Abstract
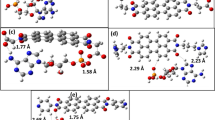
The interaction between one polychlorobiphenyl (3,3′,4,4′,-tetrachlorobiphenyl, coded PCB77) and the four DNA nucleic acid–base is studied by means of quantum mechanics calculations in stacked conformations. It is shown that even if the intermolecular dispersion energy is the largest component of the total interaction energy, some other contributions play a non negligible role. In particular the electrostatic dipole-dipole interaction and the charge transfer from the nucleobase to the PCB are responsible for the relative orientation of the monomers in the complexes. In addition, the charge transfer tends to flatten the PCB, which could therefore intercalate more easily between DNA base pairs. From these seminal results, we predict that PCB could intercalate completely between two base pairs, preferably between Guanine:Cytosine pairs.

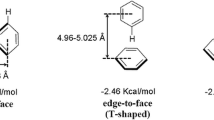
Molecular orbital interaction diagram of stacked PCB77 and Adenine.



Similar content being viewed by others
References
Jimenez B, Wright C, Kelly M, Startin JR (1996) Levels of PCDDs, PCDFs and non-ortho PCBs in dietary supplement fish oil obtained in spain. Chemosphere 32:461–467
Kurokowa Y, Matsueda T, Nakamura M, Takada S, Fukamachi K (1996) Characterization of non-ortho coplanar PCBs, polychlorinated dibenzo-p-dioxines and dibenzofurans in the atmosphere. Chemosphere 32:491–500
Morgan DP, Roan CC (1971) Absorption, storage, and metabolic conversion of ingested DDT and DDT metabolites in man. Arch Environ Health 22:301–308
Kimbrough RD (1995) Polychlorinated biphenyls (PCBs) and human health: an update. Crit Rev Toxicol 25:133–163
Faroon OM, Keith S, Jones D, De Rosa C (2001) Carcinogenic effects of polychlorobiphenyls. Toxicol Ind Health 17:41–62
Safe SH (1994) Polychlorinated biphenyls (PCBs): environnemental impact, biochemical and toxic responses, and implications for risk assessment. Crit Rev Toxicol 24:87–149
Preston BD, Miller JA, Miller EC (1984) Reactions of 2,2′,5,5′-tetrachlorobiphenyl 3,4-oxide with methionine, cysteine and glutathione in relation to the formation of methylthio-metabolites of 2,2′,5,5′-tetrachlorobiphenyl in the rat and mouse. Chem Biol Interact 50:289–312
Narbonne JF, Daubeze M (1980) In vitro binding hexachlorobiphenyl to DNA and proteins. Toxicology 16:173–175
Poland A, Glover E, Kende AS (1976) Stereospecific high binding affinity of 2,3,7,8-teratchlorodibenzo-p-dioxin by cytosols. Evidence that the binding species is a receptor for induction of aryl hydrocarbon hydroxylase. J Biol Chem 251:4936–4946
Cheney BV, Tolly T (1979) Electronic factor affecting receptor binding of binding of dibenzo-p-dioxins and dibenzofurans. Int J Quant Chem 16:87–110
Parthasarathi R, Padmanabhan J, Sbramanian V, Maiti B, Chattaraj PK (2003) J Phys Chem A 107:10346–10352
Yoshimura H, Yoshihara S, Ozawa N, Miki M (1979) Possible correlation between induction modes of hepatic enzymes by PCBs and their toxicity in rats. Ann NY Acad Sci 320:179–192
Jimenez B, Hernandez LM, Eljarrat E, Rivera J, Gonzalez MJ (1996) Level of PCDDs, PCDFs, and non-ortho PCBs in serum samples of non-exposed individuals living in Madrid (Spain). Chemosphere 33:2403–2410
Tanabe S, Kannan N, Wakimoto T, Tatsukawa R (1987) Method for the determination of three toxic nonortho chlorine substituted coplanar PCBs in environmental samples at part-per-trillion levels. Int J Environ Anal Chem 29:199–213
Faqi AS, Dalsenter PR, Merker HJ, Chahoud I (1998) Effects on developmental landmarks and reproductive capability of 3,3′,4,4′-tetrachlorobiphenyl and 3,3′,4,4′-pentachlorobiphenyl in offspring of rats exposed during pregnancy. Hum Exp Toxicol 17:365–372
Gray LE Jr, Wolf C, Mann P, Price M, Cooper RL, Ostby J (1999) Adminstration of potentially antiangenic pesticides and toxic substances (dibutyl- and dielthyl hexyl phtalate, PCB169, and ethane dimethane sulfonate) during sexuel differentiation produces divers profiles of reproductive malformations in the male rate. Toxicol Ind Health 15:94–118
Hong C, Bush B, Xiao J (1992) Coplanar PCBs in fish and mussels from marine and estuarine waters of New York states. Ecotoxicol Environ Safety 23:118–131
Sargent S, Roloff B, Meisner L (1989) In vitro chromosome damage due to PCB interactions. Mutat Res 224:79–88
Wasson JS, Huff JE, Loprieno N (1977) A review of the genetic toxicology of chlorinated dibenzo-p-dioxins. Mutat Res 47:141–160
Poland A, Glover E (1980) 2,3,7,8-tetrachlorodibenzo-p-dioxin: segregation of toxicity with the Ah locus. Mol Pharmacol 17:86–94
Safe S (1986) Comparative toxicology and mechanism of action of polychlorinated dibenzofurans. Annu Rev Pharmacol Toxicol 26:371–399
Swart M, Wijst TV, Guerra CF, Bickelhaupt FM (2007) π-π stacking tackled with density functional theory. J Mol Model 13:1245–1257
Vosko SH, Wilk L, Nusair M (1980) Accurate spin-dependent electron liquid correlation energies for local spin density calculations: A critical analysis. Can J Phys 58:1200–1211
Zimmerli U, Parrinello M, Koumoustsakos P (2004) Dispersion corrections to density functionals for water aromatic interactions. J Chem Phys 120:2693–2699
Grimme S (2004) Accurate description of van der Waals complexes by density functional theory including empirical corrections. J Comput Chem 25:1463–1473
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian03, revision B.05. Gaussian, Inc, Wallingford
Sponer J, Leszczynski J, Hobza P (1996) Nature of nucleic acid − base stacking: nonempirical ab initio and empirical potential characterization of 10 stacked base dimers. Comparison of stacked and H-bonded base pairs. J Phys Chem 100:5590–5596
Hobza P, Sponer J (1999) Structure, energetics, and dynamics of the nucleic acid base pairs: nonempirical Ab initio calculations. Chem Rev 99:3247–3276
Sponer J, Florian J, Ng HL, Sponer JE, Spackova N (2000) Local conformational variations observed in B-DNA crystals do not improve base stacking: computational analysis of base stacking in a d(CATGGGCCCATG)2 B↔A intermediate crystal structure. Nucleic Acids Res 28:4893–4902
Swart M, Ehlers AW, Lammertsma K (2004) Performance of the OPBE exchange-correlation functional. Mol Phys 102:2467–2474
Cassandra DM, Wetmore C, Wetmore SD (2009) Noncovalent interactions involving histidine: the effect of charge on π − π stacking and T-shaped interactions with the DNA nucleobases. J Phys Chem B 113:16046–16058
Rutledge LR, Wheaton CA, Wetmore SD (2007) A computational characterization of the hydrogen-bonding and stacking interactions of hypoxanthine. Phys Chem Chem Phys 9:497–509
Mulliken RS (1955) Electronic population analysis on LCAO MO molecular wave functions I. J Chem Phys 23:1833–1840
Acknowledgments
SA is very thankful to the Algerian government for a bursary. AM and XA thank the Agence National de la Recherche for funding ANR-09-BLAN-0191-01 PhotoBioMet.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 527 kb)
Rights and permissions
About this article
Cite this article
Abtouche, S., Very, T., Monari, A. et al. Insight on the interaction of polychlorobiphenyl with nucleic acid–base. J Mol Model 19, 581–588 (2013). https://doi.org/10.1007/s00894-012-1580-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-012-1580-3