Abstract
Purpose
Paget’s disease (PD) of the breast is an uncommon disease of the nipple usually accompanied by an underlying carcinoma, often HER2 + , and accounting for 0.5–5% of all breast cancer. To date, histogenesis of PD of the breast remains controversial, as two theories—transformation and epidermotropic—have been proposed to explain this disease. Currently, animal models recapitulating PD of the nipple have not been described.
Methods
HER2-enriched DT13 breast cancer cells were injected into the mammary fat pad of NOD scid gamma null (NSG) female mice. Immunohistochemical staining and pathological studies were performed on tumor samples, and diagnosis of PD of the nipple was confirmed by expression of proteins characteristic of Paget cells (epidermal growth factor 2 (HER2), androgen receptor (AR), cytokeratin 7 (CK7), cytokeratin 8/18 (CK8/18), and mucin 1 (MUC1)). In addition, DT13 cells grown in 2D culture and in soft agar assays were sensitive to in vitro treatment with pharmacological inhibitors targeting Her2, adenylyl cyclase, mTOR, and PI3K signaling pathways.
Results
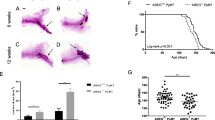
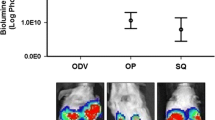
Mice developed tumors and nipple lesions that were detected exclusively on the tumor-bearing mammary fat pad. Tumor cells were positive for proteins characteristic of Paget cells. In vitro, DT13 cells were sensitive to inhibition of Her2, adenylyl cyclase, mTOR, and PI3K signaling pathways.
Conclusions
Our results suggest that injection of HER2 + DT13 cells into the mammary fat pad of NSG mice recapitulates critical aspects of the pathophysiology of PD of the nipple, supporting the epidermotropic theory as the more likely to explain the histogenesis of this disease.



Similar content being viewed by others
Abbreviations
- PD:
-
Paget’s disease
- NAC:
-
Nipple-areola complex
- HER2:
-
Human epidermal growth factor receptor 2
- ER:
-
Estrogen receptor
- PR:
-
Progesterone receptor
- AR:
-
Androgen receptor
- NSG:
-
NOD scid gamma null mice
References
Karakas C (2011) Paget’s disease of the breast. J Carcinog 10:31. https://doi.org/10.4103/1477-3163.90676
Lim HS, Jeong SJ, Lee JS, Park MH, Kim JW, Shin SS, Park JG, Kang HK (2011) Paget disease of the breast: mammographic, US, and MR imaging findings with pathologic correlation. Radiographics 31(7):1973–1987. https://doi.org/10.1148/rg.317115070
Sek P, Zawrocki A, Biernat W, Piekarski JH (2010) HER2 molecular subtype is a dominant subtype of mammary Paget’s cells. An immunohistochemical study. Histopathology 57(4):564–571. https://doi.org/10.1111/j.1365-2559.2010.03665.x
Fu W, Lobocki CA, Silberberg BK, Chelladurai M, Young SC (2001) Molecular markers in Paget disease of the breast. J Surg Oncol 77(3):171–178
Lester T, Wang J, Bourne P, Yang Q, Fu L, Tang P (2009) Different panels of markers should be used to predict mammary Paget’s disease associated with in situ or invasive ductal carcinoma of the breast. Ann Clin Lab Sci 39(1):17–24
Liegl B, Horn LC, Moinfar F (2005) Androgen receptors are frequently expressed in mammary and extramammary Paget’s disease. Mod Pathol 18(10):1283–1288. https://doi.org/10.1038/modpathol.3800437
Schelfhout VR, Coene ED, Delaey B, Thys S, Page DL, De Potter CR (2000) Pathogenesis of Paget’s disease: epidermal heregulin-alpha, motility factor, and the HER receptor family. J Natl Cancer Inst 92(8):622–628
Yamamoto T, Ikawa S, Akiyama T, Semba K, Nomura N, Miyajima N, Saito T, Toyoshima K (1986) Similarity of protein encoded by the human c-erb-B-2 gene to epidermal growth factor receptor. Nature 319(6050):230–234. https://doi.org/10.1038/319230a0
Dey N, De P, Leyland-Jones B (2017) PI3K-AKT-mTOR inhibitors in breast cancers: from tumor cell signaling to clinical trials. Pharmacol Ther 175:91–106. https://doi.org/10.1016/j.pharmthera.2017.02.037
Goka ET, Lippman ME (2015) Loss of the E3 ubiquitin ligase HACE1 results in enhanced Rac1 signaling contributing to breast cancer progression. Oncogene 34(42):5395–5405. https://doi.org/10.1038/onc.2014.468
Loibl S, Gianni L (2017) HER2-positive breast cancer. Lancet 389(10087):2415–2429. https://doi.org/10.1016/S0140-6736(16)32417-5
Ursini-Siegel J, Schade B, Cardiff RD, Muller WJ (2007) Insights from transgenic mouse models of ERBB2-induced breast cancer. Nat Rev Cancer 7(5):389–397. https://doi.org/10.1038/nrc2127
Drews-Elger K, Brinkman JA, Miller P, Shah SH, Harrell JC, da Silva TG, Ao Z, Schlater A, Azzam DJ, Diehl K, Thomas D, Slingerland JM, Perou CM, Lippman ME, El-Ashry D (2014) Primary breast tumor-derived cellular models: characterization of tumorigenic, metastatic, and cancer-associated fibroblasts in dissociated tumor (DT) cultures. Breast Cancer Res Treat. https://doi.org/10.1007/s10549-014-2887-9
Iorns E, Drews-Elger K, Ward TM, Dean S, Clarke J, Berry D, El Ashry D, Lippman M (2012) A new mouse model for the study of human breast cancer metastasis. PLoS ONE 7(10):e47995. https://doi.org/10.1371/journal.pone.0047995
Nanni P, Nicoletti G, Palladini A, Croci S, Murgo A, Ianzano ML, Grosso V, Stivani V, Antognoli A, Lamolinara A, Landuzzi L, di Tomaso E, Iezzi M, De Giovanni C, Lollini PL (2012) Multiorgan metastasis of human HER-2 + breast cancer in Rag2-/-;Il2rg-/- mice and treatment with PI3K inhibitor. PLoS ONE 7(6):e39626. https://doi.org/10.1371/journal.pone.0039626
Saci A, Cantley LC, Carpenter CL (2011) Rac1 regulates the activity of mTORC1 and mTORC2 and controls cellular size. Mol Cell 42(1):50–61. https://doi.org/10.1016/j.molcel.2011.03.017
Campa CC, Ciraolo E, Ghigo A, Germena G, Hirsch E (2015) Crossroads of PI3K and Rac pathways. Small GTPases 6(2):71–80. https://doi.org/10.4161/21541248.2014.989789
Jegg AM, Ward TM, Iorns E, Hoe N, Zhou J, Liu X, Singh S, Landgraf R, Pegram MD (2012) PI3K independent activation of mTORC1 as a target in lapatinib-resistant ERBB2 + breast cancer cells. Breast Cancer Res Treat 136(3):683–692. https://doi.org/10.1007/s10549-012-2252-9
Funding
This research was supported by NIH Grant 1R01 CA113674 to DEA, Award Number T32CA119929 from the National Cancer Institute supported KDE, and Breast Cancer Research Foundation (BCRF) award to MEL.
Author information
Authors and Affiliations
Contributions
KDE: designed the study, collected and analyzed the data, and wrote the manuscript; ACSL: contributed to data analysis and writing of the manuscript; AMJ: contributed to data collection and analysis; ABE, CRGF and MEL carried out all diagnoses and pathological interpretations. CRGF, PM, DEA and MEL read and critically revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All animal studies were performed according to a protocol approved by the Institutional Animal Care & Use Committee (IACUC) at the University of Miami.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Drews-Elger, K., Sandoval-Leon, A.C., Ergonul, A.B. et al. Paget’s disease of the nipple in a Her2-positive breast cancer xenograft model. Breast Cancer Res Treat 179, 577–584 (2020). https://doi.org/10.1007/s10549-019-05490-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05490-8