Summary
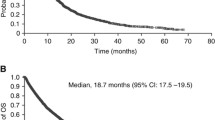
Sunitinib is associated with a robust objective response rate in patients with metastatic clear cell renal cell carcinoma (RCC). The primary objective of this phase II clinical trial was to assess the overall response rate for sunitinib in patients with papillary metastatic RCC as well as other non-clear cell histologies. A Simon 2-stage design was used to determine the number of papillary metastatic RCC patients for enrollment, and allowed for descriptive response data for other non-clear cell histologies. Twenty-three patients were enrolled, including 8 with papillary renal cell carcinoma (RCC) and the remainder with other non-clear cell histologies (unclassified in 5 patients). All patients received 50 mg of oral sunitinib in cycles of 4 weeks followed by 2 weeks of rest (4/2). The trial was stopped early because of slow accrual; no responses were observed in the 8 patients with papillary RCC. In the 22 evaluable patients, best response to sunitinib included a partial response in 1 patient with unclassified RCC, stable disease in 15, and progression in 6. The median progression-free survival was 5.5 months (95% CI, 2.5–7.1) in all 23 patients, and 5.6 months for the 8 papillary patients (95% CI, 1.4–7.1). The robust objective responses sunitinib had produced in clear cell RCC could not be demonstrated in this study comprised of patients with non-clear cell histologies.


Similar content being viewed by others
Abbreviations
- mRCC:
-
Metastatic renal cell carcinoma
- RCC:
-
Renal cell carcinoma
- HLRCC:
-
Hereditary leiomyomatosis and renal cell cancer
- CTCAE:
-
Common toxicity criteria for adverse events
- RECIST:
-
Response evaluation criteria in solid tumors
References
Motzer RJ, Bander NH, Nanus DM (1996) Renal-cell carcinoma. N Engl J Med 335:865–875
Reuter VE, Presti JC Jr (2000) Contemporary approach to the classification of renal epithelial tumors. Semin Oncol 27:124–137
van den Berg E, van der Hout AH, Oosterhuis JW, Storkel S, Dijkhuizen T, Dam A, Zweers HM, Mensink HJ, Buys CH, de Jong B (1993) Cytogenetic analysis of epithelial renal-cell tumors: relationship with a new histopathological classification. Int J Cancer 55:223–227
Weiss LM, Gelb AB, Medeiros LJ (1995) Adult renal epithelial neoplasms. Am J Clin Pathol 103:624–635
Kovacs G, Szucs S, De Riese W, Baumgartel H (1987) Specific chromosome aberration in human renal cell carcinoma. Int J Cancer 40:171–178
Fleming S (1993) The impact of genetics on the classification of renal carcinoma. Histopathology 22:89–92
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O, Oudard S, Negrier S, Szczylik C, Kim ST, Chen I, Bycott PW, Baum CM, Figlin RA (2007) Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 356:115–124
Hudes G, Carducci M, Tomczak P, Dutcher J, Figlin R, Kapoor A, Staroslawska E, Sosman J, McDermott D, Bodrogi I, Kovacevic Z, Lesovoy V, Schmidt-Wolf IG, Barbarash O, Gokmen E, O’Toole T, Lustgarten S, Moore L, Motzer RJ (2007) Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med 356:2271–2281
Motzer RJ, Escudier B, Oudard S, Hutson TE, Porta C, Bracarda S, Grunwald V, Thompson JA, Figlin RA, Hollaender N, Urbanowitz G, Berg WJ, Kay A, Lebwohl D, Ravaud A (2008) Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet 372:449–456
Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, Negrier S, Chevreau C, Solska E, Desai AA, Rolland F, Demkow T, Hutson TE, Gore M, Freeman S, Schwartz B, Shan M, Simantov R, Bukowski RM (2007) Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 356:125–134
Escudier B, Koralewski P, Pluzanska A, Ravaud A, Bracarda S, Szczylik C, Chevreau C, Filipek M, Melichar B, Moore N, on behalf of the Ai (2007) A randomized, controlled, double-blind phase III study (AVOREN) of bevacizumab/interferon-{alpha}2a vs placebo/interferon-{alpha}2a as first-line therapy in metastatic renal cell carcinoma. J Clin Oncol (Meeting Abstracts) 25:3
Rini BI, Halabi S, Rosenberg JE, Stadler WM, Vaena DA, Ou SS, Archer L, Atkins JN, Picus J, Czaykowski P, Dutcher J, Small EJ (2008) Bevacizumab plus interferon alfa compared with interferon alfa monotherapy in patients with metastatic renal cell carcinoma: CALGB 90206. J Clin Oncol 26:5422–5428
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Oudard S, Negrier S, Szczylik C, Pili R, Bjarnason GA, Garcia-del-Muro X, Sosman JA, Solska E, Wilding G, Thompson JA, Kim ST, Chen I, Huang X, Figlin RA (2009) Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol 27:3584–3590
Mai KT, Landry DC, Robertson SJ, Commons AS, Burns BF, Thijssen A, Collins J (2001) A comparative study of metastatic renal cell carcinoma with correlation to subtype and primary tumor. Pathol Res Pract 197:671–675
Motzer RJ, Bacik J, Mariani T, Russo P, Mazumdar M, Reuter V (2002) Treatment outcome and survival associated with metastatic renal cell carcinoma of non-clear-cell histology. J Clin Oncol 20:2376–2381
Upton MP, Parker RA, Youmans A, McDermott DF, Atkins MB (2005) Histologic predictors of renal cell carcinoma response to interleukin-2-based therapy. J Immunother 28:488–495
Motzer RJ, Russo P (2000) Systemic therapy for renal cell carcinoma. J Urol 163:408–417
Choueiri TK, Plantade A, Elson P, Negrier S, Ravaud A, Oudard S, Zhou M, Rini BI, Bukowski RM, Escudier B (2008) Efficacy of sunitinib and sorafenib in metastatic papillary and chromophobe renal cell carcinoma. J Clin Oncol 26:127–131
Simon R (1989) Optimal two-stage designs for phase II clinical trials. Control Clin Trials 10:1–10
Kaplan EL, Meier P (1958) Nonparametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Motzer RJ, Rini BI, Bukowski RM, Curti BD, George DJ, Hudes GR, Redman BG, Margolin KA, Merchan JR, Wilding G, Ginsberg MS, Bacik J, Kim ST, Baum CM, Michaelson MD (2006) Sunitinib in patients with metastatic renal cell carcinoma. JAMA 295:2516–2524
Ravaud A, Oudard S, Gravis-Mescam G, Sevin E, Zanetta S, Theodore C, de Fromont M, Mahier-Ait Oukhatar C, Chene G, Escudier B (2009) First-line sunitinib in type I and II papillary renal cell carcinoma (PRCC): SUPAP, a phase II study of the French Genito-Urinary Group (GETUG) and the Group of Early Phase trials (GEP). J Clin Oncol (Meeting Abstracts) 27:5146
Plimack ER, Jonasch E, Bekele BN, Smith LA, Araujo JC, Tannir NM (2008) Sunitinib in non-clear cell renal cell carcinoma (ncc-RCC): a phase II study. J Clin Oncol (Meeting Abstracts) 26:5112
Plimack ER, Jonasch E, Bekele BN, Qiao W, Ng CS, Tannir NM (2010) Sunitinib in papillary renal cell carcinoma (pRCC): results from a single-arm phase II study. J Clin Oncol (Meeting Abstracts) 28:4604
Gordon MS, Hussey M, Nagle RB, Lara PN Jr, Mack PC, Dutcher J, Samlowski W, Clark JI, Quinn DI, Pan CX, Crawford D (2009) Phase II study of erlotinib in patients with locally advanced or metastatic papillary histology renal cell cancer: SWOG S0317. J Clin Oncol 27:5788–5793
Acknowledgements
This study was supported in part by Pfizer, Inc. The authors wish to thank Carol Pearce, MSKCC Department of Medicine writer/editor, for her review of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Molina, A.M., Feldman, D.R., Ginsberg, M.S. et al. Phase II trial of sunitinib in patients with metastatic non-clear cell renal cell carcinoma. Invest New Drugs 30, 335–340 (2012). https://doi.org/10.1007/s10637-010-9491-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-010-9491-6