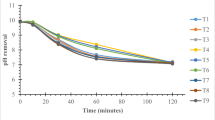
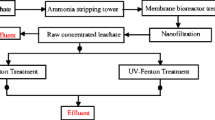
Abstract
Leachate from a closed landfill used for co-disposal of municipal and tannery waste was submitted to coagulation treatment, air stripping, adsorption on granular activated carbon, and Fenton oxidation with the aim to reduce toxicity of the leachate. Optimal operational conditions for each process were identified. The performance of the treatment was monitored by determination of organic matter (COD, DOC, BOD5), inorganic components (N-NH4 +, Cl−, alkalinity, metals), organic compounds (BTEX, PAHs, PCBs, OCPs) while changes in toxicity were followed by multiple toxicity tests. Among the applied treatment techniques, adsorption on granular activated carbon was the most efficient method for removal of organic matter and metals while air stripping was the most efficient for removal of N-NH4 + and reduction of toxicity. Lower reduction of organic matter content and toxicity was obtained during coagulation treatment. Fenton oxidation was effective for removal of COD; however, it negatively affected toxicity reduction. The combination of adsorption on granular activated carbon and air stripping led to an appreciable reduction of organic and inorganic pollutants and to leachate detoxification. Application of bioassays was helpful for assessing suitability of treatment methods and demonstrated that they are, together with physicochemical parameters, an indispensable part for monitoring of treatment efficiency.

Similar content being viewed by others
References
Abdul, J. M., Colville, A., Lim, R., Vigneswaran, S., & Kandasamy, J. (2012). Use of duckweed (Lemna disperma) to assess the phytotoxicity of the products of Fenton oxidation of metsulfuron methyl. Ecotoxicology and Environmental Safety, 83, 89–95.
Aziz, H. A., Alias, S., Adlan, M. N., Asaari, F. A. H., & Zahari, M. S. (2007). Colour removal from landfill leachate by coagulation and flocculation processes. Bioresource Technology, 98(1), 218–220.
Beaven, R. P., Knox, K., Gronow, J. R., Hjelmar, O., Greedy, D., & Scharff, H. (2014). A new economic instrument for financing accelerated landfill aftercare. Waste Management, 34(7), 1191–1198.
Benatti, C. T., da Costa, A. C., & Tavares, C. R. (2009). Characterization of solids originating from the Fenton’s process. Journal of Hazardous Materials, 163(2–3), 1246–1253.
Bernard, C., Colin, J., & Dû-Delepierre, A. L. (1997). Estimation of the hazard of landfills through toxicity testing of leachates: II. Comparison of physico-chemical characteristics of landfill leachates with their toxicity determined with a battery of tests. Chemosphere, 35(11), 2783–2796.
Bortolotto, T., Bertoldo, J. B., de Silveira, F. Z., Defaveri, T. M., Silvano, J., & Pich, C. T. (2009). Evaluation of the toxic and genotoxic potential of landfill leachates using bioassays. Environmental Toxicology and Pharmacology, 28(2), 288–293.
Bundschuh, M., Zubrod, J. P., & Schulz, R. (2011). The functional and physiological status of Gammarus fossarum (Crustacea; Amphipoda) exposed to secondary treated wastewater. Environmental Pollution, 159(1), 241–249.
Christensen, T. (2011). Solid waste technology and management. Chichester:John Wiley and Sons.
Clément, B., & Merlin, G. (1995). The contribution of ammonia and alkalinity to landfill leachate toxicity to duckweed. Science of the Total Environment, 170(1–2), 71–79.
Comstock, S. E. H., Boyer, T. H., Graf, K. C., & Townsend, T. G. (2010). Effect of landfill characteristics on leachate organic matter properties and coagulation treatability. Chemosphere, 81(7), 976–983.
Cortez, S., Teixeira, P., Oliveira, R., & Mota, M. (2010). Ozonation as polishing treatment of mature landfill leachate. Journal of Hazardous Materials, 182(1–3), 730–734.
Cossu, R. (2010). Technical evolution of landfilling. Waste Management, 30(6), 947–948.
Daphtoxkit F™ magna. (1996). Toxkit protokol, MicroBio Tests Inc.
Deng, Y., & Englehardt, J. D. (2006). Treatment of landfill leachate by Fenton process. Water Research, 40(20), 3683–3694.
Devare, M., & Bahadis, M. (1994). Biological monitoring of landfill leachate using plants and luminescent bacteria. Chemosphere, 28(2), 261–271.
Eckenfelder, W. W. (1970). Water pollution control: experimental procedures for process design. New York:Pemberton press.
EPA. (2003). Available on-line (31.12.2012): http://www.epa.gov/superfund/accomp/factsheets03/pownal.htm.
EPA 8100. (1986). Method for determination of polynuclear aromatic hydrocarbons (PAH).
Faust, R. A. (1995). Condensed toxicity summary for dibenz[a,h]antracene. Available on-line (22.4.2012): http://rais.ornl.gov/tox/profiles/dibenz_a_h_anthracene_c_V1.html.
Foo, K. Y., & Hameed, B. H. (2009). An overview of landfill leachate treatment via activated carbon adsorption process. Journal of Hazardous Materials, 171(1–3), 54–60.
Gonze, E., Commenges, N., Gonthier, Y., & Bernis, A. (2003). High frequency ultrasound as a pre- or a post-oxidation for paper mill wastewaters and landfill leachate treatment. Chemical Engineering Journal, 92(1–3), 215–225.
Guo, J.-S., Abbas, A. A., Chen, Y.-P., Liu, Z. P., Fang, F., & Chen, P. (2010). Treatment of landfill leachate using a combined stripping, Fenton, SBR, and coagulation process. Journal of Hazardous Materials, 178(1–3), 699–705.
ISO 5815-1. (2003). Water quality—determination of biochemical oxygen demand after n days (BODn), part 1: dilution and seeding method with allylthiourea addition. Edition 1, Geneva.
ISO 6060. (1989). Water quality—determination of the chemical oxygen demand. Edition 2, Geneva.
ISO 6468. (1996). Water quality—determination of certain organochlorine insecticides, polychlorinated biphenyls and chlorobenzenes—gas chromatographic method after liquid–liquid extraction. Edition 1, Geneva.
ISO 7150–1. (1984). Water quality—determination of ammonium, part 1: manual spectrometric method. Edition 1, Geneva.
ISO 8192. (2007). Water quality—test for inhibition of oxygen consumption by activated sludge for carbonaceous and ammonium oxidation. Edition 2, Geneva.
ISO 8245. (1999). Water quality—guidelines for the determination of total organic carbon (TOC) and dissolved organic carbon (DOC). Edition 2, Geneva.
ISO 9562. (2004). Water quality—determination of adsorbable organically bound halogens (AOX). Edition 3, Geneva.
ISO 10530. (1992). Water quality—determination of dissolved sulfide—photometric method using methylene blue. Edition 1, Geneva.
ISO 11348–3. (2007). Water quality—determination of the inhibitory effect of water samples on the light emission of Vibrio fischeri (Luminescent bacteria test)—part 3: method using freezed-dried bacteria. Edition 2, Geneva.
ISO 11423-1. (1997). Water quality—determination of benzene and some derivatives—part 1: head-space gas chromatographic method. Edition 1, Geneva.
ISO 20079. (2005). Water quality—determination of the toxic effect of water constituents and waste water on duckweed (Lemna minor)—duckweed growth inhibition test. Edition 1, Geneva.
Jemec, A., Tišler, T., & Žgajnar Gotvajn, A. (2012). Assessment of landfill leachate toxicity reduction after biological treatment. Archives of Environmental Contamination and Toxicology, 62(2), 210–221.
Kalčíková, G., Vávrová, M., Zagorc-Končan, J., & Žgajnar Gotvajn, A. (2011). Evaluation of the hazardous impact of landfill leachates by toxicity and biodegradability tests. Environmental Technology, 32(12), 1345–1353.
Klimiuk, E., & Kulikowska, D. (2004). Effectiveness of organics and nitrogen removal from municipal landfill leachate in single- and two-stage SBR systems. Polish Journal of Environmental Studies, 13(5), 525–532.
Kulikowska, D., & Klimiuk, E. (2008). The effect of landfill age on municipal leachate composition. Bioresource Technology, 99(13), 5981–5985.
Kurniawan, T. A., Lo, W. H., & Chan, G. Y. S. (2006). Physico-chemical treatments for removal of recalcitrant contaminants from landfill leachate. Journal of Hazardous Materials, 129(1–3), 80–100.
Municipality Vrhnika. (2009). Available on-line (29.12.2012): http://www.vrhnika.si/?m=fotoreportaze&id=8287#.
MŽP (2007). The guideline of the Department of Waste Management to determine the ecotoxicity of waste. Prague:Ministry of the Environment of the Czech Republic (In Czech).
Noma, Y., Yamane, S., & Kida, A. (2001). Adsorbable organic halides (AOX), AOX formation potential, and PCDDs/DFs in landfill leachate and their removal in water treatment processes. Journal of Material Cycles and Waste Management, 3(2), 126–134.
Official Gazette of Republic of Slovenia (2008). Decree on the emission of substance in the discharge of landfill leachate. ULRS, 6, 8219–8221 (In Slovene).
Paxéus, N. (2000). Organic compounds in municipal landfill leachates. Water Science and Technology, 42(7–8), 323–333.
Persoone, G., Jasperse, E., & Claus, C. (1984). Ecotoxicological testing for the marine envirnoment. Vol. 2. Breden:State University Ghent and Institute for Marine Scientific Research.
Qu, X., He, P. J., Shao, L. M., & Lee, D. J. (2008). Heavy metals mobility in full-scale bioreactor landfill: initial stage. Chemosphere, 70(5), 769–777.
Renou, S., Givaudan, J. G., Poulain, S., Dirassouyan, F., & Moulin, P. (2008). Landfill leachate treatment: review and opportunity. Journal of Hazardous Materials, 150(3), 468–493.
Rivas, F. J., Beltrán, F., Carvalho, F., Acedo, B., & Gimeno, O. (2004). Stabilized leachates: sequential coagulation–flocculation + chemical oxidation process. Journal of Hazardous Materials, 116(1–2), 95–102.
Singh, S. K., Moody, C. M., & Townsend, T. G. (2014). Ozonation pretreatment for stabilized landfill leachate high-pressure membrane treatment. Desalination, 344, 163–170.
Silva, A. C., Dezotti, M., & Sant’Anna Jr., G. L. (2004). Treatment and detoxification of a sanitary landfill leachate. Chemosphere, 55(2), 207–214.
Sri Shalini, S., & Joseph, K. (2012). Nitrogen management in landfill leachate: application of SHARON, ANAMMOX and combined SHARON-ANAMMOX process. Waste Management, 32(12), 2385–2400.
Standard methods for the examination of water and wastewater. (2012). Edition 22. Washington: American Water Works Association/American Public Works Association/Water Environment.
Svensson, B. M., Mathiasson, L., Mårtensson, L., & Bergström, S. (2005). Artemia salina as test organism for assessment of acute toxicity of leachate water from landfills. Environmental Monitoring and Assessment, 102(1–3), 309–321.
Tekin, H., Bilkay, O., Ataberk, S. S., Balta, T. H., Ceribasi, I. H., Sanin, F. D., Dilek, F. B., & Yetis, U. (2006). Use of Fenton oxidation to improve the biodegradability of a pharmaceutical wastewater. Journal of Hazardous Materials, 136(2), 258–265.
Tigini, V., Prigione, V., & Varese, G. C. (2014). Mycological and ecotoxicological characterisation of landfill leachate before and after traditional treatments. Science of the Total Environment, 487, 335–341.
Timur, H., & Özturk, I. (1999). Anaerobic sequencing batch reactor treatment of landfill leachate. Water Research, 33(15), 3225–3230.
Tsarpali, V., Kamilari, M., & Dailianis, S. (2012). Seasonal alterations of landfill leachate composition and toxic potency in semi-arid regions. Journal of Hazardous Materials, 233–234, 163–171.
Wilke, B. M., Riepert, F., Koch, C., & Kühne, T. (2008). Ecotoxicological characterization of hazardous waste. Ecotoxicology and Environmental Safety, 70(2), 283–293.
Žgajnar Gotvajn, A., Tišler, T., & Zagorc-Končan, J. (2009). Comparison of different treatment strategies for industrial landfill leachate. Journal of Hazardous Materials, 162(2–3), 1446–1456.
Žgajnar Gotvajn, A., Zagorc-Končan, J., & Cotman, M. (2011). Fenton’s oxidative treatment of municipal landfill leachate as an alternative to biological process. Desalination, 275(1–3), 269–275.
Ziyang, L., Youcai, Z., Tao, Y., Yu, S., Huili, C., Nanwen, Z., & Renhua, H. (2009). Natural attenuation and characterization of contaminants composition in landfill leachate under different disposing ages. Science of the Total Environment, 407(10), 3385–3391.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalčíková, G., Zupančič, M., Levei, E.A. et al. Application of multiple toxicity tests in monitoring of landfill leachate treatment efficiency. Environ Monit Assess 187, 489 (2015). https://doi.org/10.1007/s10661-015-4670-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-015-4670-8