Abstract
Canine hemangiosarcoma is a highly aggressive vascular neoplasm associated with extensive clinical and anatomical heterogeneity and a grave prognosis. Comprehensive molecular characterization of hemangiosarcoma may identify novel therapeutic targets and advanced clinical management strategies, but there are no published reports of tumor-associated genome instability and disrupted gene dosage in this cancer. We performed genome-wide microarray-based somatic DNA copy number profiling of 75 primary intra-abdominal hemangiosarcomas from five popular dog breeds that are highly predisposed to this disease. The cohort exhibited limited global genomic instability, compared to other canine sarcomas studied to date, and DNA copy number aberrations (CNAs) were predominantly of low amplitude. Recurrent imbalances of several key cancer-associated genes were evident; however, the global penetrance of any single CNA was low and no distinct hallmark aberrations were evident. Copy number gains of dog chromosomes 13, 24, and 31, and loss of chromosome 16, were the most recurrent CNAs involving large chromosome regions, but their relative distribution within and between cases suggests they most likely represent passenger aberrations. CNAs involving CDKN2A, VEGFA, and the SKI oncogene were identified as potential driver aberrations of hemangiosarcoma development, highlighting potential targets for therapeutic modulation. CNA profiles were broadly conserved between the five breeds, although subregional variation was evident, including a near twofold lower incidence of VEGFA gain in Golden Retrievers versus other breeds (22 versus 40 %). These observations support prior transcriptional studies suggesting that the clinical heterogeneity of this cancer may reflect the existence of multiple, molecularly distinct subtypes of canine hemangiosarcoma.




Similar content being viewed by others
Abbreviations
- ASD:
-
Australian Shepherd Dog
- BAC:
-
bacterial artificial chromosome
- BMD:
-
Bernese Mountain Dog
- CFA:
-
Canis familiaris
- CNA:
-
copy number aberration
- CNV:
-
copy number variant
- FCR:
-
Flat-Coated Retriever
- FFPE:
-
formalin-fixed, paraffin-embedded
- FISH:
-
fluorescence in situ hybridization
- GR:
-
Golden Retriever
- GSD:
-
German Shepherd Dog
- H&E:
-
hematoxylin and eosin
- oaCGH:
-
oligonucleotide array comparative genomic hybridization
- SSC:
-
saline–sodium citrate
References
Agulnik M, Yarber JL, Okuno SH et al (2012) An open-label, multicenter, phase II study of bevacizumab for the treatment of angiosarcoma and epithelioid hemangioendotheliomas. Ann Oncol 24:257–263
American Kennel Club (2012) American Kennel Club 2012 Dog Registration Statistics (http://www.akc.org/reg/dogreg_stats.cfm, accessed November 24th 2013)
Andersen NJ, Nickoloff BJ, Dykema KJ et al (2013) Pharmacologic inhibition of MEK signaling prevents growth of canine hemangiosarcoma. Mol Cancer Ther 12:1701--1714
Angstadt AY, Motsinger-Reif A, Thomas R et al (2011) Characterization of canine osteosarcoma by array comparative genomic hybridization and RT-qPCR: signatures of genomic imbalance in canine osteosarcoma parallel the human counterpart. Genes Chromosomes Cancer 50:859–874
Angstadt AY, Thayanithy V, Subramanian S, Modiano JF, Breen M (2012) A genome-wide approach to comparative oncology: high-resolution oligonucleotide aCGH of canine and human osteosarcoma pinpoints shared microaberrations. Cancer Genet 205:572–587
Antonescu CR, Yoshida A, Guo T et al (2009) KDR activating mutations in human angiosarcomas are sensitive to specific kinase inhibitors. Cancer Res 69:7175–7179
Baumhoer D, Gunawan B, Becker H, Fuzesi L (2005) Comparative genomic hybridization in four angiosarcomas of the female breast. Gynecol Oncol 97:348–352
Becker SE, Thomas R, Trifonov VA et al (2011) Anchoring the dog to its relatives reveals new evolutionary breakpoints across 11 species of the Canidae and provides new clues for the role of B chromosomes. Chromosome Res 19:685–708
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Methodol 57:289–300
Bergman P (2010) Hemangiosarcoma. In: Ettinger S, Feldman E (eds) Textbook of veterinary internal medicine, 7th edn. Elsevier, St Louis, pp 2175–2180
Beroukhim R, Getz G, Nghiemphu L et al (2007) Assessing the significance of chromosomal aberrations in cancer: methodology and application to glioma. Proc Natl Acad Sci U S A 104:20007–20012
Billings SD, McKenney JK, Folpe AL, Hardacre MC, Weiss SW (2004) Cutaneous angiosarcoma following breast-conserving surgery and radiation: an analysis of 27 cases. Am J Surg Pathol 28:781–788
Bonnon C, Atanasoski S (2012) c-Ski in health and disease. Cell Tissue Res 347:51–64
Breen M, Hitte C, Lorentzen TD et al (2004) An integrated 4249 marker FISH/RH map of the canine genome. BMC Genomics 5:65
Brown LF, Dezube BJ, Tognazzi K, Dvorak HF, Yancopoulos GD (2000) Expression of Tie1, Tie2, and angiopoietins 1, 2, and 4 in Kaposi’s sarcoma and cutaneous angiosarcoma. Am J Pathol 156:2179–2183
Cascone T, Heymach JV (2012) Targeting the angiopoietin/Tie2 pathway: cutting tumor vessels with a double-edged sword? J Clin Oncol 30:441–444
Chen WK, Swartz JD, Rush LJ, Alvarez CE (2009) Mapping DNA structural variation in dogs. Genome Res 19:500–509
Club AK (2006) The complete dog book, 20th edn. Ballantine Books, New York
Dews M, Homayouni A, Yu D et al (2006) Augmentation of tumor angiogenesis by a Myc-activated microRNA cluster. Nat Genet 38:1060–1065
Dickerson EB, Thomas R, Fosmire SP et al (2005) Mutations of phosphatase and tensin homolog deleted from chromosome 10 in canine hemangiosarcoma. Vet Pathol 42:618–632
Dickerson EB, Marley K, Edris W et al (2013) Imatinib and Dasatinib inhibit hemangiosarcoma and implicate PDGFR-beta and Src in tumor growth. Transl Oncol 6:158–168
Fanciulli M, Petretto E, Aitman TJ (2010) Gene copy number variation and common human disease. Clin Genet 77:201–213
Fury MG, Antonescu CR, Van Zee KJ, Brennan MF, Maki RG (2005) A 14-year retrospective review of angiosarcoma: clinical characteristics, prognostic factors, and treatment outcomes with surgery and chemotherapy. Cancer J 11:241–247
Glickman L, Glickman N, Thorpe R (2000) The Golden Retriever Club of America National Health Survey http://www.grca.org/pdf/health/healthsurvey.pdf, accessed 24th November 2013). Golden Retriever Club of America
Guo T, Zhang L, Chang NE et al (2011) Consistent MYC and FLT4 gene amplification in radiation-induced angiosarcoma but not in other radiation-associated atypical vascular lesions. Genes Chromosomes Cancer 50:25–33
Hargis AM, Ihrke PJ, Spangler WL, Stannard AA (1992) A retrospective clinicopathologic study of 212 dogs with cutaneous hemangiomas and hemangiosarcomas. Vet Pathol 29:316–328
Hedan B, Thomas R, Motsinger-Reif A et al (2011) Molecular cytogenetic characterization of canine histiocytic sarcoma: a spontaneous model for human histiocytic cancer identifies deletion of tumor suppressor genes and highlights influence of genetic background on tumor behavior. BMC Cancer 11:201
Italiano A, Chen CL, Thomas R et al (2012a) Alterations of the p53 and PIK3CA/AKT/mTOR pathways in angiosarcomas: a pattern distinct from other sarcomas with complex genomics. Cancer 118:5878--5887
Italiano A, Thomas R, Breen M et al (2012b) The miR-17-92 cluster and its target THBS1 are differentially expressed in angiosarcomas dependent on MYC amplification. Genes Chromosomes Cancer 51:569–578
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics. CA Cancer J Clin 60:277–300
Karlsson E, Sigurdsson S, Ivansson E et al (2013) Genome-wide analyses implicate 33 loci in heritable dog osteosarcoma, including regulatory variants near CDKN2A/B. Genome Biol.14:R132
Krijgsman O, Israeli D, Haan JC et al (2012) CGH arrays compared for DNA isolated from formalin-fixed, paraffin-embedded material. Genes Chromosomes Cancer 51:344–352
Lahat G, Dhuka AR, Hallevi H et al (2010) Angiosarcoma: clinical and molecular insights. Ann Surg 251:1098–1106
Lindblad-Toh K, Wade CM, Mikkelsen TS et al (2005) Genome sequence, comparative analysis and haplotype structure of the domestic dog. Nature 438:803–819
Lucas DR (2009) Angiosarcoma, radiation-associated angiosarcoma, and atypical vascular lesion. Arch Pathol Lab Med 133:1804–1809
Manner J, Radlwimmer B, Hohenberger P et al (2010) MYC high level gene amplification is a distinctive feature of angiosarcomas after irradiation or chronic lymphedema. Am J Pathol 176:34–39
Mars Veterinary M (2011) Results of nation’s first ever mutt census reveal paw print Retrieved from http://www.marsveterinary.com/blogs/news/resultsofnationsfirstevermuttcensusrevealpawprint.html Accessed 30 Nov 2013
Nicholas TJ, Cheng Z, Ventura M et al (2009) The genomic architecture of segmental duplications and associated copy number variants in dogs. Genome Res 19:491–499
Roy P, Clark MA, Thomas JM (2004) Stewart-Treves syndrome—treatment and outcome in six patients from a single centre. Eur J Surg Oncol 30:982–986
Saharinen P, Eklund L, Pulkki K, Bono P, Alitalo K (2011) VEGF and angiopoietin signaling in tumor angiogenesis and metastasis. Trends Mol Med 17:347–362
Sarver AL, Phalak R, Thayanithy V, Subramanian S (2010) S-MED: sarcoma microRNA expression database. Lab Investig 90:753–761
Schultheiss PC (2004) A retrospective study of visceral and nonvisceral hemangiosarcoma and hemangiomas in domestic animals. J Vet Diagn Investig 16:522–526
Shearin AL, Ostrander EA (2010) Leading the way: canine models of genomics and disease. Dis Model Mech 3:27–34
Shearin AL, Hedan B, Cadieu E et al (2012) The MTAP-CDKN2A locus confers susceptibility to a naturally occurring canine cancer. Cancer Epidemiol Biomarkers Prev 21:1019–1027
Shlien A, Malkin D (2010) Copy number variations and cancer susceptibility. Curr Opin Oncol 22:55–63
Tamburini BA, Trapp S, Phang TL et al (2009) Gene expression profiles of sporadic canine hemangiosarcoma are uniquely associated with breed. PLoS One 4:e5549
Tamburini BA, Phang TL, Fosmire SP et al (2010) Gene expression profiling identifies inflammation and angiogenesis as distinguishing features of canine hemangiosarcoma. BMC Cancer 10:619
Taylor BS, Barretina J, Maki RG et al (2011) Advances in sarcoma genomics and new therapeutic targets. Nat Rev Cancer 11:541–557
Thamm D (2007) Hemangiosarcoma. In: Withrow S, Vail D (eds) Small animal clinical oncology, 4th edn. WB Saunders Elsevier, St Louis, pp 785–795
Thomas R, Duke SE, Wang HJ et al (2009) ‘Putting our heads together’: insights into genomic conservation between human and canine intracranial tumors. J Neurooncol 94:333–349
Thomas R, Seiser EL, Motsinger-Reif A et al (2011) Refining tumor-associated aneuploidy through ‘genomic recoding’ of recurrent DNA copy number aberrations in 150 canine non-Hodgkin lymphomas. Leuk Lymphoma 52:1321–1335
Thompson SL, Compton DA (2011) Chromosomes and cancer cells. Chromosome Res 19:433–444
TM O, Tan M, Tarango M et al (2009) Differential expression of SKI oncogene protein in hemangiomas. Otolaryngol Head Neck Surg 141:213–218
Tonomura N, Thomas R, Karlsson EK et al. Genome-wide association study identifies shared risk loci common to two malignancies in golden retrievers. Nat Commun. (submitted)
Vail DM, MacEwen EG (2000) Spontaneously occurring tumors of companion animals as models for human cancer. Cancer Invest 18:781–792
van Essen HF, Ylstra B (2012) High-resolution copy number profiling by array CGH using DNA isolated from formalin-fixed, paraffin-embedded tissues. Methods Mol Biol 838:329–341
Yonemaru K, Sakai H, Murakami M, Yanai T, Masegi T (2006) Expression of vascular endothelial growth factor, basic fibroblast growth factor, and their receptors (flt-1, flk-1, and flg-1) in canine vascular tumors. Vet Pathol 43:971–980
Yonemaru K, Sakai H, Murakami M et al (2007) The significance of p53 and retinoblastoma pathways in canine hemangiosarcoma. J Vet Med Sci 69:271–278
Acknowledgments
This study was supported by grant D10CA-501 (MB, JFM, KLT) from the Golden Retriever Foundation and Morris Animal Foundation, and grant 1131 from the American Kennel Club Canine Health Foundation (JFM). We thank Sandra Horton and staff members of the North Carolina State University Histopathology laboratory for their expertise in the preparation and diagnostic evaluation of clinical specimens. Additional pathology support was provided by Drs Gerry O’Sullivan and Sandra Yi, and the Comparative Pathology Shared Resource Core of the Masonic Cancer Center, University of Minnesota, supported by grant P30CA077598 from the National Institutes of Health. We thank Mitzi Lewellyn for co-ordination of clinical specimen collection, Katie Kennedy for assistance with FISH analysis and Kate Kelley, Alex Hanes Sparrow, Milcah Scott, and Megan Duckett for technical assistance. Enrollment and diagnostic evaluation of cases recruited by North Carolina State University was supported by the Canine Cancer Genomics Fund. Cases recruited through University of Minnesota were supported in part by the Starlight Fund, The Land of PureGold Foundation, the WillPower Fund, and other philanthropic funding at the University of Minnesota Animal Cancer Care and Research Program. We gratefully acknowledge the many dog owners, breeders, and veterinarians who have supported this study through provision of clinical specimens and patient data.
Ethical standards
Experiments described in this manuscript comply with the current laws of the country in which they were performed (USA). All institutional and national guidelines for the care and use of laboratory animals were followed.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Conly Rieder
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(XLS 309 kb)
Supplementary figure 1
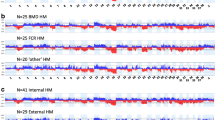
Comparison of oaCGH data obtained using DNA isolated from fresh and FFPE-derived tissue from the same tumor specimen. Examples are provided from two tumors for which the genomic profile shown in blue (1) was obtained using DNA isolated from fresh, unfixed tissue. The profile shown in red (2) was obtained using FFPE-derived tissue. These two profiles are then overlaid (3), showing that, although the data derived from FFPE tissue exhibit a higher level of background noise in oaCGH analysis, the regions of genomic balance identified are highly consistent with those derived from fresh, unfixed tissue from the same specimen (PPT 201 kb)
Rights and permissions
About this article
Cite this article
Thomas, R., Borst, L., Rotroff, D. et al. Genomic profiling reveals extensive heterogeneity in somatic DNA copy number aberrations of canine hemangiosarcoma. Chromosome Res 22, 305–319 (2014). https://doi.org/10.1007/s10577-014-9406-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-014-9406-z