Abstract
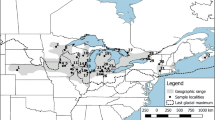
A genetic boundary at the Mississippi River, USA, has been suggested for the Northern Leopard Frog, Rana pipiens, which was recently proposed for listing as federally threatened in the western USA. This suggestion was made on the basis of limited geographic sampling of a mitochondrial gene. However, mitochondrial DNA represents a very small part of the genome and is not necessarily indicative of patterns in nuclear DNA. We tested the hypothesis that eastern and western populations are separated by a distinct genetic boundary by sequencing mitochondrial DNA more extensively across the range, including focused sampling in the zone of hypothetical introgression, and by analyzing four nuclear sequences and seven microsatellite loci. We confirmed previous results that eastern and western populations have unique mitochondrial sequences that are deeply divergent (3.8 %) and which overlap only in a narrow region around the Mississippi River. Nuclear sequences also show divergent eastern and western lineages in some cases but with a broader zone of geographic overlap. Microsatellite data correspond closely to mitochondrial data, differing between east and west and changing abruptly near the Mississippi River. These data collectively demonstrate that eastern and western clades of this species introgress considerably in some markers but are distinct and defined by clear and narrow boundaries in others. We demonstrate that the Mississippi River forms an important, albeit somewhat permeable, boundary between genetic lineages in this species. This genetic boundary coincides with previously described discontinuities in morphological features.











Similar content being viewed by others
References
Al-Rabab’ah MA, Williams CG (2002) Population dynamics of Pinus taeda L. based on nuclear microsatellites. For Ecol Manag 163:263–271
Austin JD, Lougheed SC, Boag PT (2004) Discordant temporal and geographic patterns in maternal lineages of eastern North American frogs, Rana catesbeiana (Ranidae) and Pseudacris crucifer (Hylidae). Mol Phylogenet Evol 32:799–816
Avise JC (2000) Phylogeography. Harvard University Press, Cambridge
Bonnet E, Van de Peer Y (2002) zt: a software tool for simple and partial Mantel tests. J Stat Softw 7:1–12
Bossuyt F, Milinkovitch MC (2000) Convergent adaptive radiations in Madagascan and Asian ranid frogs reveal covariation between larval and adult traits. Proc Natl Acad Sci USA 97:6585–6590
Brant SV, Ortí G (2003) Phylogeography of the Northern short-tailed shrew, Blarina brevicauda (Insectivora: Soricidae): past fragmentation and postglacial recolonization. Mol Ecol 12:1435–1449
Clarkson RW, Rorabaugh JC (1989) Status of leopard frogs (Rana pipiens complex: Ranidae) in Arizona and southeastern California. Southwest Nat 34:531–538
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1659
Cope ED (1889) The Batrachia of North America. Bull US Natl Mus 34:1–525
Di Candia MR, Routman EJ (2007) Cytonuclear discordance across a leopard frog contact zone. Mol Phylogenet Evol 45:564–575
Dunlap DG, Platz JE (1981) Geographic variation of proteins and call in Rana pipiens from the northcentral United States. Copeia 1981:876–879
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Fitzpatrick BM, Shaffer HB (2007) Hybrid vigor between native and introduced salamanders raises new challenges for conservation. Proc Natl Acad Sci USA 104:15793–15798
Flot J-F (2010) SEQPHASE: a web tool for interconverting PHASE input/output files and FASTA sequence alignments. Mol Ecol Resour 10:162–166
Fontenot BE, Makowsky R, Chippindale PT (2011) Nuclear-mitochondrial discordance and gene flow in a recent radiation of toads. Mol Phylogenet Evol 59:66–80
Frantz AC, Cellina S, Krier A, Schley L, Burke T (2009) Using spatial Bayesian methods to determine the genetic structure of a continuously distributed population: clusters or isolation by distance? J Appl Ecol 46:493–505
Frost JS, Bagnara JT (1976) A new species of leopard frog (Rana pipiens complex) from Northwestern Mexico. Copeia 1976:332–338
Germaine SS, Hays DW (2009) Distribution and postbreeding environmental relationships of Northern Leopard Frogs (Rana [Lithobates] pipiens) in Washington. West N Am Nat 69:537–547
Goldberg CS, Field KJ, Sredl MJ (2004) Mitochondrial DNA sequences do not support species status of the Ramsey Canyon Leopard Frog (Rana subaquavocalis). J Herpetol 38:313–319
Green DE (2001) Toe-clipping of frogs and toads. In: Standard operating procedure. United States Geological Survey, National Wildlife Health Center
Guillot G, Mortier F, Estoup A (2005) GENELAND: a computer package for landscape genetics. Mol Ecol Notes 5:712–715
Hekkala ER, Saumure RA, Jaeger JR, Herrmann H-W, Sredl MJ, Bradford DF, Drabeck D, Blum MJ (2011) Resurrecting an extinct species: archival DNA, taxonomy, and conservation of the Vegas Valley Leopard Frog. Cons Genet 12:1379–1385
Hillis DM, Wilcox TP (2005) Phylogeny of the New World true frogs (Rana). Mol Phylogenet Evol 34:299–314
Hoffman EA, Blouin MS (2004a) Evolutionary history of the Northern Leopard Frog: reconstruction of phylogeny, phylogeography, and historical changes in population demography from mitochondrial DNA. Evolution 58:145–159
Hoffman EA, Blouin MS (2004b) Historical data refute recent range contraction as a cause of low genetic diversity in isolated frog populations. Mol Ecol 13:271–276
Hoffman EA, Ardren WR, Blouin MS (2003) Nine polymorphic microsatellite loci for the Northern Leopard Frog (Rana pipiens). Mol Ecol Notes 3:115–116
Jacobsen F, Omland KE (2011) Species tree inference in a recent radiation of orioles (Genus Icterus): multiple markers and methods reveal cytonuclear discordance in the northern oriole group. Mol Phylogenet Evol 61:460–469
Jaeger JR, Riddle BR, Jennings RD, Bradford DF (2001) Rediscovering Rana onca: evidence for phylogenetically distinct leopard frogs from the border region of Nevada, Utah, and Arizona. Copeia 2001:339–354
Johnson KP, Clayton DH (2000) Nuclear and mitochondrial genes contain similar phylogenetic signal for pigeons and doves (Aves: Columbiformes). Mol Phylogenet Evol 14:141–151
Kauffeld CF (1937) The status of the leopard frogs, Rana brachycephala and Rana pipiens. Herpetologica 1:84–87
Kim J-B, Matsui M, Lee J-E, Min M-S, Suh J-H, Yang S-Y (2004) Notes on a discrepancy in mitochondrial DNA and allozyme differentiation in a pond frog Rana nigromaculata. Zool Sci 21:39–42
Larson KA (2004) Advertisement call complexity in Northern Leopard Frogs, Rana pipiens. Copeia 2004:676–682
McAllister KR (2005) Northern Leopard Frog. In: Jones LLC, Leonard WP, Olson DH (eds) Amphibians of the Pacific Northwest. Seattle Audubon Society, Seattle, pp 206–209
Miller MA, Pfeiffer W, Schwartz T (2010) Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In: Proceedings of the gateway computing environments workshop (GCE), pp 1-8, New Orleans
Moore JA (1944) Geographic variation in Rana pipiens Schreber of eastern North America. Bull Am Mus Nat Hist 82:345–369
Moriarty ME (2009) Endangered and threatened wildlife and plants; 90-day finding on a petition to list the Northern Leopard Frog (Lithobates [Rana] pipiens) in the western United States as threatened. Fed Regist 74:31389–31401
Moriarty EC, Cannatella DC (2004) Phylogenetic relationships of the North American chorus frogs (Pseudacris: Hylidae). Mol Phylogenet Evol 30:409–420
Müllenbach R, Lagoda PJL, Welter C (1989) An efficient salt–chloroform extraction of DNA from blood and tissues. Trends Genet 5:391
Oyler-McCance SJ, Kahn NW, Burnham KP, Braun CE, Quinn TW (1999) A population genetic comparison of large- and small-bodied sage grouse in Colorado using microsatellite and mitochondrial DNA markers. Mol Ecol 8:1457–1465
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Platz JE, Frost JS (1984) Rana yavapaiensis, a new species of leopard frog (Rana pipiens complex). Copeia 1984:940–948
Platz JE, Mecham JS (1979) Rana chiricahuensis, a new species of leopard frog (Rana pipiens complex) from Arizona. Copeia 1979:383–390
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Prychitko TM, Moore WS (1997) The utility of DNA sequences of an intron from the β-fibrinogen gene in phylogenetic analysis of woodpeckers (Aves: Picidae). Mol Phylogenet Evol 8:193–204
Qiu X, Arthington JD, Riley DG, Chase CC Jr, Phillips WA, Coleman SW, Olson TA (2007) Genetic effects on acute phase protein response to the stresses of weaning and transportation in beef calves. J Anim Sci 85:2367–2374
Sage RD, Atchley WR, Capanna E (1993) House mice as models in systematic biology. Syst Biol 42:523–561
Schueler FW (1973) Frogs of the Ontario coast of Hudson Bay and James Bay. Can Field Nat 87:409–418
Schueler FW (1982) Geographic variation in skin pigmentation and dermal glands in the Northern Leopard Frog, Rana pipiens. Publ Zool Natl Mus Nat Sci Natl Mus Can 16:1–80
Selander RK, Hunt WG, Yang SY (1969) Protein polymorphism and genic heterozygosity in two European subspecies of the house mouse. Evolution 23:379–390
Sites JW Jr, Marshall JC (2003) Delimiting species: a renaissance issue in systematic biology. Trends Ecol Evol 18:462–470
Sites JW Jr, Marshall JC (2004) Operational criteria for delimiting species. Annu Rev Ecol Evol Syst 35:199–227
Soltis DE, Morris AB, McLachlan JS, Manos PS, Soltis PS (2006) Comparative phylogeography of unglaciated eastern North America. Mol Ecol 15:4261–4293
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22:2688–2690
Stamatakis A, Hoover P, Rougemont J (2008) A rapid bootstrap algorithm for the RAxML web servers. Syst Biol 57:758–771
Stephens M, Donnelly P (2003) A comparison of Bayesian methods for haplotype reconstruction from population genotype data. Am J Hum Genet 73:1162–1169
Walker D, Moler PE, Buhlmann KA, Avise JC (1998) Phylogeographic patterns in Kinosternon subrubrum and K. baurii based on mitochondrial DNA restriction analyses. Herpetologica 54:174–184
Weibel AC, Moore WS (2002) A test of a mitochondrial gene-based phylogeny of woodpeckers (genus Picoides) using an independent nuclear gene, β-fibrinogen intron 7. Mol Phylogenet Evol 22:247–257
Werner JK (2003) Status of the Northern Leopard Frog (Rana pipiens) in western Montana. Northwest Nat 84:24–30
Wilson GA, Fulton TL, Kendell K, Scrimgeour G, Paszkowski CA, Coltman DW (2008) Genetic diversity and structure in Canadian Northern Leopard Frog (Rana pipiens) populations: implications for reintroduction programs. Can J Zool 86:863–874
Wright AH, Wright AA (1949) Handbook of frogs and toads of the United States and Canada, 3rd edn. Comstock, Ithaca
Yang D-S, Kenagy GJ (2009) Nuclear and mitochondrial DNA reveal contrasting evolutionary processes in populations of deer mice (Peromyscus maniculatus). Mol Ecol 18:5115–5125
Acknowledgments
C. Drost assisted in many ways including helping acquire financial support and samples. Funding was provided by the Utah State University Ecology Center, the United States Bureau of Reclamation, and the United States Geological Survey. For help collecting or acquiring samples, we thank D. Alonso, B. and B. Alsop, C. Avenengo, A. and R. Bekintis, B. Blake, K. Boyle, C. Bubac, M. Carrick, G. Casper, A. Durso, C. Friesen, M. French, L. Gelczis, A. Gooley, D. Guericke, K. Haxton, M. Hayes, S. Jones, K. Kendell, D. Kimberling, M. Koepsel, J. Massie, M. Mazza, D. McAlpine, K. and R. Moloney, D. Penttila, D. and D. Peterson, J. Poston, R. Quinn, J. Slawson, B. Smith, L. Smith, N. Smith, A. Spriggs, and K. and K. Taylor. For help with laboratory work, we thank J. Baker, S. Bardot, C. Callahan, J. P. Chong, M. Culumber, D. Fixsen, and C. Rowe. For comments on design, analysis, and writing, we thank K. Beard, M. Pfrender, and H. Rai. The protocols used here were approved by the Utah State University Institutional Animal Care and Use Committee (approvals 1138 and 1469) and Northern Arizona University IACUC (approval 07-003). New collections were conducted under the following permissions (some samples were donated from pre-existing collections): Colorado Division of Wildlife Scientific Collection License 08HP2004A001, Illinois Scientific Permits NH09.5256 and NH10.5256, Iowa Collectors Permit SC 896, Kentucky Educational Wildlife Collecting Permit SC1011073, Michigan Scientific Collector’s Permit 16627, Montana Scientific Collector’s Permit 2010-080, Minnesota Special Permits 15676 and 16627, Nebraska Game and Parks Commission Scientific and Educational Permits 951 and 980, New Brunswick Scientific Permits SP09-004 and SP10-011, Ontario Wildlife Scientific Collector’s Authorization 1051331, Ouray National Wildlife Refuge Special Use Permit 08-002, Pennsylvania Scientific Collector Permit 457, Quebec Permis Scientifiques 2009-04-14-106-10-S–F and 2010-04-29-124-10-S–F, Rhode Island Scientific Collector’s Permit 2009-38, South Dakota Scientific Collector’s Permit 58, Utah Division of Wildlife Certificates of Registration 1COLL7519 and 1COLL7723, Wisconsin Scientific Research Licenses SRL-SCR-003-2010, and Wyoming Game and Fish Department Chapter 33 Permit 681.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
O’Donnell, R.P., Mock, K.E. Two frog species or one? A multi-marker approach to assessing the distinctiveness of genetic lineages in the Northern Leopard Frog, Rana pipiens . Conserv Genet 13, 1167–1182 (2012). https://doi.org/10.1007/s10592-012-0384-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-012-0384-4