Abstract
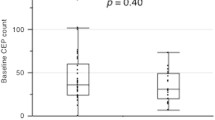
Animal models have demonstrated the critical role of bone marrow-derived VEGFR1+ hematopoietic progenitor cells (HPCs) and VEGFR2+ endothelial progenitor cells (EPCs) in metastatic progression. We explored whether these cells could predict relapse and response in breast cancer (BC) patients. One hundred and thirty-two patients with stages 1–4 BC were enrolled on 2 studies. Circulating CD45+/CD34+/VEGFR1+ HPCs and CD45dim/CD133+/VEGFR2+ EPCs were assessed from peripheral blood mononuclear cells using flow cytometry. Changes in HPCs and EPCs were analyzed in (1) patients without overt disease that relapsed and (2) metastatic patients according to response by RECIST. At study entry, 102 patients were without evidence of disease and 30 patients had metastatic BC. Seven patients without evidence of BC by exam, labs, and imaging developed recurrence while on study. Median HPC/ml (range) increased from 645.8 (23.5–1,914) to 2,899 (1,176–37,336), P = 0.016, followed by an increase in median EPC/ml from 21.3 (4.7–42.5) to 94.7 (28.2–201.3), P = 0.016, prior to clinical relapse. In metastatic patients with progressive disease, median HPC/ml increased from 1,696 (10–16,470) to 5,124 (374–77,605), P = 0.0009, and median EPC/ml increased from 26 (0–560) to 71 (0–615) prior to progression, P = 0.10. In patients with responding disease, median HPC/ml decreased from 6,147 (912–85,070) to 633 (47–18,065), P = 0.05, and EPC/ml decreased from 46 (0–197) to 23 (0–105), P = 0.41, at response. There were no significant changes in these cells over time in patients with stable disease. Circulating bone marrow-derived HPCs and EPCs predict relapse and disease progression in BC patients.




Similar content being viewed by others
References
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA Cancer J Clin 60:277–300
Brewster AM, Hortobagyi GN, Broglio KR et al (2008) Residual risk of breast cancer recurrence 5 years after adjuvant therapy. J Natl Cancer Inst 100:1179–1183
Chiang AC, Massague J (2008) Molecular basis of metastasis. N Engl J Med 359:2814–2823
Saphner T, Tormey DC, Gray R (1996) Annual hazard rates of recurrence for breast cancer after primary therapy. J Clin Oncol 14:2738–2746
Perou CM, Sorlie T, Eisen MB et al (2000) Molecular portraits of human breast tumours. Nature 406:747–752
Sorlie T, Perou CM, Tibshirani R et al (2001) Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98:10869–10874
Sorlie T, Tibshirani R, Parker J et al (2003) Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA 100:8418–8423
Cheang MC, van de Rijn M, Nielsen TO (2008) Gene expression profiling of breast cancer. Annu Rev Pathol 3:67–97
Harris L, Fritsche H, Mennel R et al (2007) American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol 25:5287–5312
Folkman J (1971) Tumor angiogenesis: therapeutic implications. N Engl J Med 285:1182–1186
Carmeliet P, Jain RK (2000) Angiogenesis in cancer and other diseases. Nature 407:249–257
Lyden D, Hattori K, Dias S et al (2001) Impaired recruitment of bone marrow derived endothelial and a hematopoietic precursor cells blocks tumor angiogenesis and growth. Nat Med 7:1194–1201
Kaplan RN, Riba RD, Zacharoulis S et al (2005) VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438:820–827
Gao D, Nolan DJ, Mellick AS, Bambino K, McDonnell K, Mittal V (2008) Endothelial progenitor cells control the angiogenic switch in mouse lung metastasis. Science 319:195–198
Fujita K, Nakayama M, Nakai Y et al (2009) Vascular endothelial growth factor receptor 1 expression in pelvic lymph nodes predicts the risk of cancer progression after radical prostatectomy. Cancer Sci 100:1047–1050
Taylor M, Rossler J, Geoerger B et al (2009) High levels of circulating VEGFR2+ Bone marrow-derived progenitor cells correlate with metastatic disease in patients with pediatric solid malignancies. Clin Cancer Res 15:4561–4571
Gao D, Mittal V (2009) The role of bone-marrow-derived cells in tumor growth, metastasis initiation and progression. Trends Mol Med 15:333–343
Roodhart JM, Langenberg MH, Daenen LG, Voest EE (2009) Translating preclinical findings of (endothelial) progenitor cell mobilization into the clinic; from bedside to bench and back. Biochim Biophys Acta 1796:41–49
Dome B, Timar J, Dobos J et al (2006) Identification and clinical significance of circulating endothelial progenitor cells in human non-small cell lung cancer. Cancer Res 66:7341–7347
Furstenberger G, von Moos R, Lucas R et al (2006) Circulating endothelial cells and angiogenic serum factors during neoadjuvant chemotherapy of primary breast cancer. Br J Cancer 94:524–531
Massa M, Rosti V, Ramajoli I et al (2005) Circulating CD34+, CD133+, and vascular endothelial growth factor receptor 2-positive endothelial progenitor cells in myelofibrosis with myeloid metaplasia. J Clin Oncol 23:5688–5695
Richter-Ehrenstein C, Rentzsch J, Runkel S, Schneider A, Schonfelder G (2007) Endothelial progenitor cells in breast cancer patients. Breast Cancer Res Treat 106:343–349
Naik RP, Jin D, Chuang E et al (2008) Circulating endothelial progenitor cells correlate to stage in patients with invasive breast cancer. Breast Cancer Res Treat 107:133–138
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247
Bertolini F, Shaked Y, Mancuso P, Kerbel RS (2006) The multifaceted circulating endothelial cell in cancer: towards marker and target identification. Nat Rev Cancer 6:835–845
Mancuso P, Antoniotti P, Quarna J et al (2009) Validation of a standardized method for enumerating circulating endothelial cells and progenitors: flow cytometry and molecular and ultrastructural analyses. Clin Cancer Res 15:267–273
Shaked Y, Henke E, Roodhart JM et al (2008) Rapid chemotherapy-induced acute endothelial progenitor cell mobilization: implications for antiangiogenic drugs as chemosensitizing agents. Cancer Cell 14:263–273
Farace F, Massard C, Borghi E, Bidart JM, Soria JC (2007) Vascular disrupting therapy-induced mobilization of circulating endothelial progenitor cells. Ann Oncol 18:1421–1422
Khan SS, Solomon MA, McCoy JP Jr (2005) Detection of circulating endothelial cells and endothelial progenitor cells by flow cytometry. Cytometry B Clin Cytom 64:1–8
Bahlmann FH, De Groot K, Spandau JM et al (2004) Erythropoietin regulates endothelial progenitor cells. Blood 103:921–926
Friedrich EB, Werner C, Walenta K, Bohm M, Scheller B (2009) Role of extracellular signal-regulated kinase for endothelial progenitor cell dysfunction in coronary artery disease. Basic Res Cardiol 104:613–620
Goss GD, Arnold A, Shepherd FA et al (2010) Randomized, double-blind trial of carboplatin and paclitaxel with either daily oral cediranib or placebo in advanced non-small-cell lung cancer: NCIC clinical trials group BR24 study. J Clin Oncol 28:49–55
Spratlin JL, Cohen RB, Eadens M et al (2010) Phase I pharmacologic and biologic study of ramucirumab (IMC-1121B), a fully human immunoglobulin G1 monoclonal antibody targeting the vascular endothelial growth factor receptor-2. J Clin Oncol 28:780–787
Schwartz JD, Rowinsky EK, Youssoufian H, Pytowski B, Wu Y (2010) Vascular endothelial growth factor receptor-1 in human cancer: concise review and rationale for development of IMC-18F1 (Human antibody targeting vascular endothelial growth factor receptor-1). Cancer 116:1027–1032
Jain S, Ward M, Chuang E, Cigler T, Moore A, Donovan D et al (2010) The effect of tetrathiomolybdate (TM) on circulating endothelial progenitor cells in women at moderate to high risk of breast cancer recurrence. Proc Breast Cancer Symp; abstr 297
Acknowledgments
We greatly appreciate the generous support from the following: Anne Moore Breast Cancer Research Fund, Stephen and Madeline Anbinder Foundation, Rozaliya Kosmandel Research Fund, Susan G Komen for the Cure, New York Community Trust, and Cancer Research and Treatment Fund. We would also like to thank the Weill Cornell Translational Core Laboratory, Rakhi Naik, MD for her initial contribution to the design of Study 1, and Dingchen Gao and Raul Catena for their valuable comments on this manuscript. Dr. Paul Christos was partially supported by the following grant: Clinical Translational Science Center (CTSC) (UL1-RR024996).
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jain, S., Ward, M.M., O’Loughlin, J. et al. Incremental increase in VEGFR1+ hematopoietic progenitor cells and VEGFR2+ endothelial progenitor cells predicts relapse and lack of tumor response in breast cancer patients. Breast Cancer Res Treat 132, 235–242 (2012). https://doi.org/10.1007/s10549-011-1906-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-011-1906-3