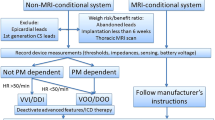
Abstract
Feasibility, safety, and utility of brain MRI for patients with non-MRI-conditioned cardiac implantable electrical devices (CIEDs) remains controversial. While a growing number of studies have shown safe employment in select patients under strict protocols, there is an increasing clinical need for further off-label investigations. To assess the feasibility and utility of brain MRI in neurological and neurosurgical patients with non-MRI-conditioned CIEDs using off-label protocol. We retrospectively evaluated 126 patients with non-MRI-conditioned CIEDs referred to our hospital between 2014 to 2018 for MRI under an IRB-approved protocol. A total of 126 off-label brain MRI scans were performed. The mean age was 67.5 ± 13.0. Seventy percent of scans were performed on female patients. Indications for MRI are neurosurgical (45.2%), neurological (51.6%), and others (3.2%). MRI utilization for tumor cases was highest for tumor cases (68.3%), but employment was valuable for vascular (12.7%), deep brain stimulators (3.2%), and other cases (15.9%). In the tumor category, (37.2%) of the scans were performed for initial diagnosis and pre-surgical planning, (47.7%) for post-intervention evaluation/surveillance, (15.1%) for stereotactic radiosurgery treatment (CyberKnife). No clinical complications were encountered. No functional device complications of the CIED were identified during and after the MRI in 96.9% of the studies. A 49.6% of the off-label brain MRI scans performed led to a clinically significant decision and/or intervention for the patients. A 42.9% of obtained MRI studies did not change the plan of care. A 7.9% of post-scan decision-making data was not available. We demonstrate that off-label brain MRI scans performed on select patients under a strict protocol is feasible, safe, and relevant. Almost 50% of scans provided critical information resulting in clinical intervention of the patients.



Similar content being viewed by others
References
Cadieu R, Peron M, Le Ven F et al (2017) Central nervous system MRI and cardiac implantable electronic devices. J Neuroradiol 44(1):1–9. https://doi.org/10.1016/j.neurad.2016.09.004
Cohen JD, Costa HS, Russo RJ (2012) Determining the risks of magnetic resonance imaging at 1.5 tesla for patients with pacemakers and implantable cardioverter defibrillators. Am J Cardiol 110(11):1631–1636. https://doi.org/10.1016/j.amjcard.2012.07.030
Indik JH, Gimbel JR, Abe H, Alkmim-Teixeira R, Birgersdotter-Green U, Clarke GD, Dickfeld TML, Froelich JW, Grant J, Hayes DL, Heidbuchel H, Idriss SF, Kanal E, Lampert R, Machado CE, Mandrola JM, Nazarian S, Patton KK, Rozner MA, Russo RJ, Shen WK, Shinbane JS, Teo WS, Uribe W, Verma A, Wilkoff BL, Woodard PK (2017) 2017 HRS expert consensus statement on magnetic resonance imaging and radiation exposure in patients with cardiovascular implantable electronic devices. Heart Rhythm 14(7):e97–e153
Nazarian S, Hansford R, Roguin A, Goldsher D, Zviman MM, Lardo AC, Caffo BS, Frick KD, Kraut MA, Kamel IR, Calkins H, Berger RD, Bluemke DA, Halperin HR (2011) A prospective evaluation of a protocol for magnetic resonance imaging of patients with implanted cardiac devices. Ann Intern Med 155(7):415–424. https://doi.org/10.7326/0003-4819-155-7-201110040-00004
Nazarian S, Roguin A, Zviman MM, Lardo AC, Dickfeld TL, Calkins H, Weiss RG, Berger RD, Bluemke DA, Halperin HR (2006) Clinical utility and safety of a protocol for noncardiac and cardiac magnetic resonance imaging of patients with permanent pacemakers and implantable-cardioverter defibrillators at 1.5 tesla. Circulation 114(12):1277–1284. https://doi.org/10.1161/CIRCULATIONAHA.105.607655
Ono M, Suzuki M, Isobe M (2017) Feasibility, safety, and potential demand of emergent brain magnetic resonance imaging of patients with cardiac implantable electronic devices. J Arrhythmia 33(5):455–458
Roguin A (2009) Magnetic resonance imaging in patients with implantable cardioverter-defibrillators and pacemakers. J Am Coll Cardiol 54(6):556–557. https://doi.org/10.1016/j.jacc.2009.04.047
Roguin A, Schwitter J, Vahlhaus C, Lombardi M, Brugada J, Vardas P, Auricchio A, Priori S, Sommer T (2008) Magnetic resonance imaging in individuals with cardiovascular implantable electronic devices. Europace 10(3):336–346. https://doi.org/10.1093/europace/eun021
Shah AD, Patel AU, Knezevic A et al (2017) Clinical performance of magnetic resonance imaging conditional and nonconditional cardiac implantable electronic devices. Pacing Clin Electrophysiol 40(5):467–475
Russo RJ, Costa HS, Silva PD, Anderson JL, Arshad A, Biederman RWW, Boyle NG, Frabizzio JV, Birgersdotter-Green U, Higgins SL, Lampert R, Machado CE, Martin ET, Rivard AL, Rubenstein JC, Schaerf RHM, Schwartz JD, Shah DJ, Tomassoni GF, Tominaga GT, Tonkin AE, Uretsky S, Wolff SD (2017) Assessing the risks associated with MRI in patients with a pacemaker or defibrillator. N Engl J Med 376(8):755–764. https://doi.org/10.1056/NEJMoa1603265
Nazarian S, Beinart R, Halperin HR (2013) Magnetic resonance imaging and implantable devices. Circ Arrhythm Electrophysiol 6(2):419–428. https://doi.org/10.1161/CIRCEP.113.000116
Verma A, Ha ACT, Dennie C, Essebag V, Exner DV, Khan N, Lane C, Leipsic J, Philippon F, Sampaio M, Schieda N, Seifer C, Berthiaume A, Campbell D, Chakraborty S (2014) Canadian Heart Rhythm Society and Canadian Association of Radiologists Consensus Statement on magnetic resonance imaging with cardiac implantable electronic devices. Can J Cardiol 30(10):1131–1141. https://doi.org/10.1016/j.cjca.2014.07.010
Levine GN, Gomes AS, Arai AE et al (2007) Safety of magnetic resonance imaging in patients with cardiovascular devices: an American Heart Association scientific statement from the committee on diagnostic and interventional cardiac catheterization, council on clinical cardiology, and the council on cardiovascular radiology and intervention: endorsed by the American College of Cardiology Foundation, the north American Society for Cardiac Imaging, and the Society for Cardiovascular Magnetic Resonance. Circulation. 116(24):2878–2891
Naehle CP, Strach K, Thomas D, Meyer C, Linhart M, Bitaraf S, Litt H, Schwab JO, Schild H, Sommer T (2009) Magnetic resonance imaging at 1.5-T in patients with implantable cardioverter-defibrillators. J Am Coll Cardiol 54(6):549–555
Gimbel JR, Bailey SM, Tchou PJ, Ruggieri PM, Wilkoff BL (2005) Strategies for the safe magnetic resonance imaging of pacemaker-dependent patients. Pacing Clin Electrophysiol 28(10):1041–1046
Strom JB, Whelan JB, Shen C, Zheng SQ, Mortele KJ, Kramer DB (2017) Safety and utility of magnetic resonance imaging in patients with cardiac implantable electronic devices. Heart Rhythm 14(8):1138–1144
Cronin EM, Mahon N, Wilkoff BL (2012) MRI in patients with cardiac implantable electronic devices. Expert Rev Med Devices 9(2):139–146
Furusho MD, Kusayama MD, Chikata MD et al (2012) Magnetic resonance imaging in a patient with an implantable cardiac defibrillator. J Arrhythmia 28(6):353–355
Nordbeck P, Ertl G, Ritter O (2015) Magnetic resonance imaging safety in pacemaker and implantable cardioverter defibrillator patients: how far have we come? Eur Heart J 36(24):1505–1511
Kraff O, Ladd ME (2016) MR safety update 2015: where do the risks come from? Curr Radiol Rep 4(6):34. https://doi.org/10.1007/s40134-016-0163-y
Yadava M, Nugent M, Krebsbach A, Minnier J, Jessel P, Henrikson CA (2017) Magnetic resonance imaging in patients with cardiac implantable electronic devices: a single-center prospective study. J Interv Card Electrophysiol 50(1):95–104
Miller JD, Nazarian S, Halperin HR (2016) Implantable electronic cardiac devices and compatibility with magnetic resonance imaging. J Am Coll Cardiol 68(14):1590–1598. https://doi.org/10.1016/j.jacc.2016.06.068
van der Graaf AWM, Bhagirath P, Götte MJW (2014) MRI and cardiac implantable electronic devices; current status and required safety conditions. Neth Heart J 22(6):269–276. https://doi.org/10.1007/s12471-014-0544-x
Jung JI (2016) Magnetic resonance imaging for patients with cardiac implantable electronic devices: reduced concerns regarding safety, but scrutiny remains critical. Korean Circ J 46(6):765–767. https://doi.org/10.4070/kcj.2016.46.6.765
Sabzevari K, Oldman J, Herrey AS, Moon JC, Kydd AC, Manisty C (2016) Provision of magnetic resonance imaging for patients with ‘MR-conditional’ cardiac implantable electronic devices: an unmet clinical need. Europace euw063. doi:https://doi.org/10.1093/europace/euw063
Nazarian S, Reynolds MR, Ryan MP, Wolff SD, Mollenkopf SA, Turakhia MP (2016) Utilization and likelihood of radiologic diagnostic imaging in patients with implantable cardiac defibrillators. J Magn Reson Imaging 43(1):115–127. https://doi.org/10.1002/jmri.24971
Funding
During the initial part of the development (initial cohort data collection phase), Dr. Daniel Kramer was supported by a Paul B. Beeson Career Development Awards in Aging Research program (award no. K23AG045963) and the Greenwall Faculty Scholars Program, and he is a consultant to Circulatory Systems Advisory Panel of the Food and Drug Administration.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Daniel Kramer is also a consultant to Circulatory Systems Advisory Panel of the Food and Drug Administration.
Ethical approval
Our study has full Institutional Review Board approval. (IRB was approved by IRB review team at Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA USA).
Informed consent
Patients described in this article provided consent for the use of their medical information and imaging.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mayeku, J., Kramer, D., Mahadevan, A. et al. Feasibility, safety, and utility of brain MRI for patients with non-MRI-conditioned CIED. Neurosurg Rev 43, 1539–1546 (2020). https://doi.org/10.1007/s10143-019-01149-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-019-01149-6