Abstract
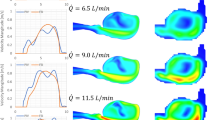
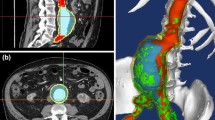
Hemodynamic conditions are hypothesized to affect the initiation, growth, and rupture of abdominal aortic aneurysms (AAAs), a vascular disease characterized by progressive wall degradation and enlargement of the abdominal aorta. This study aims to use magnetic resonance imaging (MRI) and computational fluid dynamics (CFD) to quantify flow stagnation and recirculation in eight AAAs by computing particle residence time (PRT). Specifically, we used gadolinium-enhanced MR angiography to obtain images of the vessel lumens, which were used to generate subject-specific models. We also used phase-contrast MRI to measure blood flow at supraceliac and infrarenal locations to prescribe physiologic boundary conditions. CFD was used to simulate pulsatile flow, and PRT, particle residence index, and particle half-life of PRT in the aneurysms were computed. We observed significant regional differences of PRT in the aneurysms with localized patterns that differed depending on aneurysm geometry and infrarenal flow. A bulbous aneurysm with the lowest mean infrarenal flow demonstrated the slowest particle clearance. In addition, improvements in particle clearance were observed with increase of mean infrarenal flow. We postulate that augmentation of mean infrarenal flow during exercise may reduce chronic flow stasis that may influence mural thrombus burden, degradation of the vessel wall, and aneurysm growth.











Similar content being viewed by others
Abbreviations
- AAA:
-
Abdominal aortic aneurysm
- AP:
-
Anterior to posterior
- IR:
-
Infrarenal
- LR:
-
Left to right
- MRI:
-
Magnetic resonance imaging
- PRI:
-
Particle residence index
- PRT:
-
Particle residence time
- RCR:
-
Resistance (proximal)–Capacitance–Resistance (distal)
- SC:
-
Supraceliac
References
Bluestein, D., L. Niu, R. T. Schoephoerster, and M. K. Dewanjee. Steady flow in an aneurysm model: correlation between fluid dynamics and blood platelet deposition. J. Biomech. Eng. 118:280–286, 1996.
Bluestein, D., L. Niu, R. T. Schoephoerster, and M. K. Dewanjee. Fluid mechanics of arterial stenosis: relationship to the development of mural thrombus. Ann. Biomed. Eng. 25(2):344–356, 1997.
Butty, V. D., K. Gudjonsson, P. Buchel, V. B. Makhijani, Y. Ventikos, and D. Poulikakos. Residence times and basins of attraction for a realistic right internal carotid artery with two aneurysms. Biorheology 29:387–393, 2002.
Cao, J., and S. E. Rittgers. Particle motion within in vitro models of stenosed internal carotid and left anterior descending coronary arteries. Ann. Biomed. Eng. 26:190–199, 1998.
Cheng, C. P., R. J. Herfkens, and C. A. Taylor. Abdominal aortic hemodynamic conditions in healthy subjects aged 50–70 at rest and during lower limb exercise: in vivo quantification using MRI. Atherosclerosis 168:323–331, 2003.
Dalman, R. L., M. M. Tedesco, J. Myers, and C. A. Taylor. AAA disease: mechanism, stratification, and treatment. Ann. N. Y. Acad. Sci. 1085:92–109, 2006.
Egelhoff, C. J., R. S. Budwig, D. F. Elger, T. A. Khraishi, and K. H. Johansen. Model studies of the flow in abdominal aortic aneurysms during resting and exercise conditions. J. Biomech. 32:1319–1329, 1999.
Fallon, A. M., U. M. Marzec, S. R. Hanson, and A. P. Yoganathan. Thrombin formation in vitro in response to shear-induced activation of platelets. Thromb. Res. 121(3):397–406, 2007.
Fillinger, M. F., J. Racusin, R. K. Baker, J. L. Cronenwett, A. Teutelink, M. L. Schermerhorn, R. M. Zwolak, R. J. Powell, D. B. Walsh, and E. M. Rzucidlo. Anatomic characteristics of ruptured abdominal aortic aneurysm on conventional CT scans: implications for rupture risk. J. Vasc. Surg. 39:1243–1252, 2004.
Finol, E. A., K. Keyhani, and C. H. Amon. The effect of asymmetry in abdominal aortic aneurysms under physiologically realistic pulsatile flow conditions. J. Biomech. Eng. 125:207–217, 2003.
Fogelson, A. L. Continuum models of platelet aggregation: formulation and mechanical properties. SIAM J. Appl. Math. 52:1089–1110, 1992.
Folie, B. J., and L. V. McIntire. Mathematical analysis of mural thrombogenesis: concentration profiles of platelet-activating agents and effects of viscous shear flow. Biophys. J. 56:1121–1141, 1989.
Fontaine, V., M. P. Jacob, X. Houard, P. Rossignol, D. Plissonnier, E. Angles-Cano, and J. B. Michel. Involvement of the mural thrombus as a site of protease release and activation in human aortic aneurysms. Am. J. Pathol. 161:1701–1710, 2002.
Gear, A. R. L. Platelet adhesion, shape change, and aggregation: rapid initiation and signal transduction events. Can. J. Physiol. Pharmacol. 72:285–294, 1994.
Gillum, R. F. Epidemiology of aortic aneurysm in the United States. J. Clin. Epidemiol. 48:1289–1298, 1995.
Glagov, S., C. K. Zarins, D. G. Giddens, and D. N. Ku. Hemodynamics and atherosclerosis. Insights and perspectives gained from studies of human arteries. Arch. Pathol. Lab. Med. 112:1018–1031, 1988.
Golledge, J., P. S. Tsao, R. L. Dalman, and P. E. Norman. Circulating markers of abdominal aortic aneurysm presence and progression. Circulation 118:2382–2392, 2008.
Jesty, J., W. Yin, P. Perrotta, and D. Bluestein. Platelet activation in a circulating flow loop: combined effects of shear stress and exposure time. Platelets 14(3):143–149, 2003.
Karino, T., and H. L. Goldsmith. Aggregation of human platelets in an annular vortex distal to a tubular expansion. Microvasc. Res. 17:217–237, 1979.
Karino, T., and H. L. Goldsmith. Adhesion of human platelets to collagen on the walls distal to a tubular expansion. Microvasc. Res. 17:238–262, 1979.
Khanafer, K. M., P. Gadhoke, R. Berguer, and J. L. Bull. Modeling pulsatile flow in aortic aneurysms: effect of non-Newtonian properties of blood. Biorheology 43:661–679, 2006.
Kim, H. J., C. A. Figueroa, T. J. Hughes, K. C. Jansen, and C. A. Taylor. Augmented lagrangian method for constraining the shape of velocity profiles at outlet boundaries for three-dimensional finite element simulations of blood flow. Comput. Methods Appl. Mech. Eng. 198:3551–3566, 2009.
Kunov, M. J., D. A. Steinman, and C. R. Ethier. Particle volumetric residence time calculation in arterial geometries. J. Biomech. Eng. 118:158–164, 1996.
Les, A. S., S. C. Shadden, C. A. Figueroa, J. M. Park, M. M. Tedesco, R. J. Herfkens, R. L. Dalman, and C. A. Taylor. Quantification of hemodynamics in abdominal aortic aneurysms during rest and exercise using magnetic resonance imaging and computational fluid dynamics. Ann. Biomed. Eng. 38:1288–1313, 2010.
Les, A. S., J. J. Yeung, G. M. Schultz, R. J. Herfkens, R. L. Dalman, and C. A. Taylor. Supraceliac and infrarenal aortic flow in patients with abdominal aortic aneurysms: mean flows, waveforms, and allometric scaling relationships. Cardiovasc. Eng. Technol. 1:39–51, 2010.
Leuprecht, A., and K. Perktold. Computer simulation of non-Newtonian effects on blood flow in large arteries. Comput. Methods Biomech. Biomed. Eng. 4:149–163, 2001.
McPhee, J. T., J. S. Hill, and M. H. Eslami. The impact of gender on presentation, therapy and mortality of abdominal aortic aneurysm in the United States, 2001–2004. J. Vasc. Surg. 45:891–899, 2007.
Moore, Jr., J. E., and D. N. Ku. Pulsatile velocity measurements in a model of the human abdominal aorta under resting conditions. J. Biomech. Eng. 116:337–346, 1994.
Perktold, K. On the paths of fluid particles in an axisymmetrical aneurysm. J. Biomech. 20:311–317, 1987.
Raines, J. K., M. Y. Jaffrin, and A. H. Shapiro. A computer simulation of arterial dynamics in the human leg. J. Biomech. 7:77–91, 1974.
Satta, J., E. Laara, and T. Juvonen. Intraluminal thrombus predicts rupture of an abdominal aortic aneurysm. J. Vasc. Surg. 23:737–739, 1996.
Spilker, R. L., and C. A. Taylor. Tuning multiscale hemodynamic simulations to match physiological measurements. Ann. Biomed. Eng. 38:2635–2648, 2010.
Tang, B. T., C. P. Cheng, M. T. Draney, N. M. Wilson, P. S. Tsao, R. J. Herfkens, and C. A. Taylor. Abdominal aortic hemodynamics in young healthy adults at rest and during lower limb exercise: quantification using image-based computer modeling. Am. J. Physiol. Heart Circ. Physiol. 291:H668–H676, 2006.
Taylor, C. A., and D. A. Steinman. Image-based modeling of blood flow and vessel wall dynamics: applications, methods and future directions. Ann. Biomed. Eng. 38:1188–1203, 2010.
Taylor, C. A., T. J. R. Hughes, and C. K. Zarins. Effect of exercise on hemodynamic conditions in the abdominal aorta. J. Vasc. Surg. 29:1077–1089, 1999.
Taylor, C. A., T. J. R. Hughes, and C. K. Zarins. Finite element modeling of blood flow in arteries. Comput. Methods Appl. Mech. Eng. 158:155–196, 1998.
Tenforde, A. S., C. P. Cheng, G. Suh, R. J. Herfkens, R. L. Dalman, and C. A. Taylor. Quantifying in vivo hemodynamic response to exercise in patients with intermittent claudication and abdominal aortic aneurysms using cine phase-contrast MRI. J. Magn. Reson. Imaging 31:425–429, 2010.
Vignon-Clementel, I. E., C. A. Figueroa, K. E. Jensen, and C. A. Taylor. Outflow boundary conditions for three-dimensional finite element modeling of blood flow and pressure in arteries. Comput. Methods Appl. Mech. Eng. 195:3776–3796, 2006.
Vignon-Clementel, I. E., C. A. Figueroa, K. E. Jensen, and C. A. Taylor. Outflow boundary conditions for three-dimensional simulations of non-periodic blood flow and pressure fields in deformable arteries. Comput. Meth. Biomech. Eng., 2010. doi:10.1080/10255840903413565.
Vollmar, J. F., E. Paes, P. Pauschinger, E. Hense, and A. Friesch. Aortic aneurysms as late sequelae of above-knee amputation. Lancet 2:834–835, 1989.
Whiting, C. H., and K. C. Jansen. A stabilized finite element method for the incompressible Navier-Stokes equations using a hierarchical basis. Int. J. Numer. Methods Fluid 35:93–116, 2001.
Wilson, N., K. Wang, R. W. Dutton, and C. A. Taylor. A software framework for creating patient specific geometric models from medical imaging data for simulation based medical planning of vascular surgery. Lect. Notes Comput. Sci. 2208:449–456, 2001.
Wolf, Y. G., W. S. Thomas, F. J. Brennan, W. G. Goff, M. J. Sise, and E. F. Bernstein. Computed tomography scanning findings associated with rapid expansion of abdominal aortic aneurysms. J. Vasc. Surg. 20:529–535, 1994.
Womersley, J. R. Method for the calculation of velocity, rate of flow and viscous drag in arteries when the pressure gradient in known. J. Physiol. 127:553–563, 1955.
Yeung, J. J., H. J. Kim, T. A. Abbruzzese, I. E. Vignon-Clementel, M. T. Draney-Blomme, K. K. Yeung, I. Perkash, R. J. Herfkens, C. A. Taylor, and R. J. Dalman. Aortoiliac hemodynamics and morphologic adaptation to chronic spinal cord injury. J. Vasc. Surg. 44:1254–1265, 2006.
Acknowledgments
This research was supported by the National Institutes of Health (P50 HL083800, P41 RR09784), the Lucas Center for Magnetic Resonance Imaging, and NSF (CNS-0619926) for computer resources. Allen Chiou, Victoria Yeh, Yash Narang, and Bartlomiej R. Imielski provided assistance with imaging and modeling. Nan Xiao provided help with quantification of PRT data. We thank all research subjects for their participation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Aleksander S. Popel oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Suh, GY., Les, A.S., Tenforde, A.S. et al. Quantification of Particle Residence Time in Abdominal Aortic Aneurysms Using Magnetic Resonance Imaging and Computational Fluid Dynamics. Ann Biomed Eng 39, 864–883 (2011). https://doi.org/10.1007/s10439-010-0202-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-010-0202-4