Abstract
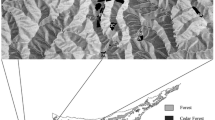
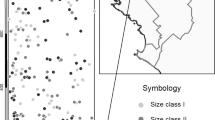
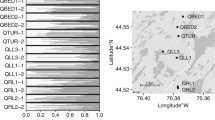
The distribution and abundance of genetic diversity in plant populations is initiated by sexually asymmetric propagule dispersal through pollen and seeds. Because these processes occur serially, it is not transparent how each contributes to subsequent patterns of genetic diversity. Using combined seedling/seed coat assay for naturally distributed seedlings of Quercus lobata Née, we extracted male and female gametic genotypes, and then assessed (wind-vectored) paternal and (gravity- and animal-vectored) maternal contributions to spatially distributed allelic diversity. We evaluated 200 naturally recruited seedlings from 4 open patches away from any adult canopies (denoted ‘open’), and 174 seedlings from 14 patches immediately beneath adult canopies (denoted ‘canopy’). The open patches included 19 % long distant dispersal events of >1 km while the canopy patches contained seedlings from one tree overhead. For each patch type, we partitioned average allelic diversity for six microsatellite loci for the whole study site (γ) into separate within-patch (α) and among-patch (β) components, translated into among-patch divergence (δ). We found that α-diversity resulting from seed dispersal was much less than that from pollen dispersal in both patch types, while total γ-diversity across the site was similar. Divergence (δ) among canopy patches was significantly greater than δ among open patches. We then evaluated spatial genetic autocorrelation (kinship) patterns for both open and canopy strata, separately for male and female gametes. Female gametes showed sharply declining kinship with increasing distance for canopy patches and modestly for open patches. In sharp contrast, male gametes from both patches showed only subtle decline of kinship, but seedlings still showed significant structure across patch types. On balance, sexual asymmetry in propagule dispersal shapes both the abundance and distribution of allelic diversity, with pollen dispersal promoting overall diversity but reducing spatial structure, but seed-dispersal reduces overall diversity and markedly increases spatial genetic structure.




Similar content being viewed by others
References
Austerlitz F, Smouse PE (2002) Two-generation analysis of pollen flow across a landscape IV. Estimating the dispersal parameter. Genetics 161:355–363
Austerlitz F, Dick CW, Dutech C, Klein EK, Oddou-Muratorio S, Smouse PE, Sork VL (2004) Using genetic markers to estimate the pollen dispersal curve. Mol Ecol 13(4):937–954
Austerlitz F, Dutech C, Smouse PE, Davis F, Sork VL (2007) Estimating anisotropic pollen dispersal: a case study in Quercus lobata. Heredity 99(2):193–204. doi:10.1038/sj.hdy.6800983
Chybicki IJ, Burczyk J (2010) Realized gene flow within mixed stands of Quercus robur L. and Q. petraea (Matt.) L. revealed at the stage of naturally established seedling. Mol Ecol 19(10):2137–2151. doi:10.1111/j.1365-294X.2010.04632.x
Crawford TJ (1984) The estimation of neighborhood parameters for plant populations. Heredity 52:273–283
Darley-Hill S, Johnson WC (1981) Acorn dispersal by the blue jay (Cyanocitta cristata). Oecologia 50(2):231–232
Dutech C, Sork VL, Irwin AJ, Smouse PE, Davis FW (2005) Gene flow and fine-scale genetic structure in a wind-pollinated tree species Quercus lobata (Fagaceaee). Am J Bot 92(2):252–261
Ennos RA (1994) Estimating the relative rates of pollen and seed migration among plant populations. Heredity 72:250–259. doi:10.1038/hdy.1994.35
Garcia C, Grivet D (2011) Molecular insights into seed dispersal mutualisms driving plant population recruitment. Acta Oecol 37(6):632–640. doi:10.1016/J.Actao.04.009
Garcia C, Jordano P, Godoy JA (2007) Contemporary pollen and seed dispersal in a Prunus mahaleb population: patterns in distance and direction. Mol Ecol 16(9):1947–1955. doi:10.1111/j.1365-294X.2006.03126.x
Gomez JM (2003) Spatial patterns in long-distance dispersal of Quercus ilex acorns by jays in a heterogeneous landscape. Ecography 26(5):573–584
Grivet D, Smouse PE, Sork VL (2005) A novel approach to an old problem: tracking dispersed seeds. Mol Ecol 14(11):3585–3595
Grivet D, Robledo-Arnuncio JJ, Smouse PE, Sork VL (2009) Relative contribution of contemporary pollen and seed dispersal to the effective parental size of seedling population of California valley oak (Quercus lobata, Née). Mol Ecol 18(19):3967–3979. doi:10.1111/j.1365-294X.2009.04326.x
Hamilton MB, Miller JR (2002) Comparing relative rates of pollen and seed gene flow in the island model using nuclear and organelle measures of population structure. Genetics 162(4):1897–1909
Heuertz M, Vekemans X, Hausman JF, Palada M, Hardy OJ (2003) Estimating seed versus pollen dispersal from spatial genetic structure in the common ash. Mol Ecol 12(9):2483–2495. doi:10.1046/j.135-294X.2003.01923.x
Heywood JS (1991) Spatial analysis of genetic-variation in plant populations. Annu Rev Ecol Syst 22:335–355. doi:10.1146/annurev.es.22.110191.002003
Holbrook KM, Smith TB, Hardesty BD (2002) Implications of long-distance movements of frugivorous rain forest hornbills. Ecography 25(6):745–749
Isagi Y, Saito D, Kawaguchi H, Tateno R, Watanabe S (2007) Effective pollen dispersal is enhanced by the genetic structure of an Aesculus turbinata population. J Ecol 95(5):983–990. doi:10.1111/j.1365-2745.2007.01272.x
Jost L (2007) Partitioning diversity into independent alpha and beta components. Ecology 88(10):2427–2439
Jost L (2008) GST and its relatives do not measure differentiation. Mol Ecol 17(18):4015–4026. doi:10.1111/j.1365-294X.2008.03887.x
Jost L (2010) Independence of alpha and beta diversities. Ecology 91(7):1969–1974
Jost L, DeVries P, Walla T, Greeney H, Chao A, Ricotta C (2010) Partitioning diversity for conservation analyses. Divers Distrib 16(1):65–76. doi:10.1111/j.1472-4642.2009.00626.x
Karubian J, Sork VL, Roorda T, Duraes R, Smith TB (2010) Destination-based seed dispersal homogenizes genetic structure of a tropical palm. Mol Ecol 19(8):1745–1753
Koenig WD, McEntee JP, Walters EL (2008) Acorn harvesting by acorn woodpeckers: annual variation and comparison with genetic estimates. Evol Ecol Res 10:811–822
Koenig WD, Krakauer AH, Monahan WB, Haydock J, Knops JMH, Carmen WJ (2009) Mast-producing trees and the geographical ecology of western scrub-jays. Ecography 32(4):561–570. doi:10.1111/j.1600-0587.2008.05617.x
Krauss SL, He T, Barrett LG, Lamont BB, Enright NJ, Miller BP, Hanley ME (2009) Contrasting impacts of pollen and seed dispersal on spatial genetic structure in the bird-pollinated Banksia hookeriana. Heredity 102(3):274–285. doi:10.1038/hdy.2008.118
Moran EV, Clark JS (2012) Between-site differences in the scale of dispersal and gene flow in red oak. PLoS One 7(5):e36492. doi:10.1371/journal.pone.0036492
Nei M (1972) Genetic distance between populations. Am Nat 106:283–292
Nei M (1973) Analysis of gene diversity in subdivided populations. Proc Natl Acad Sci USA 70(12):3321–3323
Nielsen R, Tarpy DR, Reeve HK (2003) Estimating effective paternity number in social insects and the effective number of alleles in a population. Mol Ecol 12:3157–3164
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6(1):288–295
Peakall R, Smouse PE (2012) GenAlEx 6.5: genetic analysis in Excel. Population genetic software for teaching and research-an update. Bioinformatics 28(19):2537–2539
Pluess AR, Sork VL, Dolan B, Davis FW, Grivet D, Merg K, Smouse PE (2009) Short distance pollen movement in a wind-pollinated tree, Quercus lobata (Fagaceae). For Ecol Manage 258(5):735–744. doi:10.1016/j.foreco.2009.05.014
R Core Team (2014) R: a language and environment for statistical computing
Ravigne V, Olivieri I, Gonzalez-Martinez SC, Rousset F (2006) Selective interactions between short-distance pollen and seed dispersal in self-compatible species. Evolution 60(11):2257–2271. doi:10.1554/05-352.1
Rousset F (2004) Genetic structure and selection in subdivided populations. Princeton University Press, Princeton
Schupp EW, Jordano P, Gómez JM (2010) Seed dispersal effectiveness revisited: a conceptual review. New Phytol 188(2):333–353
Scofield DG (2015a) diversityDispersal: analyse individual and genetic diversity of dispersal processes in biological populations. R package version 0.9.9000. https://github.com/douglasgscofield/diversityDispersal
Scofield DG (2015b) readGenalex: read and manipulate GenAlEx-format genotype files. R package version 0.4.1. http://CRAN.R-project.org/package=readGenalex
Scofield DG, Sork VL, Smouse PE (2010) Influence of acorn woodpecker social behaviour on transport of coast live oak (Quercus agrifolia) acorns in a southern California oak savanna. J Ecol 98(3):561–572
Scofield DG, Alfaro VR, Sork VL, Grivet D, Martinez E, Papp J, Smouse PE (2011) Foraging patterns of acorn woodpeckers (Melanerpes formicivorus) on valley oak (Quercus lobata Née) in two California oak savanna-woodlands. Oecologia 166(1):187–196
Scofield DG, Smouse PE, Karubian J, Sork VL (2012) Use of Alpha, Beta, and Gamma diversity measures to characterize seed dispersal by animals. Am Nat 180(6):719–732. doi:10.1086/668202
Simpson EH (1949) Measurement of diversity. Nature 163:688
Smouse PE, Peakall R (1999) Spatial autocorrelation analysis of individual multiallele and multilocus genetic structure. Heredity 82:561–573
Smouse PE, Dyer RJ, Westfall RD, Sork VL (2001) Two-generation analysis of pollen flow across a landscape. I. Male gamete heterogeneity among females. Evolution 55(2):260–271
Smouse PE, Peakall R, Gonzales E (2008) A heterogeneity test for fine-scale genetic structure. Mol Ecol 17(14):3389–3400
Smouse PE, Sork VL, Scofield DG, Grivet D (2012) Using seedling and pericarp tissues to determine maternal parentage of dispersed valley oak recruits. J Hered 103(2):250–259
Snedecor GW, Cochran WG (1989) Statistical methods, 8 ed. Iowa State University Press
Sork VL, Smouse PE (2006) Genetic analysis of landscape connectivity in tree populations. Landsc Ecol 21(6):821–836. doi:10.1007/s10980-005-5415-9
Sork V, Dyer R, Davis F, Smouse P (2002a) Mating system in California Valley oak, Quercus lobata Neé. In: Standiford RB, McCreary D, Purcell KL (eds) Proceedings of the fifth symposium on Oak Woodlands: Oaks in California’s changing landscape. 2001 October 22–25; San Diego, CA, Gen. Tech. Rep. PSW-GTR-184. Pacific Southwest Research Station, Forest Service, U.S. Department of Agriculture, Albany, CA, pp 427–444
Sork VL, Davis FW, Smouse PE, Apsit VJ, Dyer RJ, Fernandez-M JF, Kuhn B (2002b) Pollen movement in declining populations of California Valley oak, Quercus lobata: where have all the fathers gone? Mol Ecol 11(9):1657–1668
Thompson PT, Smouse PE, Scofield DG, Sork VL (2014) What seeds tell us about bird movement: a multi-year analysis of Acorn Woodpecker foraging patterns on two oak species. Mov Ecol. doi:10.1186/2051-3933-2-12
Tuomisto H (2010) A consistent terminology for quantifying species diversity? Yes, it does exist. Oecologia 164(4):853–860. doi:10.1007/s00442-010-1812-0
Tyler CM, Kuhn B, Davis FW (2006) Demography and recruitment limitations of three oak species in California. Q Rev Biol 81(2):127–152
Vekemans X, Hardy OJ (2004) New insights from fine-scale spatial genetic structure analyses in plant populations. Mol Ecol 13(4):921–935
Wenny DG (2001) Advantages of seed dispersal: a re-evaluation of directed dispersal. Evol Ecol Res 3(1):51–74
Whittaker RH (1960) Vegetation of the Siskiyou Mountains, Oregon and California. Ecol Monogr 30(3):279–338
Whittaker RH (1972) Evolution and measurement of species diversity. Taxon 21(2/3):213–251
Wright S (1943) Isolation by distance. Genetics 28:114–138
Wright S (1946) Isolation by distance under diverse systems of mating. Genetics 31:39–59
Zavaleta ES, Hulvey KB, Fulfrost B (2007) Regional patterns of recruitment success and failure in two endemic California oaks. Divers Distrib 13(6):735–745
Acknowledgments
The authors thank F. Davis (UCSB) for our early collaboration. The fieldwork was performed at the University of California Natural Reserve System Sedgwick Reserve administered by the UC Santa Barbara. We thank M. Williams, K. McCurdy, A. Lentz, and the staff at the Sedgwick Reserve for logistical support over the years. We thank F. Austerlitz and J. J. Arnuncio for intellectual stimulation. For technical assistance in genotyping and scoring genotypes of difficult tissue, we are grateful to C. Winder of the Sork lab and J. Papp and U. Dandekar of the UCLA Genotyping Core facility. We thank members of the Sork lab for comments on earlier versions of this manuscript. The research and V.L.S, D.G., D.G.S. were supported by NSF-DEB-0514956 and NSF-DEB-0089445; P.E.S. was supported by NSF-DEB-0514956 and USDA/NJAES-17111 and -17160.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sork, V.L., Smouse, P.E., Grivet, D. et al. Impact of asymmetric male and female gamete dispersal on allelic diversity and spatial genetic structure in valley oak (Quercus lobata Née). Evol Ecol 29, 927–945 (2015). https://doi.org/10.1007/s10682-015-9769-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-015-9769-4