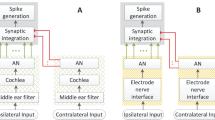
Abstract
Bilateral cochlear implants (CIs) provide improvements in sound localization and speech perception in noise over unilateral CIs. However, the benefits arise mainly from the perception of interaural level differences, while bilateral CI listeners’ sensitivity to interaural time difference (ITD) is poorer than normal. To help understand this limitation, a set of ITD-sensitive neural models was developed to study binaural responses to electric stimulation. Our working hypothesis was that central auditory processing is normal with bilateral CIs so that the abnormality in the response to electric stimulation at the level of the auditory nerve fibers (ANFs) is the source of the limited ITD sensitivity. A descriptive model of ANF response to both acoustic and electric stimulation was implemented and used to drive a simplified biophysical model of neurons in the medial superior olive (MSO). The model’s ITD sensitivity was found to depend strongly on the specific configurations of membrane and synaptic parameters for different stimulation rates. Specifically, stronger excitatory synaptic inputs and faster membrane responses were required for the model neurons to be ITD-sensitive at high stimulation rates, whereas weaker excitatory synaptic input and slower membrane responses were necessary at low stimulation rates, for both electric and acoustic stimulation. This finding raises the possibility of frequency-dependent differences in neural mechanisms of binaural processing; limitations in ITD sensitivity with bilateral CIs may be due to a mismatch between stimulation rate and cell parameters in ITD-sensitive neurons.











Similar content being viewed by others
References
Agmon-Snir H, Carr CE, Rinzel J (1998) The role of dendrites in auditory coincidence detection. Nature 393:268–272
American Academy of Otolaryngology-Head and Neck Surgery (2014) Position statement on cochlear implants
Barnes-Davies M, Barker MC, Osmani F, Forsythe ID (2004) Kv1 currents mediate a gradient of principal neuron excitability across the tonotopic axis in the rat lateral superior olive. Eur J Neurosci 19:325–333
Batra R, Kuwada S, Fitzpatrick DC (1997) Sensitivity to interaural temporal disparities of low- and high-frequency neurons in the superior olivary complex. I. Heterogeneity of responses. J Neurophysiol 78:1222–1236
Baumann VJ, Lehnert S, Leibold C, Koch U (2013) Tonotopic organization of the hyperpolarization-activated current (IH) in the mammalian medial superior olive. Front Neural Circuits 7:117
Bernstein LR, Trahiotis C (2002) Enhancing sensitivity to interaural delays at high frequencies by using “transposed stimuli”. J Acoust Soc Am 112:1026–1036
Brand A, Behrend O, Marquardt T, Mcalpine D, Grothe B (2002) Precise inhibition is essential for microsecond interaural time difference coding. Nature 417:543–547
Brew HM, Forsythe ID (2005) Systematic variation of potassium current amplitudes across the tonotopic axis of the rat medial nucleus of the trapezoid body. Hear Res 206:116–132
Bruce IC, Irlicht LS, White MW, O'Leary SJ, Dynes S, Javel E, Clark GM (1999a) A stochastic model of the electrically stimulated auditory nerve: pulse-train response. IEEE Trans Biomed Eng 46:630–637
Bruce IC, White MW, Irlicht LS, O’Leary SJ, Dynes S, Javel E, Clark GM (1999b) A stochastic model of the electrically stimulated auditory nerve: single-pulse response. IEEE Trans Biomed Eng 46:617–629
Brughera AR, Stutman ER, Carney LH, Colburn HS (1996) A model with excitation and inhibition for cells in the medial superior olive. Audit Neurosci 2:219–233
Brughera A, Dunai L, Hartmann WM (2013) Human interaural time difference thresholds for sine tones: the high-frequency limit. J Acoust Soc Am 133:2839–2855
Butler BE, Lomber SG (2013) Functional and structural changes throughout the auditory system following congenital and earlyonset deafness: implications for hearing restoration. Front Syst Neurosci 7:92
Cant NB, Casseday JH (1986) Projections from the anteroventral cochlear nucleus to the lateral and medial superior olivary nuclei. J Comp Neurol 247:457–476
Cant NB, Morest DK (1979) Organization of the neurons in the anterior division of the anteroventral cochlear nucleus of the cat. Light-Microscopic Obs Neurosci 4:1909–1923
Chung Y, Hancock KE, Delgutte B (2014a) Neural coding of interaural time difference in an awake rabbit model of bilateral cochlear implants. Abstr Assoc Res Otolaryngol 36:654
Chung Y, Hancock KE, Nam SI, Delgutte B (2014b) Coding of electric pulse trains presented through cochlear implants in the auditory midbrain of awake rabbit: comparison with anesthetized preparations. J Neurosci Off J Soc Neurosci 34:218–231
Colburn HS, Esquissaud P (1976) An auditory-nerve model for interaural time discrimination of high-frequency complex stimuli. J Acoust Soc Am Supplement:S23
Colburn HS, Chung Y, Zhou Y, Brughera A (2009) Models of brainstem responses to bilateral electrical stimulation. J Assoc Res Otolaryngol 10:91–110
Couchman K, Grothe B, Felmy F (2010) Medial superior olivary neurons receive surprisingly few excitatory and inhibitory inputs with balanced strength and short-term dynamics. J Neurosci Off J Soc Neurosci 30:17111–17121
Day ML, Semple MN (2011) Frequency-dependent interaural delays in the medial superior olive: implications for interaural cochlear delays. J Neurophysiol 106:1985–1999
de Boer E (1969) Encoding of frequency information in the discharge pattern of auditory nerve fibers. Int J Audiol 8:547–556
Dynes SB, Delgutte B (1992) Phase-locking of auditory-nerve discharges to sinusoidal electric stimulation of the cochlea. Hear Res 58:79–90
Eatock RA (2003) Auditory physiology: listening with K+ channels. Curr Biol 13:R767–R769
Goldberg JM, Brown PB (1969) Response of binaural neurons of dog superior olivary complex to dichotic tonal stimuli: some physiological mechanisms of sound localization. J Neurophysiol 32:613–636
Grothe B, Pecka M, Mcalpine D (2010) Mechanisms of sound localization in mammals. Physiol Rev 90:983–1012
Guinan JJ Jr, Norris BE, Guinan SS (1972) Single auditory units in the superior olivary complex. II: locations of unit categories and tonotopic organization. Int J Neurosci 4:147–166
Hamlet WR, Liu YW, Tang ZQ, Lu Y (2014) Interplay between low threshold voltage-gated K(+) channels and synaptic inhibition in neurons of the chicken nucleus laminaris along its frequency axis. Front Neural Circuits 8:51
Hancock KE, Noel V, Ryugo DK, Delgutte B (2010) Neural coding of interaural time differences with bilateral cochlear implants: effects of congenital deafness. J Neurosci 30:14068–14079
Hancock KE, Chung Y, Delgutte B (2012) Neural ITD coding with bilateral cochlear implants: effect of binaurally coherent jitter. J Neurophysiol 108:714–728
Hancock KE, Chung Y, Delgutte B (2013) Congenital and prolonged adult-onset deafness cause distinct degradations in neural ITD coding with bilateral cochlear implants. J Assoc Res Otolaryngol: JARO 14:393–411
Henkel CK, Brunso-Bechtold JK (1990) Dendritic morphology and development in the ferret medial superior olivary nucleus. J Comp Neurol 294:377–388
Javel E, Shepherd RK (2000) Electrical stimulation of the auditory nerve. III. Response initiation sites and temporal fine structure. Hear Res 140:45–76
Javel E, Viemeister NF (2000) Stochastic properties of cat auditory nerve responses to electric and acoustic stimuli and application to intensity discrimination. J Acoust Soc Am 107:908–921
Johnson DH (1980) The relationship between spike rate and synchrony in responses of auditory-nerve fibers to single tones. J Acoust Soc Am 68:1115–1122
Johnston J, Forsythe ID, Kopp-Scheinpflug C (2010) Going native: voltage-gated potassium channels controlling neuronal excitability. J Physiol 588:3187–3200
Joris PX, Carney LH, Smith PH, Yin TC (1994) Enhancement of neural synchronization in the anteroventral cochlear nucleus. I. Responses to tones at the characteristic frequency. J Neurophysiol 71:1022–1036
Kalluri S, Delgutte B (2003) Mathematical models of cochlear nucleus onset neurons: I. Point neuron with many weak synaptic inputs. J Comput Neurosci 14:71–90
Karino S, Smith PH, Yin TC, Joris PX (2011) Axonal branching patterns as sources of delay in the mammalian auditory brainstem: a re-examination. J Neurosci Off J Soc Neurosci 31:3016–3031
Kiang NY, Moxon EC (1972) Physiological considerations in artificial stimulation of the inner ear. Ann Otol Rhinol Laryngol 81:714–730
Kotak VC, Takesian AE, Sanes DH (2008) Hearing loss prevents the maturation of gabaergic transmission in the auditory cortex. Cereb Cortex 18:2098–2108
Kuba H, Yamada R, Fukui I, Ohmori H (2005) Tonotopic specialization of auditory coincidence detection in nucleus laminaris of the chick. J Neurosci Off J Soc Neurosci 25:1924–1934
Kuba H, Ishii TM, Ohmori H (2006) Axonal site of spike initiation enhances auditory coincidence detection. Nature 444:1069–1072
Kuwada S, Yin TC (1983) Binaural interaction in low-frequency neurons in inferior colliculus of the cat. I. Effects of long interaural delays, intensity, and repetition rate on interaural delay function. J Neurophysiol 50:981–999
Kuwada S, Stanford TR, Batra R (1987) Interaural phase-sensitive units in the inferior colliculus of the unanesthetized rabbit: effects of changing frequency. J Neurophysiol 57:1338–1360
Leao RN, Sun H, Svahn K, Berntson A, Youssoufian M, Paolini AG, Fyffe RE, Walmsley B (2006) Topographic organization in the auditory brainstem of juvenile mice is disrupted in congenital deafness. J Physiol 571:563–578
Litovsky RY, Jones GL, Agrawal S, van Hoesel R (2010) Effect of age at onset of deafness on binaural sensitivity in electric hearing in humans. J Acoust Soc Am 127:400–414
Manis PB, Marx SO (1991) Outward currents in isolated ventral cochlear nucleus neurons. J Neurosci 11:2865–2880
Mathews PJ, Jercog PE, Rinzel J, Scott LL, Golding NL (2010) Control of submillisecond synaptic timing in binaural coincidence detectors by K(v)1 channels. Nat Neurosci 13:601–609
Mckinney MF, Delgutte B (1999) A possible neurophysiological basis of the octave enlargement effect. J Acoust Soc Am 106:2679–2692
Miller CA, Robinson BK, Rubinstein JT, Abbas PJ, Runge-Samuelson CL (2001) Auditory nerve responses to monophasic and biphasic electric stimuli. Hear Res 151:79–94
Mountain DC, Anderson D, Bresnahan G, Deligeorges S, Hubbard A, Vajda V (2005) EarLab: large-scale simulation of auditory pathways. In: The mid-winter meeting of the association for research in otolaryngology
Nourski KV, Abbas PJ, Miller CA (2006) Effects of remaining hair cells on cochlear implant function, 15th quarterly progress report, NIH contract N01-DC-2-1005
Ohmori H (2014) Neuronal specializations for the processing of interaural difference cues in the chick. Front Neural Circuits 8:47
Pecka M, Brand A, Behrend O, Grothe B (2008) Interaural time difference processing in the mammalian medial superior olive: the role of glycinergic inhibition. J Neurosci Off J Soc Neurosci 28:6914–6925
Poon BB, Eddington DK, Noel V, Colburn HS (2009) Sensitivity to interaural time difference with bilateral cochlear implants: development over time and effect of interaural electrode spacing. J Acoust Soc Am 126:806–815
Reyes AD, Rubel EW, Spain WJ (1996) In vitro analysis of optimal stimuli for phase-locking and time-delayed modulation of firing in avian nucleus laminaris neurons. J Neurosci Off J Soc Neurosci 16:993–1007
Rothman JS, Manis PB (2003a) Differential expression of three distinct potassium currents in the ventral cochlear nucleus. J Neurophysiol 89:3070–3082
Rothman JS, Manis PB (2003b) The roles potassium currents play in regulating the electrical activity of ventral cochlear nucleus neurons. J Neurophysiol 89:3097–3113
Rothman JS, Young ED, Manis PB (1993) Convergence of auditory nerve fibers onto bushy cells in the ventral cochlear nucleus: implications of a computational model. J Neurophysiol 70:2562–2583
Sachs MB, Abbas PJ (1974) Rate versus level functions for auditorynerve fibers in cats: tone-burst stimuli. J Acoust Soc Am 56:1835–1847
Schroeder MR, Hall JL (1974) Model for mechanical to neural transduction in the auditory receptor. J Acoust Soc Am 55:1055–1060
Scott LL, Mathews PJ, Golding NL (2005) Posthearing developmental refinement of temporal processing in principal neurons of the medial superior olive. J Neurosci Off J Soc Neurosci 25:7887–7895
Scott LL, Hage TA, Golding NL (2007) Weak action potential backpropagation is associated with high-frequency axonal firing capability in principal neurons of the gerbil medial superior olive. J Physiol 583:647–661
Shepherd RK, Hardie NA (2001) Deafness-induced changes in the auditory pathway: implications for cochlear implants. Audiol Neurootol 6:305–318
Shepherd RK, Javel E (1997) Electrical stimulation of the auditory nerve. I. Correlation of physiological responses with cochlear status. Hear Res 108:112–144
Smith PH (1995) Structural and functional differences distinguish principal from nonprincipal cells in the guinea pig MSO slice. J Neurophysiol 73:1653–1667
Smith ZM, Delgutte B (2007) Sensitivity to interaural time differences in the inferior colliculus with bilateral cochlear implants. J Neurosci 27:6740–6750
Smith ZM, Delgutte B (2008) Sensitivity of inferior colliculus neurons to interaural time differences in the envelope versus the fine structure with bilateral cochlear implants. J Neurophysiol 99:2390–2407
Smith DJ, Rubel EW (1979) Organization and development of brain stem auditory nuclei of the chicken: dendritic gradients in nucleus laminaris. J Comp Neurol 186:213–239
Smith PH, Joris PX, Yin TC (1993) Projections of physiologically characterized spherical bushy cell axons from the cochlear nucleus of the cat: evidence for delay lines to the medial superior olive. J Comp Neurol 331:245–260
Smith KR, Saberi K, Hickok G (2007) An event-related fMRI study of auditory motion perception: no evidence for a specialized cortical system. Brain Res 1150:94–99
Snyder R, Leake P, Rebscher S, Beitel R (1995) Temporal resolution of neurons in cat inferior colliculus to intracochlear electrical stimulation: effects of neonatal deafening and chronic stimulation. J Neurophysiol 73:449–467
Svirskis G, Kotak V, Sanes DH, Rinzel J (2004) Sodium along with low-threshold potassium currents enhance coincidence detection of subthreshold noisy signals in MSO neurons. J Neurophysiol 91:2465–2473
Tillein J, Hubka P, Syed E, Hartmann R, Engel AK, Kral A (2010) Cortical representation of interaural time difference in congenital deafness. Cereb Cortex 20:492–506
Trussell LO (1997) Cellular mechanisms for preservation of timing in central auditory pathways. Curr Opin Neurobiol 7:487–492
Trussell LO (1999) Synaptic mechanisms for coding timing in auditory neurons. Annu Rev Physiol 61:477–496
Vale C, Sanes DH (2000) Afferent regulation of inhibitory synaptic transmission in the developing auditory midbrain. J Neurosci Off J Soc Neurosci 20:1912–1921
van Hoesel RJ (2007) Sensitivity to binaural timing in bilateral cochlear implant users. J Acoust Soc Am 121:2192–2206
van Hoesel RJ (2012) Contrasting benefits from contralateral implants and hearing aids in cochlear implant users. Hear Res 288:100–113
von Hehn CA, Bhattacharjee A, Kaczmarek LK (2004) Loss of Kv3.1 tonotopicity and alterations in camp response element-binding protein signaling in central auditory neurons of hearing impaired mice. J Neurosci Off J Soc Neurosci 24:1936–1940
Wang GI, Delgutte B (2012) Sensitivity of cochlear nucleus neurons to spatio-temporal changes in auditory nerve activity. J Neurophysiol 108:3172–3195
Yin TC, Chan JC (1990) Interaural time sensitivity in medial superior olive of cat. J Neurophysiol 64:465–488
Yin TC, Kuwada S, Sujaku Y (1984) Interaural time sensitivity of high-frequency neurons in the inferior colliculus. J Acoust Soc Am 76:1401–1410
Young ED, Sachs MB (1979) Representation of steady-state vowels in the temporal aspects of the discharge patterns of populations of auditory-nerve fibers. J Acoust Soc Am 66:1381–1403
Zhang F, Miller CA, Robinson BK, Abbas PJ, Hu N (2007) Changes across time in spike rate and spike amplitude of auditory nerve fibers stimulated by electric pulse trains. J Assoc Res Otolaryngol 8:356–372
Zhou Y, Carney LH, Colburn HS (2005) A model for interaural time difference sensitivity in the medial superior olive: interaction of excitatory and inhibitory synaptic inputs, channel dynamics, and cellular morphology. J Neurosci 25:3046–3058
Acknowledgments
This work was supported by the National Institutes of Health (Grants R01 DC005775 to B.D. and R01 DC000100 to H.S.C.) and the Hearing Health Foundation (Emerging Research Grant to Y.C.).
Conflict of Interest
None of the authors has conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chung, Y., Delgutte, B. & Colburn, H.S. Modeling Binaural Responses in the Auditory Brainstem to Electric Stimulation of the Auditory Nerve. JARO 16, 135–158 (2015). https://doi.org/10.1007/s10162-014-0492-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10162-014-0492-6