Abstract
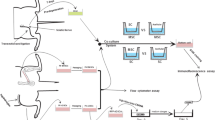
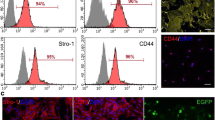
Traumatic brain injuries and degenerative neurological disorders such as Alzheimer’s dementia, Parkinson’s disease, amyotrophic lateral sclerosis and many others are characterized by loss of brain cells and supporting structures. Restoring microanatomy and function using stem cells is a promising therapeutic approach. Among the many various sources, adipose-derived stem cells (ADSCs) are one of the most easily harvested alternatives, they multiply rapidly, and they demonstrate low immunogenicity with an ability to differentiate into several cell types. The objective of this study was to evaluate the effect of xenotransplanted human ADSCs on post-traumatic regeneration of rat sciatic nerve. Peripheral reconstruction following complete sciatic transection and autonerve grafting was complemented by intra-operative injection of hADSCs into the proximal and distal stumps. The injury caused gliosis and apoptosis of sensory neurons in the lumbar 5 (L5) ganglia in the control rodents; however, animals treated with hADSCs demonstrated a smaller amount of cellular loss. Formation of amputation neuroma, which hinders axonal repair, was less prominent in the experimental group, and immunohistochemical analysis of myelin basic protein showed good myelination 65 days after surgery. At this point, control groups still exhibited high levels of microglia/macrophage-specific marker Iba-1 and proliferating cell nuclear antigen, the mark of an ongoing inflammation and incomplete axonal growth 2 months after the injury. This report demonstrates that hADSCs promote neuronal survival in the spinal ganglion, fuel axonal repair and stimulate the regeneration of peripheral nerves.











Similar content being viewed by others
References
Dubovy P. Schwann cells and endoneurial extracellular matrix molecules as potential cues for sorting of regenerated axons: a review. Anat Sci Int. 2004;79(4):198–208. doi:10.1111/j.1447-073x.2004.00090.x.
Dezawa M, Ishikawa H, Hoshino M, Itokazu Y, Nabeshima Y. Potential of bone marrow stromal cells in applications for neuro-degenerative, neuro-traumatic and muscle degenerative diseases. Curr Neuropharmacol. 2005;3(4):257–66.
Radtke C, Wewetzer K, Reimers K, Vogt PM. Transplantation of olfactory ensheathing cells as adjunct cell therapy for peripheral nerve injury. Cell Transplant. 2011;20(2):145–52. doi:10.3727/096368910X522081.
Kingham PJ, Kalbermatten DF, Mahay D, Armstrong SJ, Wiberg M, Terenghi G. Adipose-derived stem cells differentiate into a Schwann cell phenotype and promote neurite outgrowth in vitro. Exp Neurol. 2007;207(2):267–74. doi:10.1016/j.expneurol.2007.06.029.
Arkhipova SS, Raginov IS, Mukhitov AR, Chelyshev YA. Satellite cells of sensory neurons after various types of sciatic nerve trauma in the rat. Neurosci Behav Physiol. 2010;40(6):609–14. doi:10.1007/s11055-010-9303-7.
Gordon T, Tyreman N, Raji MA. The basis for diminished functional recovery after delayed peripheral nerve repair. J Neurosci. 2011;31(14):5325–34. doi:10.1523/JNEUROSCI.6156-10.2011.
McKay Hart A, Brannstrom T, Wiberg M, Terenghi G. Primary sensory neurons and satellite cells after peripheral axotomy in the adult rat: timecourse of cell death and elimination. Exp Brain Res. 2002;142(3):308–18. doi:10.1007/s00221-001-0929-0.
Raginov IS, Chelyshev IuA. Post-traumatic survival in different subpopulations of sensory neurons. Morfologiia. 2003;124(4):47–50.
Tandrup T, Woolf CJ, Coggeshall RE. Delayed loss of small dorsal root ganglion cells after transection of the rat sciatic nerve. J Comp Neurol. 2000;422(2):172–80. doi:10.1002/(SICI)1096-9861(20000626)422:2<172:AID-CNE2>3.0.CO.
Vigneswara V, Berry M, Logan A, Ahmed Z. Caspase-2 is upregulated after sciatic nerve transection and its inhibition protects dorsal root ganglion neurons from apoptosis after serum withdrawal. PLoS One. 2013;8(2):e57861. doi:10.1371/journal.pone.0057861.PONE-D-12-35011.
Weerasuriya A, Mizisin AP. The blood-nerve barrier: structure and functional significance. Methods Mol Biol. 2011;686:149–73. doi:10.1007/978-1-60761-938-3_6.
Tamaki T, Hirata M, Soeda S, Nakajima N, Saito K, Nakazato K, et al. Preferential and comprehensive reconstitution of severely damaged sciatic nerve using murine skeletal muscle-derived multipotent stem cells. PLoS One. 2014;9(3):e91257. doi:10.1371/journal.pone.0091257.PONE-D-13-38421.
Perry VH, Teeling J. Microglia and macrophages of the central nervous system: the contribution of microglia priming and systemic inflammation to chronic neurodegeneration. Semin Immunopathol. 2013;35(5):601–12. doi:10.1007/s00281-013-0382-8.
Kalinina NI, Sysoeva VY, Rubina KA, Parfenova YV, Tkachuk VA. Mesenchymal stem cells in tissue growth and repair. Acta Naturae. 2011;3(4):30–7.
Faroni A, Terenghi G, Reid AJ. Adipose-derived stem cells and nerve regeneration: promises and pitfalls. Int Rev Neurobiol. 2013;108:121–36. doi:10.1016/B978-0-12-410499-0.00005-8.
Solovyeva VV, Salafutdinov II, Martynove EV. Human adipose derived stem cells do not alter cytokine secretion in response to the genetic modification with pEGFP-N2 Plasmid DNA. World Appl Sci J. 2013;26(7):968–72.
Momeni HR, Soleimani Mehranjani M, Shariatzadeh MA, Haddadi M. Caspase-mediated apoptosis in sensory neurons of cultured dorsal root ganglia in adult mouse. Cell J. 2013;15(3):212–7.
Chen L, Qiu R, Xu Q. Mesenchymal stem cell therapy for neurodegenerative diseases. J Nanosci Nanotechnol. 2014;14(1):969–75.
Masgutov RF, Salafutdinov II, Bogov AA. The stimulation of posttraumatic regeneration of the sciatic nerve of a rat with a plasmid expressing a vascular endothelial growth factor and the basic fibroblast growth factor. Cell Transplant Tissue Eng. 2011;6(3):67–70.
Cheutin T, Gorski SA, May KM, Singh PB, Misteli T. In vivo dynamics of Swi6 in yeast: evidence for a stochastic model of heterochromatin. Mol Cell Biol. 2004;24(8):3157–67.
Lo DC. Neurotrophic factors and synaptic plasticity. Neuron. 1995;15(5):979–81.
Goldberg JL, Barres BA. Nogo in nerve regeneration. Nature. 2000;403(6768):369–70. doi:10.1038/35000309.
Ribeiro-Resende VT, Carrier-Ruiz A, Lemes RM, Reis RA, Mendez-Otero R. Bone marrow-derived fibroblast growth factor-2 induces glial cell proliferation in the regenerating peripheral nervous system. Mol Neurodegener. 2012;7:34. doi:10.1186/1750-1326-7-34.
Otoshi K, Kikuchi S, Konno S, Sekiguchi M. The reactions of glial cells and endoneurial macrophages in the dorsal root ganglion and their contribution to pain-related behavior after application of nucleus pulposus onto the nerve root in rats. Spine (Phila Pa 1976). 2010;35(3):264–71. doi:10.1097/BRS.0b013e3181b8b04f.
Rodriguez J, Lazebnik Y. Caspase-9 and APAF-1 form an active holoenzyme. Genes Dev. 1999;13(24):3179–84.
Cheng C, Zochodne DW. Sensory neurons with activated caspase-3 survive long-term experimental diabetes. Diabetes. 2003;52(9):2363–71.
Wood MD, Kemp SW, Weber C, Borschel GH, Gordon T. Outcome measures of peripheral nerve regeneration. Ann Anat. 2011;193(4):321–33. doi:10.1016/j.aanat.2011.04.008.
Abram SE, Yi J, Fuchs A, Hogan QH. Permeability of injured and intact peripheral nerves and dorsal root ganglia. Anesthesiology. 2006;105(1):146–53.
Berg A, Zelano J, Pekna M, Wilhelmsson U, Pekny M, Cullheim S. Axonal regeneration after sciatic nerve lesion is delayed but complete in GFAP- and vimentin-deficient mice. PLoS One. 2013;8(11):e79395. doi:10.1371/journal.pone.0079395.PONE-D-13-32448.
Ginhoux F, Lim S, Hoeffel G, Low D, Huber T. Origin and differentiation of microglia. Front Cell Neurosci. 2013;7:45. doi:10.3389/fncel.2013.00045.
Kettenmann H, Hanisch UK, Noda M, Verkhratsky A. Physiology of microglia. Physiol Rev. 2011;91(2):461–553. doi:10.1152/physrev.00011.2010.
Shachpazyan NR, Astrelina TA, Yakovleva MV. Mesenchymal stem cells derived from various human tissues: biological properties, evaluation of their quality and safety for clinical use. Cell Transplant Tissue Eng. 2012;7(1):23–33.
Reid AJ, Sun M, Wiberg M, Downes S, Terenghi G, Kingham PJ. Nerve repair with adipose-derived stem cells protects dorsal root ganglia neurons from apoptosis. Neuroscience. 2011;199:515–22. doi:10.1016/j.neuroscience.2011.09.064.
Marconi S, Castiglione G, Turano E, Bissolotti G, Angiari S, Farinazzo A, et al. Human adipose-derived mesenchymal stem cells systemically injected promote peripheral nerve regeneration in the mouse model of sciatic crush. Tissue Eng Part A. 2012;18(11–12):1264–72. doi:10.1089/ten.TEA.2011.0491.
Acknowledgments
The study was supported by Grant 13-04-12035 from Russian Foundation for Basic Research. This work was performed in accordance with Program of Competitive Growth of Kazan Federal University and subsidy allocated to Kazan Federal University for the state assignment in the sphere of scientific activities. Yana O. Mukhamedshina was supported by Presidential grant for government support of young scientists (PhD) from the Russian Federation (4020.2015.7). Some of the experiments were conducted using equipment at the Interdisciplinary center for collective use of Kazan Federal University supported by Ministry of Education of Russia (ID RFMEFI59414X0003), Interdisciplinary center for analytical microscopy, and Pharmaceutical Research and Education Center, Kazan (Volga Region) Federal University, Kazan, Russia. The authors are also indebted to Drs. Gallyamov and Bogov for their assistance in animal handling.
Conflict of interest
Authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Masgutov, R.F., Masgutova, G.A., Zhuravleva, M.N. et al. Human adipose-derived stem cells stimulate neuroregeneration. Clin Exp Med 16, 451–461 (2016). https://doi.org/10.1007/s10238-015-0364-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-015-0364-3