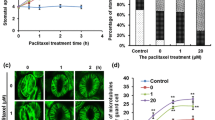
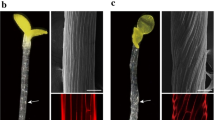
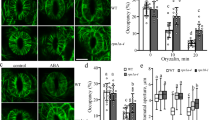
Abstract
Microtubules are highly dynamic cellular structures that are required for many biological processes. Cortical microtubules in plant play crucial roles during cell expansion. Its proper dynamics are required for plant growth and responses to environmental stimuli. Arabidopsis mutants, such as sav2/tub4 P287L, display a variety of growth defects, including short and twisting hypocotyls in dark and shade. Both microtubule organization and dynamics are altered in sav2. Here, we have identified a suppressor of sav2 (sus2), which surprisingly contains a missense mutation in another β tubulin gene, TUB6. The mutation results in a L246F substitution in TUB6. It locates at the interface of αβ-intradimer. This mutation partially suppressed the swirling microtubule arrangement in sav2 hypocotyl cells, leading to the partial rescue of sav2 phenotypes. As the mutant behaves as a semi-dominant mutation and the CFP-labeled tub6L246F can incorporate into microtubules, we propose that the incorporation of tub6L246F interferes with the normal function of microtubules. tub6 L246F single mutant is hypersensitive to drugs disrupting microtubule dynamics, such as colchicine, suggesting the mutation may affect microtubule dynamics. Moreover, we found the colchicine hypersensitivity of tub6L246F can be suppressed by tub4P287L, while tub6L246F interferes with the rescuing effect of EB1 on sav2. As P287 locates around M-loop, which is involved in interactions between microtubule protofilaments, we propose that altered interactions at αβ-intradimer interface may affect microtubule dynamics through M-loop mediated interactions between microtubule protofilaments.





Similar content being viewed by others
References
Brooks BR, Brooks CL, Mackerell AD, Nilsson L, Petrella RJ, Roux B, Won Y, Archontis G, Bartels C, Boresch S, Caflisch A, Caves L, Cui Q, Dinner AR, Feig M, Fischer S, Gao J, Hodoscek M, Im W, Kuczera K, Lazaridis T, Ma J, Ovchinnikov V, Paci E, Pastor RW, Post CB, Pu JZ, Schaefer M, Tidor B, Venable RM, Woodcock HL, Wu X, Yang W, York DM, Karplus M (2009) CHARMM: the biomolecular simulation program. J Comput Chem 30(10):1545–1614. doi:10.1002/jcc.21287
Chaffey N, Barlow P (2002) Myosin, microtubules, and microfilaments: co-operation between cytoskeletal components during cambial cell division and secondary vascular differentiation in trees. Planta 214(4):526–536. doi:10.1007/s004250100652
Chan J, Calder GM, Doonan JH, Lloyd CW (2003) EB1 reveals mobile microtubule nucleation sites in Arabidopsis. Nat Cell Biol 5(11):967–971. doi:10.1038/ncb1057
Chan J, Eder M, Crowell EF, Hampson J, Calder G, Lloyd C (2011) Microtubules and CESA tracks at the inner epidermal wall align independently of those on the outer wall of light-grown Arabidopsis hypocotyls. J Cell Sci 124(7):1088–1094. doi:10.1242/jcs.086702
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16(6):735–743. doi:10.1046/j.1365-313x.1998.00343.x
Crowell EF, Timpano H, Desprez T, Franssen-Verheijen T, Emons AM, Hofte H, Vernhettes S (2011) Differential regulation of cellulose orientation at the inner and outer face of epidermal cells in the Arabidopsis hypocotyl. Plant Cell 23(7):2592–2605. doi:10.1105/tpc.111.087338
Hamant O, Heisler MG, Jonsson H, Krupinski P, Uyttewaal M, Bokov P, Corson F, Sahlin P, Boudaoud A, Meyerowitz EM, Couder Y, Traas J (2008) Developmental patterning by mechanical signals in Arabidopsis. Science 322(5908):1650–1655. doi:10.1126/science.1165594
Hashimoto T (2013) Dissecting the cellular functions of plant microtubules using mutant tubulins. Cytoskeleton 70(4):191–200. doi:10.1002/cm.21099
Hashimoto T (2015) Microtubules in plants. Arabidopsis Book 13:e0179. doi:10.1199/tab.0179
Hastie SB (1991) Interactions of colchicine with tubulin. Pharmacol Ther 51(3):377–401. doi:10.1016/0163-7258(91)90067-V
Howard J, Hyman AA (2009) Growth, fluctuation and switching at microtubule plus ends. Nat Rev Mol Cell Biol 10(8):569–574. doi:10.1038/nrm2713
Ishida T, Kaneko Y, Iwano M, Hashimoto T (2007) Helical microtubule arrays in a collection of twisting tubulin mutants of Arabidopsis thaliana. Proc Natl Acad Sci USA 104(20):8544–8549. doi:10.1073/pnas.0701224104
Jordan MA, Wilson L (2004) Microtubules as a target for anticancer drugs. Nat Rev Cancer 4(4):253–265. doi:10.1038/nr1317
Kopczak SD, Haas NA, Hussey PJ, Silflow CD, Snustad DP (1992) The small genome of Arabidopsis contains at least six expressed α-tubulin genes. Plant Cell 4(5):539–547. doi:10.1105/Tpc.4.5.539
Krissinel E, Henrick K (2007) Inference of macromolecular assemblies from crystalline state. J Mol Biol 372(3):774–797. doi:10.1016/j.jmb.2007.05.022
Lloyd C (2011) Dynamic microtubules and the texture of plant cell walls. Int Rev Cell Mol Biol 287(287):287–329. doi:10.1016/B978-0-12-386043-9.00007-4
Lowe J, Li H, Downing KH, Nogales E (2001) Refined structure of αβ-tubulin at 3.5 Å resolution. J Mol Biol 313(5):1045–1057. doi:10.1006/jmbi.2001.5077
Mathur J, Hulskamp M (2002) Microtubules and microfilaments in cell morphogenesis in higher plants. Curr Biol 12(19):R669–R676. doi:10.1016/S0960-9822(02)01164-8
Nick P, Bergfeld R, Schafer E, Schopfer P (1990) Unilateral reorientation of microtubules at the outer epidermal wall during phototropic and gravitropic curvature of maize coleoptiles and sunflower hypocotyls. Planta 181(2):162–168. doi:10.1007/Bf02411533
Nogales E, Wolf SG, Downing KH (1998) Structure of the αβ tubulin dimer by electron crystallography. Nature 391(6663):199–203. doi:10.1038/34465
Nogales E, Whittaker M, Milligan RA, Downing KH (1999) High-resolution model of the microtubule. Cell 96(1):79–88. doi:10.1016/S0092-8674(00)80961-7
Phillips JC, Braun R, Wang W, Gumbart J, Tajkhorshid E, Villa E, Chipot C, Skeel RD, Kale L, Schulten K (2005) Scalable molecular dynamics with NAMD. J Comput Chem 26(16):1781–1802. doi:10.1002/jcc.20289
Ravelli RBG, Gigant B, Curmi PA, Jourdain I, Lachkar S, Sobel A, Knossow M (2004) Insight into tubulin regulation from a complex with colchicine and a stathmin-like domain. Nature 428(6979):198–202. doi:10.1038/nature02393
Sambade A, Pratap A, Buschmann H, Morris RJ, Lloyd C (2012) The influence of light on microtubule dynamics and alignment in the Arabidopsis hypocotyl. Plant Cell 24(1):192–201. doi:10.1105/tpc.111.093849
Sandblad L, Busch KE, Tittmann P, Gross H, Brunner D, Hoenger A (2006) The Schizosaccharomyces pombe EB1 homolog Mal3p binds and stabilizes the microtubule lattice seam. Cell 127(7):1415–1424. doi:10.1016/j.cell.2006.11.025
Schymkowitz J, Borg J, Stricher F, Nys R, Rousseau F, Serrano L (2005) The FoldX web server: an online force field. Nucleic Acids Res 33:W382–W388. doi:10.1093/nar/gki387
Sedbrook JC, Kaloriti D (2008) Microtubules, MAPs and plant directional cell expansion. Trends Plant Sci 13(6):303–310. doi:10.1016/j.tplants.2008.04.002
Shearwin KE, Timasheff SN (1994) Effect of colchicine analogs on the dissociation of αβ-tubulin into subunits-the locus of colchicine binding. Biochemistry 33(4):894–901. doi:10.1021/Bi00170a007
Shibaoka H (1994) Plant hormone-induced changes in the orientation of cortical microtubules-alterations in the cross-linking between microtubules and the plasma-membrane. Annu Rev Plant Physiol Plant Mol Biol 45:527–544. doi:10.1146/annurev.pp.45.060194.002523
Skoufias DA, Wilson L (1992) Mechanism of inhibition of microtubule polymerization by colchicine-inhibitory potencies of unliganded colchicine and tubulin colchicine complexes. Biochemistry 31(3):738–746. doi:10.1021/Bi00118a015
Snustad DP, Haas NA, Kopczak SD, Silflow CD (1992) The small genome of Arabidopsis contains at least nine expressed β-tubulin genes. Plant Cell 4(5):549–556. doi:10.1105/Tpc.4.5.549
Thitamadee S, Tuchihara K, Hashimoto T (2002) Microtubule basis for left-handed helical growth in Arabidopsis. Nature 417(6885):193–196. doi:10.1038/417193a
Ueda K, Matsuyama T, Hashimoto T (1999) Visualization of microtubules in living cells of transgenic Arabidopsis thaliana. Protoplasma 206(1–3):201–206. doi:10.1007/Bf01279267
Vaughan KT (2005) TIP maker and TIP marker; EB1 as a master controller of microtubule plus ends. J Cell Biol 171(2):197–200. doi:10.1083/jcb.200509150
Vineyard L, Elliott A, Dhingra S, Lucas JR, Shaw SL (2013) Progressive transverse microtubule array organization in hormone-induced Arabidopsis hypocotyl cells. Plant Cell 25(2):662–676. doi:10.1105/tpc.112.107326
Xiong X, Xu DY, Yang ZN, Huang H, Cui XF (2013) A single amino-acid substitution at Lysine 40 of an Arabidopsis thaliana α-tubulin causes extensive cell proliferation and expansion defects. J Integr Plant Biol 55(3):209–220. doi:10.1111/jipb.12003
Yu J, Qiu H, Liu X, Wang ML, Gao YL, Chory J, Tao Y (2015) Characterization of tub4 P287L, a β-tubulin mutant, revealed new aspects of microtubule regulation in shade. J Integr Plant Biol 57(9):757–769. doi:10.1111/jipb.12363
Acknowledgments
We thank Dr. Takashi Hashimoto for providing tub4 P287L seeds; Dr. Rong Yu and Tonglin Mao for providing 35S::GFP-TUA6 and 35S::EB1b-GFP seeds. This work was supported by the National Natural Science Foundation of China, 31171162, 31271298 to Y.T; Fundamental Research Funds for the Central Universities 2012121041 to Y.T.
Author information
Authors and Affiliations
Corresponding author
Additional information
J. Yu and K. Dong have equally contributed to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10725_2016_186_MOESM1_ESM.tif
Sup Fig. 1 Phenotypic characterization of sus1 mutants. a Mutation sites on TUB4 amino acid in the sus1 intragenic suppressors. b Positions (shown in white) of sus1 mutations in the 3D structural model of the αβ-tubulin dimer described by Nogales et al. (1998; PDB accession number, 1TUB). c mTUB4 fragment (with P287L mutation only) under the control of its own promoter complemented sus1 in shade. Representative seedlings are shown. The scale bars representing 2 mm. d Quantitative measurements of hypocotyl length and width of Col-0, sav2 and sus1 mutants grown in continuous white light (Wc) and shade. Error bars represent standard error of mean (SEM) (n≥10); ***P < 0.001 by Student’s t-test; NS, not significant (TIF 25139 KB)
10725_2016_186_MOESM2_ESM.tif
Sup Fig. 2 cMT patterns in hypocotyl epidermal cells of shade treated Col-0, sav2, sus2 and tub6 L246F. Scale bar represent 20 μm. (TIF 24570 KB)
10725_2016_186_MOESM3_ESM.tif
Sup Fig. 3 sus2 behaves in a semi-dominant manner. Segregation of WT-like, intermediate and sav2-like seedlings in dark-grown sus2(♀)×sav2 (♂) and sav2(♀)×sus2 (♂) F1 and F2 population. Segregation ratio in F2 population fitted the expected 1:2:1(χ2 = 0.113 and 0.148 < χ2 0.05,2 = 5.991) (TIF 6441 KB)
10725_2016_186_MOESM4_ESM.tif
Sup Fig. 4 Computational modeling showing L246F interacting with GTP after mutation. GTP-interacting residues in the wild type (top panel) and in the simulated mutant protein structure (lower panel) are shown. Yellow arrow heads indicate the residue equivalent to residue 246 in Arabidopsis TUB6 (TIF 32981 KB)
10725_2016_186_MOESM5_ESM.tif
Sup Fig. 5 tub6L246F did not exert its suppression effect through increasing its expression. a Quantitative RT-PCR results showing relative expression level of TUB6 in Col-0, sav2, sus2, and tub6 L246F under Wc and shade. Five-d-old light grown seedlings were treated with Wc or simulated shade for 2 h. Error bars represent standard error of mean (n=3). b Protein level of α- and β-tubulin did not change in sav2 and sus2. Tubulin was detected by western using anti-α-tubulin and anti-β-tubulin antibody. Commassie blue (CBB) stain of total proteins are shown as loading control. (TIF 14207 KB)
Rights and permissions
About this article
Cite this article
Yu, J., Dong, K., He, Q. et al. Studies on suppressors of sav2/shade avoidance 2 revealed altered interaction at the interface of αβ-tubulin intradimer affects microtubule dynamics. Plant Growth Regul 81, 71–79 (2017). https://doi.org/10.1007/s10725-016-0186-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-016-0186-7