenesis of diabetic cardiomyopathy. Heart Fail Rev 19:75–86
Dhalla NS, Takeda N, Rodriguez-Leyva D, Elimban V (2014) Mechanisms of subcellular remodeling in heart failure due to diabetes. Heart Fail Rev 19:87–99. https://doi.org/10.1007/s10741-013-9385-8
Dhalla NS, Liu X, Panagia V, Takeda N (1998) Subcellular remodelling and heart dysfunction in chronic diabetes. Cardiovasc Res 40(2):239–247
Brownlee M (2005) The pathobiology of diabetic complications: a unifying mechanism. Nature 54:1615–1625
Doron A (2003) Cross-linking of glycated collagen in the pathogenesis of arterial and myocardial stiffening of ageing and diabetes. J Hypertens 21:3–12
Asbun J, Villareal FJ (2006) The pathogenesis of myocardial fibrosis in the setting of diabetic cardiomyopathy. J Am Coll Cardiol 47:693–700
Masoudi FA, Inzucchi SE (2007) Diabetes mellitus and heart failure: epidemiology, mechanisms, and pharmacotherapy. Am J Cardiol 99:113–132
Giacco F, Brownlee M (2010) Oxidative stress and diabetic complications. Circ Res 107:1058–1070
Scott JA, King GL (2004) Oxidative stress and antioxidant treatment in diabetes. Ann N Y Acad Sci 1031:204–213
Boudina S, Abel ED (2010) Diabetic cardiomyopathy, causes and effects. Rev Endocr Metab Disord 11:31–39
Dobrin JS, Lebeche D (2010) Diabetic cardiomyopathy: signalling defects and therapeutic approaches. Exp Rev Cardiovasc Ther 8:373–391
Candido R, Forbes JM, Thomas MC, Thallas V, Dean RG, Burns WC, Tikellis C, Ritchie RH, Twigg SM, Cooper ME, burrell LM (2003) A breaker of advanced glycation end products attenuates diabetes-induced myocardial structural changes. Circ Res 92:785–792
Young LH, Wackers FJ, Chyun DA, Davey JA, Barrett EJ, Taillefer R, Heller GV, Iskandrian AE, Wittlin SD, Filipchunk N, Ratner RE, Inzucchi SE (2009) DIAD investigators. Cardiac outcomes after screening for asymptomatic coronary artery disease in patients with type 2 diabetes: the DIAD study, a randomized controlled trial. JAMA 301:1547–1555
Cai L, Wang Y, Zhou G, Chen T, Li X, Kang YJ (2006) Attenuation by metallo-thionein of early cardiac cell death via suppression of mitochondrial oxidative stress results in a prevention of diabetic cardiomyopathy. J Am Coll Cardiol 48:1688–1697
Guo Z, Xia Z, Jiang J, McNeil JH (2007) Down-regulation of NADPH oxidase, antioxidant enzymes and inflammatory markers in the heart of streptozotocin-induced diabetic rats by N enzymes. Am J Physiol Heart Circ Physiol 292:H1728–H1736
Schleicher ED, Weigert C (2000) Role of the hexosamine biosynthetic pathway in diabetic nephropathy. Kidney Intern. 58:S-13–S-18
Mohora M, Greabu M, Muscurel C, Duta C, Totan A (2007) The sources and the targets of oxidative stress in the aetiology of diabetic complications. Rom J Biophys 17:63–84
Belke DD, Dillmann WH (2004) Altered cardiac calcium handling in diabetes. Curr Hypertens Rep 6:424–429
Woodcock EA, Matkovich SJ (2005) Cardiomyocytes structure, function and associated pathologies. Inter J Biochem Cell Biol 37:1746–1751
Roderick HL, Bootman MD (2007) Pacemaking, arrhythmias, inotropy and hypertrophy: the many possible facets of IP3 signalling in cardiac myocytes. J Physiol 581(3):883–884
Bers DM, Eisner DA, Valdivia HH (2003) Sarcopalsmic reticulum Ca2+ transport and heart failure: roles of diastolic leak and Ca2+ Circ Res. 93:487-490
Hilgemnn DW (2004) New insights into the molecular and cellular workings of the cardiac Na+/Ca2+ exchanger. Am J Physiol Cell Physiol 287(5):C1167–C1172
Mackrill JJ (2010) Ryanodine receptor calcium channels and their partners as drug targets. Biochem Pharmacol 79:1535–1543
Kushnir AA, Betzenhauser MJ, Marks AR (2010) Ryanodine receptor studies using genetically engineered mice. FEBS Lett 584:1956–1965
Hasenfuss G, Schillinger W (2004) Is modulation of sodium-calcium exchange a therapeutic option in heart failure? Circ Res 95:225–227
Franzini-Armstrong C (1970) Studies of the triad. J Cell Biol 47:488–499
Campbell K, Franzini-Armstrong C, Shamoo A (1980) Further characterisation of light and heavy sarcoplasmic reticulum vesicles. Identification of the ‘sarcoplasmic reticulumfeet’ associated with heavy sarcoplasmic reticulum-vesicles. Biochem Biophys Acta 602:97–116
Lai FA, Anderson K, Rousseau E, Liu QY, Meissner G (1988) Evidence for a Ca2+ channel within the ryanodine receptor complex from cardiac sarcoplasmic reticulum. Biochem Biophys Res Commun 151:441–449
Takeshima H, Nishimura S, Matsumoto T, Ishida H, Kangawa K, Minamino N, Matsuo H, Ueda M, Hanaoka M, Hirose T (1989) Primary structure and expression from complementary DNA of skeletal muscle ryanodine receptor. Nature 339:439–445
Zorzato F, Fujii J, Otsu K, Phillips M, Green NM, Lai FA, Meissner G, Maclennan DH (1990) Molecular cloning of cDNA encoding human and rabbit forms of the Ca2+ release channel (ryanodine receptor) of skeletal muscle sarcoplasmic reticulum. J Biol Chem 265:2244–2256
Tunwell RE, Wickenden C, Bertrand BM, Shevchenko VI, Walsh MB, Allen PD, Lai FA (1996) The human cardiac muscle ryanodine receptor-calcium release channel: identification, primary structure and topological analysis. Biochem J 318:477–487
Hakamata Y, Nakai J, Takeshima H, Imoto K (1992) Primary structure and distribution of a novel ryanodine receptor/calcium release channel from rabbit brain. FEBS Lett 312:229–235
Hillyard I, Procia L (1956) Action of ryanodine on isolated kitten auricle. Fed Proc 15:438–449
Meissner G (1986) Ryanodine activation and inhibition of the Ca2+ release channel of sarcoplasmic reticulum. J Biol Chem 261:6300–6306
Bers DM, Eisner DA, Valdivia HH (2003) Sarcoplasmic reticulum Ca2+ and heart failure: roles of diastolic leak and Ca2+ transport. Circ Res 93:487–490
Priori SG, Napolitano C, Tiso N, Memmi M, Vignati G, Bloise R, Sorrentino V, Danieli GA (2001) Mutations in the cardiac ryanodine receptor gene (hRyR2) underlie catecholaminergic polymorphic ventricular tachycardia. Circulation 103:196–200
George CH, Jundi H, Thomas NL, Fry DL, Lai FA (2007) Ryanodine receptors and ventricular arrhythmias: emerging trends in mutations, mechanisms and therapies. J Mol Cell Cardiol 42:34–50
Ganguly PK, Pierce GN, Dhalla KS, Dhalla NS (1983) Defective sarcoplasmic reticular calcium transport in diabetic cardiomyopathy. Am J Phys 244:E528–E535
Pierce GN, Ramjiawan B, Dhalla NS, Ferrari R (1990) Na+-H+ exchange in cardiac sarcolemmal vesicles isolated from diabetic rats. Am J Phys 258:H255–H261
Makino N, Dhalla KS, ElimbanV DNS (1987) Sarcolemmal Ca2+ transport in streptozotocin-induced diabetic cardiomyopathy in rats. Am J Phys 253:E202–E207
Lun SL, Ostadalova I, Kolar DNS (1992) Alterations in Ca2+-channels during the development of diabetic cardiomyopathy. Mol Cell Biochem 109(2):173–179
Dhalla NS, Rangi S, Zieroth S, Xu YJ (2012) Alterations in sarcoplasmic reticulum and mitochondrial functions in diabetic cardiomyopathy. Exp Clin Cardiol 17(3):115–120
Okunade GW, Miller ML, Pyne GJ, Sutliff RL, O’Connor KT, Neumann JC, Andringa A, Miller DA, Prasad V, Doetschman T, Paul RJ, Shull GE (2004) Targeted ablation of plasma membrane Ca2+−ATPase (PMCA) 1 and 4 indicates a major housekeeping function for PMCA1 and a critical role in hyperactivated sperm motility and male fertility for PMCA4. J Biol Chem 279:33742–33750
Orio P, Rojas P, Ferreira G, Latorre R (2002) New disguises for an old channel: MaxiK channel subunits. News Physiol Sci 17:156–161
Liu N, Colombi B, Memmi M, Zissimopoulos S, Rizzi N, Imbriani M, Napolitano C, Lai FA, Priori SG (2006) Arrhythmogenesis in catecholaminergic polymorphic ventricular tachycardia: insights from a RyR2 R4496C knock-in mouse model. Circ Res 99:292–298
Periasamy M, Huke S (2001) SERCA pump level is a critical determinant of Ca2+ homeostasis and cardiac contractility. J Mol Cell Cardiol 33:1053–1063
Searls YM, Loganathan R, Smirnova IV, Stehno-Bittel L (2010) Intracellular Ca2+ regulating proteins in vascular smooth muscle cells are altered with type 1 diabetes due to the direct effects of hyperglycemia. Cardiovasc Diabetol 9(8). https://doi.org/10.1186/1475/-2840-9-8
Abe T, Ohga Y, Tabayashi N, Kobayashi S (2002) Left ventricular diastolic dysfunction in type II diabetes mellitus model rats. Am J Heart Circ Physiol 282:H139–H148
Wold LE, Dutta K, Mason MM, Ren J, Cala SE, Schwanke ML, Davidoff AJ (2005) Impaired SERCA function contributes to cardiomyocyte dysfunction in insulin resistant rats. J Mol Cell Cardiol 39:297–307
Zarain-Herzberg A, Yano K, Elimban V, Dhalla NS (1994) Cardiac sarcoplasmatic reticulum Ca2+−ATPase expression in streptozotocin-induced diabetic rat. Biochem Biophys Res Commun 203:113–120
Zhong Y, Ahmed S, Grupp IL, Matlib MA (2001) Altered SR protein expression associated with contractile dysfunction in diabetic rat heart. Am J Physiol Heart Circ Physiol 281:H1137–H1147
Aoyagi T, Yonekura K, Eto Y, Matsumoto A, Yokoyama I, Sugiura S, Momomura S, Hirata Y, Baker DL, Periasamy M (1999) The sarcoplasmic reticulum Ca2+ ATPase gene promoter activity is decreased in response to severe left ventricular pressure-overload in hypertrophy rats. J Moll Cell Cardiol 31:919–916
Connelly KA, Gilbert RE, Krum H (2007) Letter by Connelly et al regarding article, “diastolic stiffness of the failing diabetic heart: importance of fibrosis, advanced glycation end products, and myocyte resting tension”. Circ. 117 (23) e483; author reply e484
Fredersdorf S, Thumann C, Zimmerman WH, Vetter R, Graf T, Luchner A, Riegger GA, Schunkert H, Eschenhagen T, Weil J (2012) Increased myocardial SERCA expression in early type 2 diabetes mellitus is insulin dependent: in vivo and in vitro data. Cardiovasc Diabetol 11(57):1–11
Lacombe K, Massari V, Girard P, Serfaty L, Gozlan J, Pialoux G, Mialhes P, Molina JM, Lascoux-Combe C, Wendum D, Carrat F, Zoulim F (2006) Major role of hepatitis B genotypes in liver fibrosis during coinfection with HIV. AIDS 20:419–427
Dhalla NS, Temsah RM, Netticadan T (2000) Role of oxidative stress in cardiovascular diseases. J Hypertens 18:655–673
Bracken NK, Singh J, Winlow W, Howarth FC (2003) Mechanism underlying contractile dysfunction in streptozotocin-induced type 1 and type 2 diabetic cardiomyopathy. In: Pierce GN, Nagano M, Zahradka P, Dhalla NS (eds) Atherosclerosis, Hypertension and diabetes. Progress in experimental cardiology, vol 8. Springer, Boston, MA, pp 387–408
Howarth FC, Singh J (1999) Altered handling of calcium during the process of excitation-contraction coupling in the streptozotocin-induced diabetic heart: a short review. Int J Diabetes 7:52–64
Pandit SV, Giles WR, Demir SS (2003) A mathematic model of the electrophysiological alterations in rat ventricular myocyte in type 1 diabetes mellitus. Biophys J 84:832–841
Howarth FC, Qureshi MA, Bracken NK, Winlow W, Singh J (2001) Time dependant effects of streptozotocin-induced diabetes on contraction in rat ventricular myocytes. Emirates J 19:25–41
Singh J, Chonkar A, Bracken N, Adeghate E, Latt Z, Hussain M (2006) Effect of streptozotocin-induced type 1 diabetes mellitus on contraction, calcium transient, and cation contents in the isolated rat heart. Ann N Y Acad Sci 1084:178–190
Sheikh AQ, Hurley JR, Huang W, Taghian T, Kogan A, Hongkwan C, Wang Y (2012) Diabetes alters intracellular calcium transients in cardiac endothelial cells. PLoS One 7(5):e36840
Belke DD, Swanson EA, Dillmann WH (2004) Decreased sarcoplasmic reticulum activity and contractility in diabetic db/db mouse heart. Diabetes 53:3201–3208
Salem KA, Qureshi MA, Sydorenko V, Iqbal T, Singh J, Howarth C (2013) Effects of exercise training on excitation-contraction coupling and related mRNA expression in hearts of Goto-Kakizaki type 2 diabetic rats. Mol Cell Biochem 380(1–2):83–96
Zhang H, Chen X, Gao E, MacDonnell SM, Wang W, Kolpakov M, Nakayama H, Zhang X, Jaleel N, Harris DM, Li Y, Tang M, Berretta R, Leri A, Kajstura J, Sabri A, Koch WJ, Molkentin JD, Houser SR (2010) Increasing cardiac contractility after myocardial infarction exacerbates cardiac injury and pump dysfunction. Circ Res 107:800–809. https://doi.org/10.1161/CIRCRESAHA.110.219220
Nemoto O, Kawaguchi M, Yaoita H, Miyake K, Maehara K, Maruyama Y (2006) Left ventricular dysfunction and remodeling in streptozotocin-induced diabetic rats. Circ J 70:327–334
D'Souza A, Howarth FC, Yanni J, Dobryznski H, Yanni J, Dobryznski H, Boyett MR, Adeghate E, Bidasee KR, Singh J (2011) Left ventricle structural remodelling in the prediabetic Goto-Kakizaki rat. Exp Physiol 96(9):b875–b888
Kitzman DW, Higginbotham MB, Cobb FR, Sheikh Sullivan MJ (1991) Exercise intolerance in patients with heart failure and preserved left ventricular systolic function: failure of the Frank-Starling mechanism. J Am Coll Cardiol 17:1065–1072
Gandhi SK, Powers JC, Nomeir AM, Fowle K, Kitzman DW, Rankin KM, Little WC (2001) The pathogenesis of acute pulmonary oedema associated with hypertension. N Engl J Med 344:17–22
Vasan RS, Larson MG, Benjamin EJ, Evans JC, Reiss CK, Levy D (1999) Congestive heart failure in subjects with normal versus reduced left ventricular ejection fraction: prevalence and mortality in a population based cohort. J Am Coll Cardiol 33:1948–1955
Pieske B, Wachter R (2008) Impact of diabetes and hypertension on the heart. Curr Opin Cardiol 23:340–349
Ren X, Ristow B, Na B, Ali S, Schiller NB, Whooley MA (2007) Prevalence and prognosis of asymptomatic left ventricular diastolic dysfunction in ambulatory patients with coronary heart disease. Am J Cardiol 99:1643–1647
Iribarren C (2001) Glycemic control and heart failure among adult patients with diabetes. Circ. 103:2668–2673
Bertoni AG, Hundley WG, Massing MW, Bonds DE, Burke GL, Goff DC Jr (2004) Heart failure prevalence, Incidence, and Mortality in the Elderly With Diabetes. Diabetes Care 27:344–348
Burkhoff D (2012) Mortality in heart failure with preserved ejection fraction: an unacceptably high rate. Eur Heart J 33:1718–1720
MacDonald MR, Petrie MC, Varyani F, Ostergren J, Michelson EL, Young JB, Solomon SD, Granger CB, Swedberg K, Yusuf S, Pfeffer MA, McMurray JJ (2008) Impact of diabetes on outcomes in patients with low and preserved ejection fraction heart failure. Eur Heart J 29:1377–1385
Poirier P, Bogaty P, Garneau C, Marois L, Dumesnil JG (2001) Diastolic dysfunction in normotensive men with well-controlled type 2 diabetes: importance of manoeuvers in echocardiographic screening for preclinical diabetic cardiomyopathy. Diabetes Care 24:1–5
Andersen NH, Poulsen SH, Helleberg K, Ivarsen P, Knudsen ST, Mogensen CE (2003) Impact of essential hypertension and diabetes mellitus on left ventricular systolic and diastolic performance. Eur J Echocardiogr 4:306–312
Magri CJ, Cassar A, Fava S, Felice H (2012) Heart failure with preserved ejection fraction and diabetes mellitus. J Diabetes Res Clin Metab. https://doi.org/10.7243/2050-0866-1-2
Ehl NF, Kuhne M, Brinkert M, Muller-Brand J, Zellweger MJ (2011) Diabetes reduces left ventricular ejection fraction-irrespective of presence and extent of coronary artery disease. Eur J Endocrinol 165:945–951
Wong CY, O’Moore-Sullivan T, Leano R, Byrne BE, Marwick TH (2004) Alterations of left ventricular myocardial characteristics associated with obesity. Circ. 110:3081–3087
Heerebeek VL, Hamdani N, Handoko ML, Falcao-Pires I, Musters RJ, Kupreishvili K, Ijsselmuiden AJ, Schalkwijk CG, Bronzwaer JG, Diamant M, Borbély A, van der Velden J, Stienen GJ, Laarman GJ, Niessen HW, Paulus WJ (2008) Diastolic stiffness of the failing diabetic heart. Importance of fibrosis, advanced glycation end products, and myocyte resting tension. Circ. 117:43–51
Borlaug BA, Lam CSP, Roger VL, Rodeheffer RJ, Redfield MM (2009) Contractility and ventricular systolic stiffening in hypertensive heart disease. J Am Coll Cardiol 54:410–418
Maeder MT, Kaye DM (2009) Heart failure with normal left ventricular ejection fraction. J Am Coll Cardiol 53:11–16
Periasamy M, Janssen ML (2008) Molecular basis of diastolic dysfunction. Heart Fail Clin 4(1):13–21
Aurigemma GP, Gaasch WH (2004) Clinical practice. Diastolic heart failure N Engl J Med 351:1097–1105
Pierce GN, Dhalla NS (1981) Cardiac myofibrillar ATPase activity in diabetic rats. J Mol Cell Cardiol 13:1063–1069
Pierce GN, Dhalla NS (1985) Mechanisms of the defect in cardiac myofibrillar function during diabetes. Am J Phys 248:E170–E175
Jweied EE, McKinney RD, Walker LA, Brodsky I, Geha AS, Massad MG, Buttrick PM, de Tombe PP (2005) Depressed cardiac myofilament function in human diabetes mellitus. Am J Physiol Heart Circ Physiol 289:H2478–H2483
Kodama S, Tanaka S, Heianza Y, Fujihara K, Horikawa C, Shimano H, Saito K, Yamada N, Ohashi Y, Sone H (2013) Association between physical activity and risk of all-cause mortality and cardiovascular disease in patients with diabetes: a meta-analysis. Diabetes Care 36:471–479
Stolen TO, Hoydal MA, Kemi OJ, Catalucci D, Ceci M, Aasum E, Larsen T, Rolim N, Condorelli G, Smith GL, Wisløff U (2009) Interval training normalizes cardiomyocyte function, diastolic Ca2+ control, and SR Ca2+ release synchronicity in a mouse model of diabetic cardiomyopathy. Circ Res 105:527–536
Epp RA, Susser SE, Morissette MP, Kehler DS, Jassal DS, Duhamel TA (2013) Exercise training prevents the development of cardiac dysfunction in the low-dose streptozotocin diabetic rats fed a high-fat diet. Can J Physiol Pharmacol 91:80–89
DeMarco VG, Aroor AR, Sowers JR (2014) The pathophysiology of hypertension in patients with obesity. Nat Rev Endocrinol 10:364–376
Voulgari C, Papadogiannis D, Tentolouris N (2010) Diabetic cardiomyopathy: from the pathophysiology of the cardiac myocytes to current diagnosis and management strategies. Vasc Health Risk Manag 6:883–903
Adler AI, Neil HA, Manley SE, Holman RR, Turner RC (1999) Hyperglycemia and hyperinsulinemia at diagnosis of diabetes and their association with subsequent cardiovascular disease in the United Kingdom prospective diabetes study (UKPDS 47). Am Heart J 138:S353–S359
Chung J, Abraszewski P, Yu X et al (2006) Paradoxical increase in ventricular torsion and systolic torsion rate in type I diabetic patients under tight glycemic control. J Am Coll Cardiol 47:384–390
Sharma AK, Srinivasan BP (2009) Triple verses glimepiride plus metformin therapy on cardiovascular risk biomarkers and diabetic cardiomyopathy in insulin resistance type 2 diabetes mellitus rats. Eur J Pharm Sci 38:433–444
UK Prospective Diabetes Study (UKPDS) Group (1998) Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352:837–853
Wong AK, Symon R, AlZadjali MA, Ang DS, Ogston S, Choy A, Petrie JR, Struthers AD, Lang CC (2012) The effect of metformin on insulin resistance and exercise parameters in patients with heart failure. Eur J Heart Fail 14:1303–1310
von Bibra H, St John Sutton M (2011) Impact of diabetes on postinfarction heart failure and left ventricular remodeling. Curr Heart Fail Rep 8:242–251
Mamas MA, Deaton C, Rutter MK, Yuille M, Willams SG, Ray SG, Gibson JM, Neyses L (2010) Impaired glucose tolerance and insulin resistance in heart failure: under recognized and undertreated? J Card Fail 16:761–768
Young LH (2003) Insulin resistance and the effects of thiazolidinediones on cardiac metabolism. Am J Med 115:75S–80S
Inzucchi SE, Zinman B, Wanner C, Ferrari R, Fitchett D, Hantel S, Espadero RM, Woerle HL, Broedi UC, Johnasen OE (2015) SGLT-2 inhibitors and cardiovascular risk: proposed pathways and review of ongoing outcome trials. Diab Vasc Dis Res 12:90–100
Doehner W, Frenneaux M, Anker SD (2014) Metabolic impairment in heart failure: the myocardial and systemic perspective. J Am Coll Cardiol 64:1388–1400
Bostick B, Habibi J, Ma L, Aroor A, Rehmer N, Hayden MR, Sowers JR (2014) Dipeptidyl peptidase inhibition prevents diastolic dysfunction and reduces myocardial fibrosis in a mouse model of western diet induced obesity. Metabolism 63:1000–1011
Chen YH, Feng B, Chen ZW (2012) Statins for primary prevention of cardiovascular and cerebrovascular events in diabetic patients without established cardiovascular diseases: a meta-analysis. Exp Clin Endocrinol Diabetes 120:116–120
Cholesterol Treatment Trialists’ (CTT) Collaborators, Kearney PM, Blackwell L, Collins R, Keech A, Simes J, Peto R, Armitage J, Baigent C (2008) Efficacy of cholesterol-lowering therapy in 18,686 people with diabetes in 14 randomised trials of statins: a meta-analysis. Lancet 371:117–125
Van Linthout S, Riad A, Dhayat N, Spillman F, Du J, Westermann D, Hilfiker-Kleiner D, Noutsias M, Laufs U, Schultheiss HO, Tschope C (2007) Anti-inflammatory effects of atorvastatin improve left ventricular function in experimental diabetic cardiomyopathy. Diabetologia 50:1977–1986
Dai QM, Lu J, Liu NF (2011) Fluvastatin attenuates myocardial interstitial fibrosis and cardiac dysfunction in diabetic rats by inhibiting over-expression of connective tissue growth factor. Chin Med J 124:89–94
Lowes BD, Gill EA, Abraham WT, Larrain JR, Robertson AD, Bristow MR, Gilbert EM (1999) Effects of carvedilol on left ventricular function mass, chamber geometry, and mitral regurgitation in chronic heart failure. Am J Cardiol 83:1201–1205
Waagstein F, Bristow M, Swedburg K (1993) Beneficial effects of metoprolol in idiopathic dilated cardiomyopathy. Lancet 342:1441–1446
The Cardiac Insufficiency Bisoprolol Study (CIBIS) Investigators and Committees (1994) A randomised trial of β-blockade in heart failure: the Cardiac Insufficiency Bisoprolol Study (CIBIS). Circ. 90:1765–1773
CIBIS II Investigators and Committees (1999) The Cardiac Insufficiency Bisoprolol Study (CIBIS II): a randomised trial. Lancet 353:9–13
Merit-HF Study Group (1999) Effect of metoprolol CR/XL in chronic heart failure: metoprolol CR/XL randomised intervention trial in congestive heart failure (MERIT-HF). Lancet 353:2001–2007
Packer M, Bristow M, Cohn J (1996) The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. N Engl J Med 334:1349–1355
Bristow MR (2001) Beta-adrenergic receptor blockade in chronic heart failure. Circulation 101:558–569
Sharma V, McNeill JH (2011) Parallel effects of β-adrenoceptor blockade on cardiac function and fatty acid oxidation in the diabetic heart: confronting the maze. World J Cardiol 3:281–302
Mancia G, Fagard R, Narkiewicz K et al (2013) 2013 ESH/ESC guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 28:2159–2219
Thomas CM, Yong QC, Seqqat R, Chandel N, Feldman DL, Baker KM, Kumar R (2013) Direct renin inhibition prevents cardiac dysfunction in a diabetic mouse model: comparison with an angiotensin receptor antagonist and angiotensin-converting enzyme inhibitor. Clin Sci (Lond) 124:529–541
Machackova J, Liu X, Lukas A, Dhalla NS (2004) Renin-angiotensin blockade attenuates cardiac myofibrillar remodelling in chronic diabetes. Mol Cell Biochem 261:271–278
Symeonides P, Koulouris S, Vratsista E, Triantafyllou K, Ioannidis G, Thalassinos N, Katritsis D (2007) Both ramipril and telmisartan reverse indices of early diabetic cardiomyopathy: a comparative study. Eur J Echocardiogr 8:480–486
Szabo C (2002) PARP as a drug target for the therapy of diabetic cardiovascular dysfunction. Drug News Perspect 15:197–205
Du X, Matsumura T, Edelstein D, Rossetti L, Zsengeller Z, Szabo C, Brownlee M (2003) Inhibition of GAPDH activity by poly (ADP-ribose) polymerase activates three major pathways of hyperglycemic damage in endothelial cells. J Clin Invest 112:1049–1057
Zhao P, Zhang J, Yin XG, Maharaj P, Narraindoo S, Cui LQ, Tang YS (2013) The effect of trimetazidine on cardiac function in diabetic patients with idiopathic dilated cardiomyopathy. Life Sci 92:633–638
Li YJ, Wang PH, Chen C, Zou MH, Wang DW (2010) Improvement of mechanical heart function by trimetazidine in db/db mice. Acta Pharmacol Sin 31:560–569
Sulaiman M, Matta MJ, Sunderesan NR, Gupta MP, Periasamy M, Gupta M (2010) Resveratrol, an activator of SIRT1, upregulates sarcoplasmic calcium ATPase and improves cardiac function in diabetic cardiomyopathy. Am J Physiol Heart Circ Physiol 298:H833–H843
Maier LS, Layug B, Karwatowska-Prokopczuk E, Belardinelli L, Sander J, Lang C, Wachter R, Edelmann F, Hasenfuss G, Jacobshagen C (2013) RAnoLazIne for the treatment of diastolic heart failure in patients with preserved ejection fraction: the RALI-DHF proof-of-concept study. JACC Heart Fail 1:115–122
Huynh K, Bernardo BC, McMullen JR, Ritchie RH (2014) Diabetic cardiomyopathy: mechanisms and new treatment strategies targeting antioxidant signaling pathways. Pharmacol Ther 142:375–415
Leon LE, Rani S, Fernandez M, Larico M, Calligaris SD (2016) Subclinical detection of diabetic cardiomyopathy withMicroRNAs: challenges and perspectives. J Diabetes Res. https://doi.org/10.1155/2016/6143129
Yildirim SS, Akman D, Catalucci D, Turan B (2013) Relationship between downregulation of miRNAs and increase of oxidative stress in the development of diabetic cardiac dysfunction: junctin as a target protein of miR-1. Cell Biochem Biophys 67:1397–1408
Thum T, Gross C, Fiedler J, Fischer T, Kissler S, Bussen M, Galuppo P, Just S, Rottbauer W, Frantz S, Castoldi M, Soutschek J, Koteliansky V, Rosenwald A, Basson MA, Licht JD, Pena JT, Rouhanifard SH, Muckenthaler MU, Tuschl T, Martin GR, Bauersachs J, Engelhardt S (2008) MicroRNA-21 contributes to myocardial disease by stimulating MAP kinase signalling in fibroblasts. Nature 456(7224):980–984. https://doi.org/10.1038/nature07511
Tan Y, Ichikawa T, Li J, Si Q, Yang H, Chen X, Goldblatt CS, Meyer CJ, Li X, Cai L, Cui T (2011) Diabetic downregulation of Nrf2 activity via ERK contributes to oxidative stress-induced insulin resistance in cardiac cells in vitro and in vivo. Diabetes 60(2):625–633. https://doi.org/10.2337/db10-1164
Mortensen SA, Rosenfeldt F, Kumar A et al (2014) The effect of coenzyme Q10 on morbidity and mortality in chronic heart failure: results from Q-SYMBIO: a randomized double-blind trial. JACC Heart Fail 2:641–649
Xu YJ, Tappia PS, Neki NS, Dhalla NS (2014) Prevention of diabetes-induced cardiovascular complications upon treatment with antioxidants. Heart Fail Rev 19:113–121
Huynh K, Kiriazis H, Du XJ, Love JE, Jandeleit-Dahm KA, Forbes JM, McMullen JR, Ritchie RH (2012) Coenzyme Q10 attenuates diastolic dysfunction, cardiomyocyte hypertrophy and cardiac fibrosis in the db/db mouse model of type 2 diabetes. Diabetologia 55:1544–1553
Katare R, Caporali A, Zentilin L, Avolio E, Sala-Newby G, Oikawa A, Cesslli D, Beltram AP, Giacca M, Emanuel C, Madeddu P (2011) Intravenous gene therapy with PIM-1 via a cardiotropic viral vector halts the progression of diabetic cardiomyopathy through promotion of prosurvival signaling. Circ Res 108:1238–1251
Meloni M, Descamps B, Caporali A, Zentilin L, Floris I, Giacca M, Emanueli C (2012) Nerve growth factor gene therapy using adeno-associated viral vectors prevents cardiomyopathy in type 1 diabetic mice. Diabetes 61:229–240
Prakoso D, De Blasio MJ, Qin C, Rosli S, Kiriazis H, Qian H, Du XJ, Weeks KL, Gregorevic P, McMullen JR, Ritchie RH (2017) Phosphoinositide 3-kinase (p110α) gene delivery limits diabetes-induced cardiac NADPH oxidase and cardiomyopathy in a mouse model with established diastolic dysfunction. Clin Sci (Lond). 131(12):1345–1360. https://doi.org/10.1042/CS20170063
Szeto HH (2006) Cell-permeable, mitochondrial-targeted, peptide antioxidants. AAPS J 8:E277–E283
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
None.
Rights and permissions
About this article
Cite this article
Singh, R.M., Waqar, T., Howarth, F.C. et al. Hyperglycemia-induced cardiac contractile dysfunction in the diabetic heart. Heart Fail Rev 23, 37–54 (2018). https://doi.org/10.1007/s10741-017-9663-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-017-9663-y