Abstract
Land-use change, a major constituent of global environmental change, potentially has significant consequences for human health in relation to mosquito-borne diseases. Land-use change can influence mosquito habitat, and therefore the distribution and abundance of vectors, and land use mediates human–mosquito interactions, including biting rate. Based on a conceptual model linking the landscape, people, and mosquitoes, this interdisciplinary study focused on the impacts of changes in land use on dengue and malaria vectors and dengue transmission in northern Thailand. Extensive data on mosquito presence and abundance, land-use change, and infection risk determinants were collected over 3 years. The results of the different components of the study were then integrated through a set of equations linking land use to disease via mosquito abundance. The impacts of a number of plausible scenarios for future land-use changes in the region, and of concomitant behavioral change were assessed. Results indicated that land-use changes have a detectable impact on mosquito populations and on infection. This impact varies according to the local environment but can be counteracted by adoption of preventive measures.
Similar content being viewed by others
Introduction
Large areas of the earth’s surface are being modified by human activities, constituting an important component of global environmental change. The associated land-use changes have been related to emerging and reemerging diseases (Patz et al., 2004), among multiple, complex factors operating at a range of temporal and spatial scales (Wilcox and Colwell, 2005). Environmental factors are of prime importance to the transmission of vector-borne diseases and include those associated with the host or the vector. The objective of this interdisciplinary study was to investigate empirically the impact of land-use change on populations of mosquito vectors of dengue and malaria, and on transmission of dengue in northern Thailand. A conceptual model linking landscape, people, and mosquitoes was first elaborated. Results of a series of empirical studies based on extensive data collection were integrated by a set of equations. Scenarios were then developed based on possible future changes in land use and/or human behavior, and quantified using the integrated model.
The complexity of vector-borne disease transmission has long been recognized and, with it, the need for integrating various factors in models to improve understanding of the system. The most widely known models of disease transmission by mosquitoes to humans are based on the Basic Reproductive Rate (R 0 ) and the Entomological Inoculation Rate (Rogers, 1988; Anderson and May, 1991; Snow and Gilles, 2002). A key variable in these models is the density of vectors (all vector species included) in relation to humans. Abundance and diversity of vector habitats, as provided by the local environment, and particularly for the immature stages, will promote high vector densities. The behavior and spatial and temporal distributions of human and vector populations are heterogeneous. Even though remote sensing allows describing landscape heterogeneity at a scale that is relevant for insect vectors (Tran and Raffy, 2006), many models do not represent the effects of land cover on vectors, nor do they represent land use as an indicator of human activities and hence of human presence near vector habitats. Most environmental factors currently included in models relate to meteorological or climatic conditions (Focks et al., 1993, 1995).
Conceptual Model
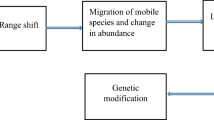
We first developed a conceptual model representing interactions between people (as agents of land-use change and disease hosts), the landscape (as being used by people for their livelihood as well as providing habitats for mosquitoes), and mosquitoes (as disease vectors) (Fig. 1).
People and Landscape
The natural, cultural, and economic environments combine to create conditions to which people in general, and land users in particular, respond and adapt by modifying their land use, e.g., their farming practice (Lambin et al., 2001, 2003). Land use also determines the location and movements of people in the landscape at certain times of the day and of the year. The location of certain activities such as farming may be close to areas with high densities of mosquito breeding sites, at times of day or during seasons that could correspond with mosquito biting peaks.
Landscape and Mosquitoes
The spatial distribution of vector-borne diseases is restricted typically by the geographical range of the vector or reservoir host and by their habitat preferences (Kitron, 1998). The immature stages of mosquitoes depend on freestanding water habitats for their survival and development (Service and Townson, 2002). These habitats include a variety of natural and artificial containers and bodies of surface water. Land-use change could allow the colonization of new habitats or the extension or reduction of a vector’s range, but could also modify the composition of the mosquito vector community, because vector species differ in their habitat preferences for the immature stages (Patz and Norris, 2004).
Mosquitoes and People
The exposure to mosquito-borne diseases depends on the prevalence of infection and on the exposure of people to biting mosquitoes. Any factor contributing to increased mosquito populations, mosquito longevity, and closer contact between humans and mosquito vectors can influence transmission dynamics. Conversely, most preventive and control measures related to the vector aim to reduce the mosquito population, the number of contacts between mosquitoes and humans, or the period when humans are infective, for example by rapid detection and treatment of disease. Social and behavioral patterns of humans have thus a significant impact on the epidemiology of mosquito-borne diseases (Fungladda and Butraporn, 1992; WHO, 1985).
Study Area
People–mosquito–landscape interactions were analyzed through extensive data collection over 3 years in seven villages.
In northern Thailand, natural forests are dominated by dry dipterocarp forests and, on moister sites, mixed deciduous forests (Schmidt-Vogt, 1999). Montane forests are found at high altitudes. Lowland farmers usually cultivate irrigated plots in the fertile valley bottoms (from one to three crops a year, including a dry-season crop other than rice) and upland plots of field or tree crops. Upland swidden (slash-and-burn) farmers grow upland rice as well as a variety of vegetables. Three study villages were located in Mae Hong Son province (Fig. 2): Ban Nong Khao Klang, a highland village with rotational swiddening; Ban Huai Pong Kha Nai, and Ban Huai Chang Kham, with irrigated farming and some upland fields. Three villages were included in Chiang Mai province: Ban Pa Nai, located in a wide irrigated valley with intensive irrigated farming supplemented by orchards; Ban Hueng Ngu, located in a narrower irrigated valley surrounded by large areas of fruit orchards; and Pong Bua Baan, a recently created village on the hill slope near Ban Hueng Ngu. Ban Pang, in Lamphun province, is in a narrow lowland valley surrounded by hill slopes where large areas were cleared for longan orchards. A peri-urban village near Chiang Mai, where agriculture is disappearing due to urban development, was also included in the entomological and epidemiological surveys.
The landscape is varied and heterogeneous but landscape units (human settlements, fields, orchards, forests) are larger than the Landsat pixel or are spatially clustered (Fig. 3). Landsat data, in combination with field observations, are thus appropriate to describe the relationship between landscape attributes, the presence of larval habitats, and human exposure to infection.
Materials and Methods
Data Collection
Land cover and land-cover change maps were derived from two Landsat images (Path 131, Row 047: 3 February 1989, TM5, 30 m resolution; and 5 March 2000, ETM+, 30 m; Eros Data Center, Sioux Falls). The images were coregistred, and radiometrically corrected using the invariant features method (Schott et al., 1988; Séguis and Puech, 1997). Image subsets corresponding to the study villages were georeferenced using the 1:50,000 topographic map of Thailand (Royal Thai Survey Department, 1992). Except for one subset where ground control points were scarce, all image subsets had root mean square errors lower than one pixel. Land cover and land-cover change were analyzed using, respectively, supervised maximum likelihood classification and change detection techniques. The accuracy assessment used aerial photographs from 1995 and field observations. Estimated Kappa classification accuracy statistics (KHAT) (Congalton, 1991) ranged between 0.75 and 0.86 for all subsets for both years. The villages were selected based on preliminary land-cover change analysis to cover a range of typical northern Thailand landscapes, and on epidemiological records obtained from local or provincial governmental agencies.
A survey of 223 farming households in seven villages allowed the identification of the most important changes in farming systems (Vanwambeke et al., 2006a). A close-ended questionnaire collected household information (occupations, composition, migrant status), agricultural inputs, cultivated areas and creation of new fields, crops grown and production sold, land tenure, forest products gathered, fishing, and fuel consumption. The questionnaire asked detailed information for 2001 and comparative information was collected for two preceding periods.
Mosquito larvae were collected in and around the study villages, up to 5 km from the village center, on eight occasions between May 2003 and April 2004, including the dry season (November–April) and wet season (May–October). A 5 km distance allows covering areas used by the villagers daily (based on village administrative boundaries and as confirmed by interviews) and potential sources of mosquitoes, considering maximum flight distance. Each of the 790 collections was associated with a description of larval habitat, date, and geographic coordinates collected with a Global Positioning System (GPS) with an average positional accuracy smaller than 10 m. Larval habitats were sampled by walking transects (over 900) that were on average 144-m long. Transects were located in each land-cover type detected by remote sensing around each village, well within the land-cover type (at more than 30 m from the edge). All larval habitats were surveyed except in land-cover types where they were very abundant, such as irrigated fields. In this case, a representative random sample was selected independently of the presence or absence of mosquito larvae. Habitats were searched for larvae by emptying the water from containers or by dipping with an enamel pan in larger water bodies. Measures of larvae numbers were recorded in categories of numbers. A sample of mosquito larvae was preserved in ethanol for identification. Morphological identification to species for Aedes or to species group for Anopheles was conducted by Ralph E. Harbach (National History Museum, London). Outdoor evening landing collections of adult females were conducted at 5–11 stations in each village, 11 times (two evenings each) between May 2001 and February 2003. In the analyses presented here, Aedes albopictus and Ae. aegypti, (= Stegomyia albopicta and St. aegypti, respectively, of Reinert et al. [2004]), and the An. minimus and An. maculatus groups were included. Anopheles dirus s.l., a complex of primary malaria vector species, was excluded from the study since very few were found in the study area. Adult data for species groups of Anopheles will be presented elsewhere.
Epidemiological data were collected in each village for either malaria or dengue, in a prospective survey over 3 years. The Medical Ethical Committee of Chiang Mai University approved the study, and local permission and collaboration were obtained. Villagers were asked to participate voluntarily in the study. After explaining the purpose of the study, written informed consent was obtained. Potential individual risk determinants were asked by formal questionnaire and included: sex, age, profession, place of birth, knowledge of dengue, daytime location, evening location, housing condition, and use of preventive measures. The dengue survey included the collection of blood samples to test for dengue antibodies (IgM). At the start of the survey, it included 1928 participants, of more than 4 years old in three villages, who were followed-up four times over 3 years (van Benthem et al., 2005; Vanwambeke et al., 2006b). The follow-up rate after 3 years was 81% (fifth survey). Surveys took place in September 2001, 2002, and 2003 (end of the rainy season) and in March 2002 and 2003 (end of the dry season). The participation rate varied from 25% in the peri-urban village to 78%–99% in the rural villages. The malaria survey followed a similar protocol, but, due to a very low incidence in the study villages, risk determinants could not be identified and are thus not reported here.
Methods
A multilevel logistic regression identified the characteristics of households and villages favoring the adoption of farming strategies. Epidemiological and entomological data were integrated with the land-cover maps in a geographic information system (GIS), which was then used to characterize each collection point (larval habitat or house) in terms of landscape structure and land use. Spatial and temporal risk determinants for recent dengue infection were analyzed using multiple logistic regression (van Benthem et al., 2005) and multilevel logistic regression (year, individual, and household) (Vanwambeke et al., 2006b). This method allows consideration of nested data, such as households in villages, or people within households, which violate the assumption of data independence (Kreft and De Leeuw, 1998). Multilevel methods combine within-group and between-group relationships (Snijders and Boskers, 1999), and integrate variables at several levels, e.g., village/household.
Integrated Model
The number of new infections for a disease d in a village v caused by a mosquito taxon c (i.e., incidence per mosquito species/species group) in a year y can be expressed as:
where Potential Biting Rate cy is the number of bites for mosquito taxon c and year y, Actual Biting Probability v is the probability for a potential bite to reach a person in village v, and Infective Bite Probability dc is the probability for a bite to be infective for disease d for mosquito taxon c. This served as the general framework for constructing the set of equations forming the model. Estimates of mosquito populations based on landscape data were used. In the case of dengue infection, transmission risk was then estimated taking human risk behaviors and preventive measures into account. The model thus included three steps: (i) production of larvae according to the availability of habitat for the immature stages (for malaria and dengue vectors), (ii) development of larvae and infection of adult mosquitoes (for malaria and dengue vectors), and (iii) for dengue only, the number of infective bites received by people according to risk behavior and use of preventive measures. The model functioned at the village level and infections (for dengue) were assumed to take place in or around houses (van Benthem et al., 2005). The detailed formulation of each step and their parameterization was based on the results of the statistical analyses of the data collected in the field and by remote sensing.
Statistical Results
Land-use Change
The main land-use changes observed between 1989 and 2000 in the rural study sites in northern Thailand were the clearing of forest for swidden farming or for permanent fields (mostly orchards), and the intensified use of irrigated fields. Clearings for permanent fields represented between 5% and 61% of the change observed in rural villages; these changes occupy up to 15% of a village’s territory (Table 1). Many clearings were related to orchard expansion, a strategy adopted by 15% of the interviewed households. The adoption of orchard expansion was related to the average orchard area per household in the village (adjusted Odds Ratio (aOR) = 3.77, 95% confidence interval (95%CI) = 0.98–14.60); traditional farming units, as proxied by the area of upland field, were less likely to expand the orchard area (aOR = 0.40, 95%CI = 0.21–1.03), as were those with a large area of orchard already under use by the household (aOR = 0.19, 95%CI = 0.07–0.52). The model had a Snijders and Bosker’s R 2 of 0.46. Intensification of irrigated land (Snijders and Bosker’s R 2 = 0.80) is related to the adoption of dry-season, drought-tolerant crops, but was mostly explained by village-level factors (intra-class correlation, i.e., proportion of observed variance at village level, 0.70). It was also related to the existence of a social network, measured by the number of other adopters in the village (aOR = 1.14, 95%CI = 1.01–1.28) (Vanwambeke et al., 2006a). Model results are summarized in Table 2.
Habitat of the Immature Stages of Mosquitoes
Data for presence/absence of larvae were collected at the level of habitats and were then related to landscape variables. The use of transects allowed the identification of larval habitats in various land-cover types, in the dry and wet seasons. The species and species groups were associated with habitat types, from which we derived a proportion of habitats used in the wet and dry season. Density and proportion of use were thus always associated with specific land-cover types (Tables 3, 4). Aedes aegypti was found exclusively in artificial containers in settled areas. Aedes albopictus was mostly found in artificial containers in villages but also in orchards, and in natural containers in both land covers. Aedes albopictus occupies a larger proportion of artificial containers in villages than Ae. aegypti. Except for artificial containers in orchards, all types of Aedes larval habitats had a higher density during the wet season. Members of the An. minimus and An. maculatus groups were found both in forest and villages, in stream habitats and ground pools. The density of stream margins is higher in villages. Stream pools are found more often in forests. Both species groups use a larger fraction of most of the larval habitats in the dry season, when some of them are denser. Anopheles minimus tends to use a larger fraction of the available larval habitats than An. maculatus. Anopheles minimus was found in the majority of stream margin habitats in villages and in a large fraction of those in forests. Members of both species groups were found most frequently in the dry season (Vanwambeke et al., 2007). A more detailed analysis of the influence of weather was not possible as we lacked localized meteorological data. Larvae collection rounds were however spread throughout the year and cover the intra-seasonal variability.
Risk Determinants of Dengue Seropositivity and Recent Dengue Infection
Six percent of the human study population showed recent infection in 2001, but rates of dengue infection varied between surveys and between sites, from less than 1% to over 25%, with a peak in 2002. Although spatial and temporal variation in significant risk determinants was observed, some risk determinants were recurrent. Factors associated with increased risk of infection were the use of abate (a larvicide) (aOR = 1.42, 95%CI = 0.99–2.02) and people spending evenings outside (aOR = 1.52, 95%CI = 1.01–2.29). Factors decreasing risk were the absence of water around the house (aOR = 0.63, 95%CI = 0.46–0.86) and the use of bednets (aOR= 0.43, 95%CI = 0.24–0.80) (van Benthem et al., 2005; Vanwambeke et al., 2006b).
Parameterization of Integrated Model
The section below provides the detailed formulation of each step of the integrated model and describes its parameterization based on the results of the above statistical analyses.
Larval Population As a Function of Landscape Structure
The first step of the model estimates the larval density likely to be found around a village. The land area considered was species/species group specific. It was defined by a circular buffer around the village corresponding to the average flight distance of the species/species group considered, avoiding overlap with neighboring villages by allocating each part of possible overlaps to the nearest village. No feedback between the adult and the larval population was included. The larval population for a given season was estimated from landscape data as:
where L x , the number of larvae of species x, is the sum of the average number of larvae present in habitats found in each land-cover type. There were H types of habitats and C types of land cover. All types of habitats were not found in each type of land cover. In one land-cover type c, for one habitat of type h, the number of larvae was the product of the area of the land cover S x(c) and the density of habitat type in that land cover D (h-c) , by the proportion of use of that habitat type in that land cover by species x U x(h-c) , and by the average number of larvae in each habitat used Avn x . Densities of habitats varied according to season (dry or wet). This product was then summed over all habitat and land-cover types. Initial values of S x(c) were calculated using the land-cover map of 2000. The variables D (h-c) and U x(h-c) were estimated from larval collection data based on transects (Tables 3, 4).
Adult Mosquito Development and Infection
The second step of the model estimates the number of larvae likely to become infective adult mosquitoes and the number of meals the females will take on humans. Attempts to link statistically larval habitats to adult mosquito abundance were unsuccessful, as the data sets were not matched temporally, and spatially explicit meteorological data were not available to produce a model with a satisfactory predictive power. This step was therefore based on parameters retrieved from the literature and on classic transmission modeling (Rogers, 1988; Smith and McKenzie, 2004).
The number of adults of species/species group x, A x , generated from the pool of larvae L x , depends on the survival rate of larvae to adulthood and the development time, which is a function of temperature. This was calculated for a model time-step of 1 month:
where s is the survival rate, P is the length of the time step in days and d(t) is development time in days as a function of temperature. This formulation is a simplification that ignores the age structure among larvae in the estimation of L x . A new pool of larvae L x is produced P/d(t) times in each time step of the model. Since land-use change is the focus of the model, average climatic conditions were used in the model. Monthly mean temperatures over a 12-year period in Chiang Mai were used.
B x , the number of potential bites given to humans by the emerged adults, depends on their longevity, length of gonotrophic cycle, and anthropophily:
where Lon is the mean longevity in days, An is the proportion of bites to humans, and G is the length of gonotrophic cycle in days. A factor of two restricts bites to females, which are assumed to represent half of the emerging adults. One bite is assumed to take place per gonotrophic cycle, except in the case of Aedes mosquitoes, for which a factor F accounting for multiple feeding behavior was included (F = 1 for Anopheles). A fixed rate of anthropophily was used. Lon and G are temperature-dependent, but the range of temperatures in the area is small enough for these to be considered fixed in the model.
The number of potential infective bites I x depends on the proportion of infective mosquitoes:
where R x (I) is the proportion of infective mosquitoes in the mosquito population.
The combination of Equations (3), (4), and (5) gives:
Literature sources for the parameters included here can be found in Appendix. Cross-referencing between several sources was often necessary. Final values were based on an expert judgment based on these sources. Equation (6) combines the Potential Biting Rate and the Infective Bite probability of the general Equation (1).
Infective Bites Received by People
The third step of the model was only developed for dengue since risk determinants for malaria infection could not be studied due to a very low incidence. It estimates, from the number of potential infective bites I x , the number of bites actually reaching susceptible people, based on data from the epidemiological survey. First, the number of potential infective bites from each species/species group was summed over a genus to the total number of potential infective bites for a disease:
This total was then used in the calculation of actual infective bites. The number of actual bites cannot be larger than I tot . Preventive measures reduce the ratio of actual to potential bites, and risk behaviors increase that ratio to a theoretical maximum value of one. In the model, the non-adoption of risk behavior was represented in the same way as the adoption of preventive measures. The efficacy of these measures was represented by the attributable risk fraction (Bruzzi et al., 1985; Rothman, 1998) estimated from the statistical analysis of dengue risk determinants.
where AB tot is the total number of infective bites received by people, M p is the rate of use of a preventive measure in the village population, E p is the efficacy of the preventive measure in protecting people from bites, M r is the rate of non-use of a risk behavior in the village population, E r is the efficacy of the non-risk behavior in protecting people from bites, and SR is the rate of susceptible people in the population. M p, E p , M r , and E r were calculated from data collected during the study. Only the most important risk determinants for dengue infection related to human behavior and use of preventive measures were retained. AB tot is equivalent to infection, but not to symptomatic dengue fever cases, since most infections are asymptomatic (Vanwambeke et al., 2006b). The sum of preventive measures and risk behavior used in Equation (8) corresponds to the Actual Biting Probability of the general Equation (1).
Error Estimation and Model Verification
Standard errors were calculated for each model parameter based on field-collected data (D (h-c) , U x(h-c) , and M p, E p , M r and E r ). Propagated errors were calculated following Mulligan and Wainwright (2004). In the calculation of the number of larvae, most parameters had a small standard error (Tables 3, 4); the largest errors were found for natural container habitats and habitats located in the forest. In those areas, habitats are not homogeneously distributed over space, and their density was therefore more difficult to estimate. In the case of human behavior, the largest errors were found for the variables related to mosquito development and habitat (water around houses and use of abate). The calculation of a standard error assumes a normal distribution, whereas U x(h-c) , M p , and M r are proportions and follow a binomial distribution. Approximations of standard errors were used but may have resulted in inflated errors.
Accurate prediction of disease incidence cannot be expected from this model, given the abbreviated structure of the model component on disease transmission. However, as no independent data were available to validate the other components of the model, a crude verification compared the model result for a baseline scenario, corresponding to the observed situation, with the observed number of recent dengue infections. Note that only passive surveillance of dengue fever cases is carried out by public health authorities in the study area, whereas the model predicts infection, and 65%–99.7% of infections were asymptomatic. We compared the number of dengue infections calculated by the model for three villages and the recent infections measured in the study population, for the dry and wet seasons (Table 5). The model predicted well the observations of September 2001 for all three sites. The match was less precise for September 2003, especially in the valley site with orchards close by. In May 2003, the model predicted correctly the observations for the dry season in one site but not in the other two. Errors could be related to risk determinants that were not included, such as protective housing characteristics in the peri-urban site. The year 2002 was a peak for dengue transmission in Thailand, and the model results did not predict the number of infections observed. Cyclical peak incidence in dengue cases has been observed in the form of waves emanating from Bangkok (Cummings et al., 2004). The explanation for these cycles is still uncertain, but recent hypotheses emphasize the role of interserotypic cross-immunity and immune selection of strains (Adams et al., 2006; Wearing and Rohani, 2006).These processes are not represented in the model. Aside from this peak year, the model produced a reasonable estimate of the number of new infections at the village level, especially in the wet season.
SCENARIOS
Scenarios provide plausible alternative images of how the future might unfold. Scenario results are not predictions. They are particularly useful when predictions cannot be made, e.g., to test the possible impact of events outside the domain of observations. Scenarios were generated at the village level to account for the diversity in environmental and social contexts. Scenarios included land-cover change (Scenarios 1 and 2), also combined with human behavioral change (Scenario 3), and changes in the density of mosquito habitats (Scenarios 4 and 5). Model outputs were compared to baseline conditions corresponding to the observed situation in the study villages. Villages were selected for scenario testing according to the importance of the vectors or disease considered. For example, low numbers of dengue vectors were found in upland villages, and transmission is currently unlikely in those areas.
Scenario 1: Forest-cover Decrease
A decrease in forest cover around villages was observed in several study sites, for example, related to agricultural expansion of orchards. As Anopheles mosquitoes partly occupy habitats in forest, this is expected to lead to a decline in their population. The scenario considers a 50% decrease in forest cover in the area within flight distance from the village. Significant impacts were noted for members of the An. minimus and An. maculatus groups, two important malaria vector taxa in Southeast Asia that inhabit both forests and village areas in the dry season. We selected two villages where large numbers of Anopheles are found and where malaria transmission had been recorded in the past few years. Forest closely surrounded the first village but was located further away from the other one, located in a valley. The decrease in forest cover resulted in a change in the population of both mosquito species group in the forested site and for the valley site. In the forested site, the difference in the An. minimus group was predicted to be slightly smaller than the decrease of An. maculatus group, and was proportional to the decrease in forest cover. In the valley site, the population of the An. minimus group was predicted to decrease much less than the population of the An. maculatus group (Table 6). This difference was due to the distinct distribution of habitats in the two villages: in the forested site, the village area provides approximately 4% of the An. minimus group, whereas in the less forested site, the village area provides approximately 27% of the population.
Scenario 2: Orchard Increase
Orchard expansion either takes place at the expense of forest, at a certain distance from villages, or by conversion of existing fields near villages. This was tested for two valley sites with high levels of dengue infection but different landscape patterns and varying importance of orchards in the farming system. Orchards increase in area in both villages, as they do in much of northern Thailand. An increase in the Ae. albopictus population is likely to result, leading to a significant effect on dengue transmission. Doubling the orchard area (S x(c) ) within the flight-distance of the mosquito had a large impact on Ae. albopictus populations (Table 7). In a site where orchards are on the valley slopes surrounding the irrigated valley floor (orchards are further than 500 m away from the village), orchards contributed 17% of the Ae. albopictus larvae in the dry season and 4% in the wet season. In another site where orchards are located in close proximity to the village (<100 m), they contributed 30% of Ae. albopictus larvae in the dry season and 8% in the wet season. The increase in number of Ae. albopictus larvae was therefore larger in this site, as orchards contribute a larger part of the population. In that site, the number of larvae was over 30% larger in the dry season.
Scenario 3: Orchard Increase and Increased Use of Preventive Measures
Orchard cultivation and the commercialization of fruit crops is generally associated with an increase in household income and with social changes related to engagement in a market economy. These changes could result in better knowledge about disease risk factors and more investment in protective measures against mosquito bites, or more generally in housing and sanitation improvements. Such effects have been observed for protection against malaria in Africa (see Ijumba and Lindsay, 2001, for examples). To what extent does better prevention compensate for the increase in potential bites caused by an increase in mosquito population in and around orchards? Actually, the rate of use of preventive measures being already very high in the valley villages studied, marginal improvements more than compensated for the increase in potential bites. Combining a 100% increase in orchard area (as in Scenario 2) with the use of preventive measures (M p ) led to a complete suppression of all Aedes bites.
Scenario 4: Dam Construction
Numerous dams have been erected in northern Thailand in the past, and various projects are currently under planning. Downstream of dams, streams create favorable habitats for mosquitoes, often under tree cover. This would favor Anopheles species that inhabit forests. This scenario simulated a change in the density of permanent stream margin and stream pool habitats. With a year-round 10% increase in stream habitats in forest areas, both An. minimus and An. maculatus populations increased by a proportion smaller than 10%, with a minor seasonal effect (Table 6). This effect is related to the respective contributions of forest and village areas in the total mosquito populations in the dry and wet seasons.
Scenario 5: Artificial Container Elimination
Dengue prevention campaigns in Thailand and elsewhere emphasize the elimination or covering of artificial containers by citizens, as they provide the main larval habitat for dengue vectors and are often found around houses on private properties. A 50% decrease in the density of artificial containers in villages during the wet season, when water-filled artificial containers are most frequently found, was simulated. Aedes aegypti, which only lay eggs in artificial containers in villages, was decreased proportionally, as expected. Aedes albopictus also breeds in artificial containers in orchards where larval habitats were not eliminated and therefore its population decreased by a smaller percentage (Table 7). Still, artificial container elimination was predicted to lead to a significant decrease in the number of infective bites received by people.
Discussion and Conclusions
The impact of land-use/land-cover change on the risk of two of the most serious mosquito-borne diseases, malaria and dengue was investigated. Extensive data collection and statistical analyses were conducted by entomologists, epidemiologists, and land-use scientists, who then combined their efforts in building an integrated understanding of the relationships between mosquito populations, disease transmission, and land use. This interdisciplinary work led to a model including explicit causal relationships based on empirical observations. This permits the examination of the effects of changes in specific aspects of the system studied, mostly land-use changes frequently encountered in northern Thailand. The integrated model explicitly includes the link between landscape attributes and larval vector ecology. It combined empirical statistical relationships with a simplified representation of the biology of vector development and vector-borne disease transmission. It details causal relationships linking changes in land cover, vector abundance, and risk of infection better than would be the case with purely empirical relationships. It integrates land use and landscape heterogeneity into approaches in epidemiology that have often assumed the environment to be a homogenous space. The model also accounted for a variety of human risk and preventive behaviors.
The data and scenario analyses suggested that land-use changes that are currently widespread across northern Thailand have a detectable impact on mosquito populations, leading to a population increase of some species or species groups, and a decrease of others. Forest decrease, associated in our scenarios with a decrease in malaria vectors, is often related to the expansion of orchards, which hosts Ae. albopictus, a dengue vector. Mosquitoes laying eggs in more than one land-cover type and/or more than one larval habitat type have more complex—and thus less easily predictable—responses to land-use/land-cover change, as was illustrated by Scenario 2 and Aedes mosquitoes. Beyond the relationship between land-use change and mosquito population, the impact on infection and disease of these changes is further complicated by human behavior. The location of human residences and activities in relation to sources of mosquitoes is a crucial element. Changes in orchard area led to an increase in Ae. albopictus population but this could be counteracted by adaptive and preventive measures. Deforestation is associated with a decrease in An. minimus populations but, as this species group also breeds in villages where it is in closer contact with humans, changes in housing infrastructure could potentially increase biting rate. Policy intervention, education campaigns, and adoption of preventive measures can counteract (or enhance) effects caused by land-use change, as indicated by Scenario 3.
The unexpected effect of the use of abate (that increases the risk of dengue infection) suggests that the adequate use of preventive measures should be monitored. Delayed or incorrect application could explain this relationship. Use of preventive measures such as abate may also reflect a high mosquito density, as found elsewhere (Thomson et al., 1996). Interactions between land-use change, use of preventive measures, and control policies often lead to non-linear effects on the presence of different mosquito species (Ijumba and Lindsay, 2001). Agricultural intensification and orchard expansion can result in greater integration of households into a market economy, more contacts with urban centers, better awareness about disease risk, and higher income to invest in preventive measures, e.g., window screens and bednets. These changes can influence disease transmission at least as strongly as effects on mosquito populations, and can act towards an increase or a decrease of the risk.
Changes in land use, preventive measures, and control policies will not necessarily have the same effects in different villages. Their impact depends on many factors, including landscape structure, type of housing, level of education, and immigration of infected individuals. Policy intervention for disease control therefore needs to be fine-tuned to local ecological and social settings. Land-use change does have an influence on mosquito populations and disease transmission risk, but its exact effect cannot be easily predicted without this local-scale contextual information.
These results cannot be balanced easily against potential effects of climate change. The relative importance of changes in climate and in land cover would likely vary between places and occur at different spatial and temporal scales. Combining the region-wide effects of climate and the landscape-level effects of land cover and land use on disease transmission is an important challenge.
The value of intact ecosystems, such as forests, in regulating pathogens and disease has been suggested by a number of authors (e.g., Costanza et al., 1997; Foley et al., 2005). The results of this study, which shows that some vectors may increase while others decrease as a result of natural forest conversion, suggests that, at least on the local landscape scale, the presence of forest ecosystems may contribute to, and not diminish, disease. Thus, it could be argued, ecosystems provide “disservices” as well as services. The potential ecosystem “disservice” of supporting vectors should be considered in land-use planning and ecosystem management. The complexity of vector-borne disease transmission calls for an integrated approach considering ecological, biological, and human aspects (Spiegel et al., 2005). Scenario formulation combined with an integrated model calibrated on a large data set allowed assessing of the implications for potential transmission of likely changes in land use, human behavior, control policies, or any combination of these. Interactions between the various changes call for further efforts in developing an interdisciplinary, integrated approach to the multiple factors that influence the intensity of disease transmission. The practice of disease control has already recognized the need for such an integrated approach (Carter et al., 2000; Reiter, 2001), but still suffers from institutional barriers to its implementation.
Feedback from a high risk of disease transmission to land management should exist in cases where the disease risk is high enough to influence land-use decisions. Land conversion that would significantly increase disease risk beyond any capacity to apply preventive measures should be avoided or regulated through policies. In the case of malaria and dengue in Thailand, such a feedback was not observed given available preventive measures that are effective and can be applied at a socially acceptable cost.
References
Adams B, Holmes E, Zhang C, Mammen MJ, Nimmannitya S, Kalayanarooj S, et al. (2006) Cross-protective immunity can account for the alternating epidemic pattern of dengue virus serotypes circulating in Bangkok. Proceedings of the National Academy of Sciences of the United States of America 103:14234–14239
Anderson RM, May RM (1991) Infectious Diseases of Human, Oxford, UK: Oxford University Press
Bruzzi P, Green SB, Byar DP, Brinton LA, Schaier C (1985) Estimating the population attributable risk for multiple risk factors using case-control data. American Journal of Epidemiology 122:904–914
Carter R, Mendis KN, Roberts D (2000) Spatial targeting of intervention against malaria. Bulletin of the World Health Organisation 78:1401–1411
Congalton RG (1991) A review of assessing the accuracy of classification of remotely sensed data. Remote Sensing of Environment 37:35–46
Costanza R, d’Arge R, de Groot R, Farber S, Grasso M, Hannon B, et al. (1997) The value of the world’s ecosystem services and natural capital. Nature 387:253–260
Cummings DAT, Irizarry RA, Huang NE, Endy TP, Nisalak A, Ungchusak K, et al. (2004) Travelling waves in the occurrence of dengue haemorrhagic fever in Thailand. Nature 427:344–347
Focks DA, Daniels E, Haile DG, Keesling JE (1995) A simulation model of the epidemiology of urban dengue fever: literature analysis, model development, preliminary validation, and samples of simulation results. American Journal of Tropical Medicine and Hygiene 53:489–506
Focks DA, Haile DG, Daniels E, Mount GA (1993) Dynamic life table model for Aedes aegypti (Diptera: Culicidae): analysis of the literature and model development. Journal of Medical Entomology 30:1003–1017
Foley JA, DeFries R, Asner GP, Barford C, Bonana G, Carpenter SR, et al. (2005) Global consequences of land use. Science 309:570–574
Fungladda W, Butraporn P (1992) Malaria-related social and behavioral risk factors in Thailand: a review. Southeast Asian Journal of Tropical Medicine and Hygiene 23(Suppl 1):57–62
Ijumba JN, Lindsay SW (2001) Impact of irrigation on malaria in Africa: paddies paradox. Medical and Veterinary Entomology 15:1–11
Kitron U (1998) Landscape ecology and epidemiology of vector-borne diseases: tools for spatial analysis. Journal of Medical Entomology 35:435–445
Kreft I, De Leeuw J (1998) Introducing Multilevel Modeling, London: Sage
Lambin EF, Geist HJ, Lepers E (2003) Dynamics of land-use and land-cover change in tropical regions. Annual Review of Environmental Resources 28:205–241
Lambin EF, Turner BL, Geist HJ, Agbola SB, Angelsen A, Bruce JW, et al. (2001) The causes of land-use and land-cover change: moving beyond the myths. Global Environmental Change 11: 216–269
Mulligan M, Wainwright J (2004) Modelling and model building. In: Wainwright J, Mulligan M (eds) Environmental Modelling. Finding Simplicity in Complexity, Chichester, UK: Wiley, pp 7–73
Patz JA, Daszak P, Tabor GM, Aguirre AA, Pearl M, Epstein J, et al. (2004) Unhealthy landscapes: policy recommendations on land use change and infectious disease emergence. Environmental Health Perspectives 112:1092–1098
Patz JA, Norris DE (2004) Land use change and human health. In: DeFries R, Asner G, Houghton R (eds) Ecosystems and Land Use Change, Geophysical Monograph 153, Washington, DC: American Geophysical Union, pp 159–167
Reinert JF, Harbach RE, Kitching IJ (2004) Phylogeny and classification of Aedini (Diptera: Culicidae), based on morphological characters of all life stages. Zoological Journal of the Linnean Society 142:289–368
Reiter P (2001) Climate change and mosquito-borne disease. Environmental Health Perspectives 109(Suppl 1):141–161
Rogers DJ (1988) The dynamics of vector-transmitted diseases in human communities. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences 321:513–539
Rothman KJ (1998) Modern Epidemiology, Philadelphia: Lippincott–Raven
Schmidt-Vogt D (1999) Swidden Farming and Fallow Vegetation in Northern Thailand, Stuttgart, Germany: Franz Steiner Verlag
Schott JR, Salvaggio C, Volchok WJ (1988) Radiometric scene normalization using pseudoinvariant features. Remote Sensing of Environment 26:1–16
Séguis L, Puech C (1997) Méthode de détermination des invariants radiométriques adaptée au paysage semi-aride de l’Afrique de l’Ouest. International Journal of Remote Sensing 18:255–271
Service MW, Townson H (2002) The Anopheles vector. In: Warrel DA, Gilles HM (eds), Essential Malariology Arnold London pp 59–84
Smith DL, McKenzie FE (2004) Statics and dynamics of malaria infection in Anopheles mosquitoes. Malaria Journal 3:13. Available: http://www.malariajournal.com/content/3/1/13 [accessed November 30, 2006]
Snijders TAB, Boskers RJ (1999) Multilevel Analysis. An Introduction to Basic and Advanced Multilevel Modeling, London: Sage
Snow RW, Gilles HM (2002) The epidemiology of malaria. In: Warrel DA, Gilles HM (eds) Essential Malariology, Arnold London pp 85–106
Spiegel J, Bennett S, Hattersley L, Hayden MH, Kittayapong P, Nalim S, et al. (2005) Barriers and bridges to prevention and control of dengue: the need for a socio-ecological approach. EcoHealth 2:273–290
Thomson M, Connor S, Bennett S, D’Alessandro U, Milligan P, Aikins M, et al. (1996) Geographical perspectives on bednet use and malaria transmission in The Gambia, West Africa. Social Science and Medicine 43:101–112
Tran A, Raffy M (2006) On the dynamics of dengue epidemics from large-scale information. Theoretical Population Biology 69:3–12
van Benthem BHB, Vanwambeke SO, Khantikul N, Burghoorn-Maas C, Panart K, Oskam L, et al. (2005) Spatial patterns of and risk factors for seropositivity for dengue infection. American Journal of Tropical Medicine and Hygiene 72:201–208
Vanwambeke SO, Somboon P, Harbach RH, Isenstadt M, Lambin EF, Walton C, et al. (2007) Landscape and land-cover factors influence the presence of Aedes and Anopheles larvae. Journal of Medical Entomology 44:133–144
Vanwambeke SO, Somboon P, Lambin EF (2006a) Rural transformation and social differentiation in northern Thailand. Journal of Land Use Science 2:1–29
Vanwambeke SO, van Benthem BHB, Khantikul N, Burghoorn-Maas C, Panart K, Oskam L, et al. (2006b) Multi-level analyses of spatial and temporal determinants for dengue infection. International Journal of Health Geographics 5:5. Available: http://www.ij-healthgeographics.com/content/5/1/5 [accessed November 30, 2006]
Wearing HJ, Rohani P (2006) Ecological and immunological determinants of dengue epidemics. Proceedings of the National Academy of Sciences of the United States of America 103:11802–11807
WHO (1985) Arthropod-borne and Rodent-borne Viral Diseases. Report of a WHO Scientific Group. WHO Technical Report Series 719
Wilcox BA, Colwell RR (2005) Emerging and reemerging infectious diseases: biocomplexity as an interdisciplinary paradigm. EcoHealth 2:1–14
Acknowledgments
This study was financially supported by EU grant QLRT-1999-31787, provided within the Quality of Life and Management of Living Resources Programme (1998–2002). Mark Isenstadt and Conor Cahill conducted the mosquito collections. We thank David J. Rogers from Oxford University for his comments on an earlier version of the manuscript. We also thank the participants of the RISKMODEL final workshop that was held in Chiang Mai on September 26–27, 2005.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
References
-
1.
Clements AN (1992) The Biology of Mosquitoes, Vol 1. Development, Nutrition and Reproduction, London: Chapman & Hall.
-
2.
Clements AN (1999) The Biology of Mosquitoes, Vol 2. Sensory Reception and Behaviour, Wallingford, Oxon, UK: CABI
-
3.
Coleman RE, Sithiprasasna R, Kankaew P, Kiattibut C, Ratanawong S, Khuntirat B, et al. (2002) Naturally occurring mixed infection of Plasmodium vivax VK210 and P. vivax VK247 in Anopheles mosquitoes (Diptera: Culicidae) in western Thailand. Journal of Medical Entomology 39:556–559
-
4.
Gilles HM, Warrell DA (2002) Essential Malariology, 4th ed., London: Arnold
-
5.
Gingrich JB, Weatherhead A, Sattabongkot J, Pilakasiri C, Wirtz RA (1990) Hyperendemic malaria in a Thai village: dependence of year-round transmission on focal and seasonally circumscribed mosquito (Diptera: Culicidae) habitats. Journal of Medical Entomology 27:1016–1026
-
6.
Green CA, Rattanarithikul R, Pongparit S, Sawadwongporn P, Baimai V (1991) A newly-recognised vector of human malarial parasites in the Oriental region, Anopheles (Cellia) pseudowillmori (Theobald, 1910). Transactions of the Royal Society of Tropical Medicine and Hygiene 85:35–36
-
7.
Harbach RE, Gingrich JB, Pang LW (1987) Some entomological observations on malaria transmission in a remote village in northwestern Thailand. Journal of the American Mosquito Control Association 3:296–301
-
8.
Horsfall WR (1955) Mosquitoes: Their Bionomics and Relation to Disease, New York: Ronald Press
-
9.
Kanda T, Bunnang D, Deesin V, Deesin T, Leemingsawat S, Komalamisra N, et al. (1995) Integration of control measures for malaria vectors in endemic areas of Thailand. Southeast Asian Journal of Tropical Medicine and Public Health 26:154–163
-
10.
Lien JC (1991) Anopheline mosquitoes and malaria parasites in Taiwan. Kaohsiung Journal of Medical Science 7:207–223
-
11.
Loong KP, Chiang GL, Yap HH (1988) Field studies of the bionomics of Anopheles maculatus and its role in malaria transmission in Malaysia. Southeast Asian Journal of Tropical Medicine and Public Health 19:724.
-
12.
Oo TT, Storch V, Becker N (2004) Review of the anopheline mosquitoes of Myanmar. Journal of Vector Ecology 29:21–40
-
13.
Rahman WA, Hassan AA, Adanan CR, Rashid MRA, Khalid AH (1992) Malaria transmission in a remote village located in northern peninsular Malaysia near the Malaysia–Thailand border. Tropical Biomedicine 9:83–89
-
14.
Ratanatham S, Upatham ES, Prasittisuk C, Rojanasunan W, Theerasilp N, Tremongkol A, et al. (1988) Bionomics of Anopheles minimus and its role in malaria transmission in Thailand. Southeast Asian Journal of Tropical Medicine and Public Health 19:283–289
-
15.
Rattanarithikul R, Konishi E, Linthicum KJ (1996) Detection of Plasmodium vivax and Plasmodium falciparum circumsporozoite antigen in anopheline mosquitoes collected in southern Thailand. American Journal of Tropical Medicine and Hygiene 54:114–121
-
16.
Reid JA (1968) Anopheline Mosquitoes of Malaya and Borneo, Government of Malaysia, Kuala Lumpur, Malaysia
-
17.
Rosenberg R, Andre RG, Somchit L (1990) Highly efficient dry season transmission of malaria in Thailand. Transactions of the Royal Society of Tropical Medicine and Hygiene 84:22–28
-
18.
Scanlon JE, Sandinhand U (1965) The distribution and biology of Anopheles balabacensis in Thailand (Diptera: Culicidae). Journal of Medical Entomology 2:61–69
-
19.
Service MW (1993) Mosquito Ecology: Field Sampling Methods, 2nd ed., Barking, UK: Elsevier Scientific Publishers
-
20.
Somboon P, Aramrattana A, Lines J, Webber R (1998) Entomological and epidemiological investigations of malaria transmission in relation to population movements in forest areas of north-west Thailand. Southeast Asian Journal of Tropical Medicine and Public Health 29:3–9
-
21.
Suwonkerd W, Overgaard HJ, Tsuda Y, Prajakwong S, Takagi M (2002) Malaria vector densities in transmission and non-transmission areas during 23 years and land use in Chiang Mai province, northern Thailand. Basic and Applied Ecology 3:197–207
-
22.
Takagi M, Suwonkerd W, Tsuda Y, Kamboonruang C, Chipralop U, Nakazawa S, et al. (1995) Seasonal density and malaria vector competence of Anopheles minimus and other anophelines at a shallow valley in northern Thailand. Japanese Journal of Tropical Medicine and Hygiene 23:177–182
-
23.
Tewari SC, Thenmozhi V, Katholi CR, Manavalan R, Munirathinam A, Gajanana A (2004) Dengue vector prevalence and virus infection in a rural area in south India. Tropical Medicine and International Health 9:499–507
-
24.
Toma T, Miyagi I, Okazawa T, Kobayashi J, Saita S, Tuzuki A, et al. (2002) Entomological surveys of malaria in Khammouane Province, Lao PDR, in 1999 and 2000. Southeast Asian Journal of Tropical Medicine and Public Health 33:532–546
-
25.
Trung HD, Van Bortel W, Sochantha T, Keokenchanh K, Quang NT, Cong LD, et al. (2004) Malaria transmission and major malaria vectors in different geographical areas of Southeast Asia. Tropical Medicine and International Health 9:230–237
-
26.
Tun-Lin W, Myat-Myat-Thu, Sein-Maung-Than, Maung-Maung-Mya (1995) Hyperendemic malaria in a forested, hilly Myanmar village. Journal of the American Mosquito Control Association 11:401–407
-
27.
Upatham ES, Prasittisuk C, Ratanatham S, Green CA, Rojanasunan W, Setakana P, et al. (1988) Bionomics of Anopheles maculatus complex and their role in malaria transmission in Thailand. Southeast Asian Journal of Tropical Medicine and Public Health 19:259–269
-
28.
Vythilingam I, Phetsouvanh R, Keokenchanh K, Yengmala V, Vanisaveth V, Phompida S, et al. (2003) The prevalence of Anopheles (Diptera: Culicidae) mosquitoes in Sekong Province, Lao PDR in relation to malaria transmission. Tropical Medicine and International Health 8:525–535
-
29.
Wharton RH, Laing ABG, Cheung WH (1963) Studies on distribution and transmission of malaria and filariasis among aborigines in Malaya. Annals of Tropical Medicine and Parasitology 57:235–254
-
30.
Wijeyaratne PM, Seawright JA, Weidhaas DE (1974) Development and survival of a natural population of Aedes aegypti. Mosquito News 34:36–42
About this article
Cite this article
Vanwambeke, S.O., Lambin, E.F., Eichhorn, M.P. et al. Impact of Land-use Change on Dengue and Malaria in Northern Thailand. EcoHealth 4, 37–51 (2007). https://doi.org/10.1007/s10393-007-0085-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10393-007-0085-5