Abstract
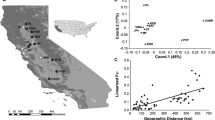
Conservation of rare species requires careful consideration to both preserve locally adapted traits and maintain genetic diversity, as species’ ranges fluctuate in response to a changing climate and habitat loss. Salt marsh systems in California have been highly modified and many salt marsh obligate species have undergone range reductions and habitat loss with concomitant losses of genetic diversity and connectivity. Remaining salt marshes are threatened by rising sea levels, and so these habitats will likely require active restoration and re-establishment efforts. This study aims to provide a reference point for the current status of genetic diversity and range-wide population structure of a federally and state listed endangered plant, salt marsh bird’s-beak (Chloropyron maritimum subsp. maritimum) that can inform future preservation and restoration efforts. We used historical data and current monitoring information to locate and sample all known occurrences throughout the range of this subspecies in southern California, and three additional occurrences from Baja California, Mexico. We used flow cytometry and single nucleotide polymorphic markers (SNPs), generated by double-digest restriction-site associated DNA sequencing (ddRAD), to assess relative ploidy, and estimate genetic diversity and population structure across the region. Overall, we found five distinct genetic clusters that coincide with geographic regions. Genetic diversity was greatest in the southern part of the range including Baja California and San Diego. These findings can bolster management and restoration efforts by identifying potentially isolated occurrences and areas that are the richest sources of allelic diversity, and by providing insight into the amount of genetic differentiation across the taxon’s range.





Similar content being viewed by others
References
Allendorf FW (1986) Genetic drift and the loss of alleles versus heterozygosity. Zoo Biology 5:181–190. https://doi.org/10.1002/zoo.1430050212
Ayres DR, Fleishman E, Launer A et al (2015) Genetic structure and demography of Chloropyron palmatum, an endangered annual plant. Madroño 62:139–149. https://doi.org/10.3120/madr-62-03-139-149.1
Broadhurst LM, Lowe A, Coates DJ et al (2008) Seed supply for broadscale restoration: maximizing evolutionary potential. Evol Appl. https://doi.org/10.1111/j.1752-4571.2008.00045.x
Bucharova A, Bossdorf O, Hölzel N et al (2019) Mix and match: regional admixture provenancing strikes a balance among different seed-sourcing strategies for ecological restoration. Conserv Genet 20:7–17. https://doi.org/10.1007/s10592-018-1067-6
Burns KJ, Barhoum DN (2006) Population-level history of the wrentit (Chamaea fasciata): implications for comparative phylogeography in the California Floristic Province. Mol Phylogenet Evol 38:117–129. https://doi.org/10.1016/j.ympev.2005.07.008
Calsbeek R, Thompson JN, Richardson JE (2003) Patterns of molecular evolution and diversification in a biodiversity hotspot: the California Floristic Province. Mol Ecol 12:1021–1029. https://doi.org/10.1046/j.1365-294X.2003.01794.x
Catchen JM, Amores A, Hohenlohe P et al (2011) Stacks: Building and genotyping loci de novo from short-read sequences. G3: Genes, Genomes. Genetics 1:171–182. https://doi.org/10.1534/g3.111.000240
Catchen JM, Hohenlohe PA, Bassham S et al (2013) Stacks: an analysis tool set for population genomics. Mol Ecol 22:3124–3140. https://doi.org/10.1111/mec.12354
Chuang T-I, Heckard LR (1971) Observations on root-parasitism in Cordylanthus (Scrophulariaceae). Am J Bot 58:218–228
Chuang TI, Heckard LR (1973) Taxonomy of Cordylanthus subgenus Hemistegia. (Scrophulariaceae) Brittonia 25:135–158. https://doi.org/10.2307/2805932
Dolezel J, Greilhuber J, Suda J (2007) Estimation of nuclear DNA content in plants using flow cytometry. Nat Protoc 2:2233–2244. https://doi.org/10.1038/nprot.2007.310
Edmands S (2001) Phylogeography of the intertidal copepod Tigriopus californicus reveals substantially reduced population differentiation at northern latitudes. Mol Ecol 10:1743–1750. https://doi.org/10.1046/j.0962-1083.2001.01306.x
Ellstrand NC, Elam DR (1993) Population genetic consequences of small population size: implications for plant conservation. Annu Rev Ecol Syst 24:217–242
Engelhardt KAM, Lloyd MW, Neel MC (2014) Effects of genetic diversity on conservation and restoration potential at individual, population, and regional scales. Biol Cons 179:6–16. https://doi.org/10.1016/j.biocon.2014.08.011
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491
Excoffier L, Foll M, Petit RJ (2009) Genetic consequences of range expansions. Annu Rev Ecol Evol Syst 40:481–501. https://doi.org/10.1146/annurev.ecolsys.39.110707.173414
Fellows MQN, Zedler JB (2005) Effects of the non-native grass, Parapholis incurva (Poaceae), on the rare and endangered hemiparasite, Cordylanthus maritimus subsp. maritimus (Scrophulariaceae). Madroño 52:91–98
Frankham R (2005) Genetics and extinction. Biol Cons 126:131–140. https://doi.org/10.1016/j.biocon.2005.05.002
Frankham R, Ballou JD, Briscoe DA (2002) Introduction to Conservation Genetics. Cambridge University Press, Cambridge
Gedan KB, Silliman BR, Bertness MD (2009) Centuries of human-driven change in salt marsh ecosystems. Ann Rev Mar Sci 1:117–141. https://doi.org/10.1146/annurev.marine.010908.163930
Gibson AL, Fishman L, Nelson CR (2017) Polyploidy: a missing link in the conversation about seed transfer of a commonly seeded native grass in western North America. Restor Ecol 25:184–190. https://doi.org/10.1111/rec.12408
Gilman IS, Tank DC (2018) Species tree estimation using ddRADseq data from historical specimens confirms the monophyly of highly disjunct species of Chloropyron (Orobanchaceae). SBOT 43:701–708. https://doi.org/10.1600/036364418X697418
Godefroid S, Piazza C, Rossi G et al (2011) How successful are plant species reintroductions? Biol Cons 144:672–682. https://doi.org/10.1016/j.biocon.2010.10.003
Harrisson KA, Pavlova A, Telonis-Scott M, Sunnucks P (2014) Using genomics to characterize evolutionary potential for conservation of wild populations. Evol Appl 7:1008–1025. https://doi.org/10.1111/eva.12149
Helenurm K, Parsons LS (1997) Genetic variation and reintroduction of Cordylanthus maritimus ssp. maritimus to Sweetwater Marsh, California. Restor Ecol 5:236–244
Hohenlohe PA, Bassham S, Etter PD et al (2010) Population genomics of parallel adaptation in threespine stickleback using sequenced RAD tags. PLoS Genet 6:e1000862. https://doi.org/10.1371/journal.pgen.1000862
Hopkins DR, Parker VT (1984) A study of the seed bank of a salt marsh in northern San Francisco. Am J Bot 71:348–355. https://doi.org/10.1002/j.1537-2197.1984.tb12522.x
Hughes AR, Inouye BD, Johnson MTJ et al (2008) Ecological consequences of genetic diversity. Ecol Lett 11:609–623. https://doi.org/10.1111/j.1461-0248.2008.01179.x
Jombart T (2008) adegenet: An R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405. https://doi.org/10.1093/bioinformatics/btn129
Jones RN, Viegas W, Houben A (2008) A century of B chromosomes in plants: so what? Ann Bot 101:767–775. https://doi.org/10.1093/aob/mcm167
Lawson DJ, Hellenthal G, Myers S, Falush D (2012) Inference of population structure using dense haplotype data. PLOS Genetics 8:e1002453. https://doi.org/10.1371/journal.pgen.1002453
Lincoln PG (1985) Pollinator effectiveness and ecology of seed set in Cordylanthus maritimus ssp. maritimus at Point Mugu, California. USFWS - Endangered Species Office, Sacramento, CA
Malinsky M, Trucchi E, Lawson DJ, Falush D (2018) RADpainter and fineRADstructure: population inference from RADseq data. Mol Biol Evol 35:1284–1290. https://doi.org/10.1093/molbev/msy023
Markert JA, Champlin DM, Gutjahr-Gobell R et al (2010) Population genetic diversity and fitness in multiple environments. BMC Evol Biol 10:205. https://doi.org/10.1186/1471-2148-10-205
Maschinski J, Albrecht MA (2017) Center for Plant Conservation’s best practice guidelines for the reintroduction of rare plants. Plant Divers 39:390–395. https://doi.org/10.1016/j.pld.2017.09.006
Maschinski J, Falk DA, Wright SJ et al (2012) Optimal locations for plant reintroductions in a changing world. In: Maschinski J, Haskins KE, Raven PH (eds) Plant Reintroduction in a Changing Climate: Promises and Perils. Island Press/Center for Resource Economics, Washington, DC, pp 109–129
McGranahan G, Balk D, Anderson B (2007) The rising tide: assessing the risks of climate change and human settlements in low elevation coastal zones. Environ Urban 19:17–37. https://doi.org/10.1177/0956247807076960
McKay JK, Christian CE, Harrison S, Rice KJ (2005) “How local is local?”-A review of practical and conceptual issues in the genetics of restoration. Restor Ecol 13:432–440. https://doi.org/10.1111/j.1526-100X.2005.00058.x
Meirmans PG, Tienderen PHV (2004) Genotype and Genodive: two programs for the analysis of genetic diversity of asexual organisms. Mol Ecol Notes 4:792–794. https://doi.org/10.1111/j.1471-8286.2004.00770.x
Michalakis Y, Excoffier L (1996) A generic estimation of population subdivision using distances between alleles with special reference for microsatellite loci. Genetics 142:1061–1064
Nerem RS, Beckley BD, Fasullo JT et al (2018) Climate-change–driven accelerated sea-level rise detected in the altimeter era. PNAS 115:2022–2025. https://doi.org/10.1073/pnas.1717312115
Newman J (1981) Aspects of Cordylanthus maritimus subsp. maritimus germination tests examined. Notes on studies performed at Pacifica Missile Test Center, Naval Air Station, Point Mugu, CA. Pt. Mugu, CA
Noe GB, Zedler JB (2001) Variable rainfall limits the germination of upper intertidal marsh plants in southern California. Estuaries 24:30–40. https://doi.org/10.2307/1352810
Noe GB, Fellows MQN, Parsons L et al (2019) Adaptive management assists reintroduction as higher tides threaten an endangered salt marsh plant. Restor Ecol 27:750–757. https://doi.org/10.1111/rec.12934
Paris JR, Stevens JR, Catchen JM, Johnston S (2017) Lost in parameter space: a road map for stacks. Methods Ecol Evol 8:1360–1373. https://doi.org/10.1111/2041-210x.12775
Parris AS, Bromirski P, Burkett V et al (2012) Global sea level rise scenarios for the United States National Climate Assessment
Parsons LS, Zedler JB (1997) Factors affecting reestablishment of an endangered annual plant at a California salt marsh. Ecol Appl 7:253–267. https://doi.org/10.1890/1051-0761(1997)007[0253:FAROAE]2.0.CO;2
Peterson BK, Weber JN, Kay EH et al (2012) Double digest RADseq: an inexpensive method for de novo SNP discovery and genotyping in model and non-model species. PLoS One 7:e37135. https://doi.org/10.1371/journal.pone.0037135
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Purer EA (1942) Plant ecology of the coastal salt marshlands of San Diego County, California. Ecol Monogr 12:81–111. https://doi.org/10.2307/1948423
San Diego Natural History Museum, Botany Dept (2018) A report of genetic sample collections and curation for six rare plants within the San Diego MSPA San Diego County, California. SANDAG
San Diego Management and Monitoring Program (2019) Management and Monitoring Strategic Plan (MSP Roadmap) 2019 Rare plant inspect and manage monitoring protocol for occurrences on conserved lands in Western San Diego County
Slatkin M (1987) Gene flow and the geographic structure of natural populations. Science 236:787–792
Spielman D, Brook BW, Briscoe DA, Frankham R (2004) Does inbreeding and loss of genetic diversity decrease disease resistance? Conserv Genet 5:439–448. https://doi.org/10.1023/B:COGE.0000041030.76598.cd
Statham MJ, Aamoth S, Barthman-Thompson L et al (2016) Conservation genetics of the endangered San Francisco Bay endemic salt marsh harvest mouse (Reithrodontomys raviventris). Conserv Genet 17:1055–1066. https://doi.org/10.1007/s10592-016-0843-4
Thorne KM, MacDonald GM, Ambrose RF et al (2016) Effects of climate change on tidal marshes along a latitudinal gradient in California. U.S. Geological Survey, Reston
Thorne K, MacDonald G, Guntenspergen G et al (2018) U.S. Pacific coastal wetland resilience and vulnerability to sea-level rise. Sci Adv 4:eaao3270. https://doi.org/10.1126/sciadv.aao3270
U.S. Fish and Wildlife Service (1978) Endangered and threatened wildlife and plants; determination of five plants as endangered species
U.S. Fish and Wildlife Service (1984) Recovery plan for salt-marsh bird’s-beak (Cordylanthus maritimus subsp. maritimus). Portland, OR
U.S. Fish and Wildlife Service (2009) Chloropyron maritimum subsp. maritimum (Cordylanthus maritimus subsp. maritimus) (salt marsh bird’s-beak) 5-year review: summary and evaluation. Carlsbad, CA
U.S. Global Change Research Program (2017) Climate science special report: fourth national climate assessment, Volume I. Washington, DC, USA
Walsh J, Kovach AI, Babbitt KJ, O’Brien KM (2012) Fine-scale population structure and asymmetrical dispersal in an obligate salt-marsh passerine, the saltmarsh sparrow (Ammodramus caudacutus). Auk 129:247–258. https://doi.org/10.1525/auk.2012.11153
Weeks AR, Sgro CM, Young AG et al (2011) Assessing the benefits and risks of translocations in changing environments: A genetic perspective. Evol Appl 4:709–725. https://doi.org/10.1111/j.1752-4571.2011.00192.x
Whorley JR, Alvarez-Castañeda ST, Kenagy GJ (2004) Genetic structure of desert ground squirrels over a 20-degree-latitude transect from Oregon through the Baja California peninsula. Mol Ecol 13:2709–2720. https://doi.org/10.1111/j.1365-294X.2004.02257.x
Williams SJ (2013) Sea-level rise implications for coastal regions. J Coastal Res 63:184–196. https://doi.org/10.2112/SI63-015.1
Wood DA, Bui T-VD, Overton CT et al (2017) A century of landscape disturbance and urbanization of the San Francisco Bay region affects the present-day genetic diversity of the California Ridgway’s rail (Rallus obsoletus obsoletus). Conserv Genet 18:131–146. https://doi.org/10.1007/s10592-016-0888-4
Wright S (1931) Evolution in Mendelian populations. Genetics 16:97–159
Wright S (1978) Evolution and the Genetics of Populations, vol 4. University of Chicago Press, Chicago
Young A, Boyle T, Brown T (1996) The population genetic consequences of habitat fragmentation for plants. Trends Ecol Evol 11:413–418. https://doi.org/10.1016/0169-5347(96)10045-8
Zedler JB, Kercher S (2005) Wetland resources: status, trends, ecosystem services, and restorability. Annu Rev Environ Resour 30:39–74. https://doi.org/10.1146/annurev.energy.30.050504.144248
Zhao Q, Bai J, Huang L et al (2016) A review of methodologies and success indicators for coastal wetland restoration. Ecol Ind 60:442–452. https://doi.org/10.1016/j.ecolind.2015.07.003
Acknowledgements
K. Preston, B. Richardson and three anonymous reviewers provided valuable comments that improved this manuscript. We thank B. Richardson, T. Tobiasson and A. Boyd for processing the flow cytometry data, and all land managers for site access. This research was supported by the San Diego Association of Governments, NAVFAC SW, the USGS Ecosystems Mission Area and Western Ecological Research Center, and used resources provided by the Core Science Analytics, Synthesis, & Libraries (CSASL) Advanced Research Computing (ARC) group at the USGS. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Milano, E.R., Mulligan, M.R., Rebman, J.P. et al. High-throughput sequencing reveals distinct regional genetic structure among remaining populations of an endangered salt marsh plant in California. Conserv Genet 21, 547–559 (2020). https://doi.org/10.1007/s10592-020-01269-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-020-01269-3