Abstract
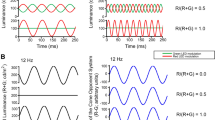
Period doubling in the full-field cone flicker electroretinogram (ERG) refers to an alternation in waveform amplitude and/or shape from cycle to cycle, presumably owing to the operation of a nonlinear gain control mechanism. This study examined the influence of stimulus chromatic properties on the characteristics of period doubling in order to better understand the underlying mechanism. ERGs were acquired from 5 visually normal subjects in response to sinusoidally modulated flicker presented at frequencies from 25 to 100 Hz. The test stimuli and the pre-stimulus adaptation were either long wavelength (R), middle wavelength (G), or an equal combination of long and middle wavelengths (Y), all equated for photopic luminance. Fourier analysis was used to obtain the response amplitude at the stimulus frequency F and at a harmonic frequency of 3F/2, which was used as the index of period doubling. The frequency–response function for 3F/2 typically showed two peaks, occurring at approximately 33.3 and 50 Hz. However, the magnitude of period doubling within these frequency regions was dependent on the chromatic properties of both the test stimulus and the pre-stimulus adaptation. Period doubling was generally smallest when an R test was used, even though the stimuli were luminance-equated and the amplitude of F did not differ between the various conditions. The pattern of results indicates that the mechanism that generates period doubling is influenced by chromatic signals from both the test stimulus and the pre-stimulus adaptation, even though the high stimulus frequencies presumably favor the achromatic luminance system.






Similar content being viewed by others
References
Marmor MF, Fulton AB, Holder GE, Miyake Y, Brigell M, Bach M (2009) ISCEV standard for full-field clinical electroretinography (2008 update). Doc Ophthalmol 118(1):69–77
Teresa RC (1984) Abnormal discharges and chaos in a neuronal model system. Biol Cybern 50(4):301–311
Canavier CC, Clark JW, Byrne JH (1990) Routes to chaos in a model of a bursting neuron. Biophys J 57(6):1245–1251
Stone L (1993) Period-doubling reversals and chaos in simple ecological models. Nature 365(6447):617–620
Crevier DW, Meister M (1998) Synchronous period-doubling in flicker vision of salamander and man. J Neurophysiol 79(4):1869–1878
Shah MR, Alexander KR, Ripps H, Qian H (2010) Characteristics of period doubling in the rat cone flicker ERG. Exp Eye Res 90(2):196–202
Qian H, Alexander KR, Ripps H (2010) Harmonic analysis of the cone flicker ERG of rabbit. Exp Eye Res 91(6):811–817
Alexander KR, Levine MW, Super BJ (2005) Characteristics of period doubling in the human cone flicker electroretinogram. Visual Neurosci 22(6):817–824
Schwartz G, Berry MJ 2nd (2008) Sophisticated temporal pattern recognition in retinal ganglion cells. J Neurophysiol 99(4):1787–1798
Alexander KR, Raghuram A (2007) Effect of contrast on the frequency response of synchronous period doubling. Vision Res 47(4):555–563
Alexander KR, Raghuram A, McAnany JJ (2008) Comparison of spectral measures of period doubling in the cone flicker electroretinogram. Doc Ophthalmol 117(3):197–203
Kremers J, Rodrigues AR, Silveira LC, da Silva Filho M (2010) Flicker ERGs representing chromaticity and luminance signals. Invest Ophthalmol Vis Sci 51(1):577–587
Eisner A, MacLeod DIA (1981) Flicker photometric study of chromatic adaptation: selective suppression of cone inputs by colored backgrounds. J Opt Soc Am 71(6):705–716
Stromeyer CF, Cole GR, Kronauer RE (1987) Chromatic suppression of cone inputs to the luminance flicker mechanism. Vision Res 27(7):1113–1137
Swanson WH (1993) Chromatic adaptation alters spectral sensitivity at high temporal frequencies. J Opt Soc Am A: 10(6):1294–1303
Wiesel TN, Hubel DH (1966) Spatial and chromatic interactions in the lateral geniculate body of the rhesus monkey. J Neurophysiol 29(6):1115–1156
Lee BB, Martin PR, Valberg A (1988) The physiological basis of heterochromatic flicker photometry demonstrated in the ganglion cells of the macaque retina. J Physiol 404:323–347
Smith VC, Lee BB, Pokorny J, Martin PR, Valberg A (1992) Responses of macaque ganglion cells to the relative phase of heterochromatically modulated lights. J Physiol 458:191–221
Lee BB, Sun H (2009) The chromatic input to cells of the magnocellular pathway of primates. J Vis 9(2):15, 1–18
Padmos P, Van Norren D (1971) Cone spectral sensitivity and chromatic adaptation as revealed by human flicker electroretinography. Vision Res 11(1):27–42
Kremers J, Stepien MW, Scholl HPN, Saito C (2003) Cone selective adaptation influences L- and M-cone driven signals in electroretinography and psychophysics. J Vis 3(2):146–160
Cao D, Pokorny J, Smith VC (2005) Associating color appearance with the cone chromaticity space. Vision Res 45(15):1929–1934
Alexander KR, Barnes CS, Fishman GA (2003) ON-pathway dysfunction and timing properties of the flicker ERG in carriers of X-linked retinitis pigmentosa. Invest Ophthalmol Vis Sci 44(9):4017–4025
Acknowledgments
This research was supported by NIH research grant 5R01EY008301 and ARRA grant 3R01EY008301-18S1 (KRA), NIH Core Grant EY001792, and an unrestricted departmental grant from Research to Prevent Blindness.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gowrisankaran, S., Alexander, K.R. Stimulus chromatic properties affect period doubling in the human cone flicker ERG. Doc Ophthalmol 125, 21–29 (2012). https://doi.org/10.1007/s10633-012-9326-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10633-012-9326-1