Abstract
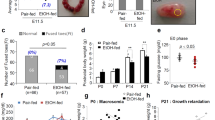
Prenatal alcohol exposure is known to cause fetal growth restriction and disturbances in amino acid bioavailability. Alterations in these parameters can persist into adulthood and low birth weight can lead to altered fetal programming. Glutamine has been associated with the synthesis of other amino acids, an increase in protein synthesis and it is used clinically as a nutrient supplement for low birth weight infants. The aim of this study was to explore the effect of repeated maternal alcohol exposure and l-glutamine supplementation on fetal growth and amino acid bioavailability during the third trimester-equivalent period in an ovine model. Pregnant sheep were randomly assigned to four groups, saline control, alcohol (1.75–2.5 g/kg), glutamine (100 mg/kg, three times daily) or alcohol + glutamine. In this study, a weekend binge drinking model was followed where treatment was done 3 days per week in succession from gestational day (GD) 109–132 (normal term ~147). Maternal alcohol exposure significantly reduced fetal body weight, height, length, thoracic girth and brain weight, and resulted in decreased amino acid bioavailability in fetal plasma and placental fluids. Maternal glutamine supplementation successfully mitigated alcohol-induced fetal growth restriction and improved the bioavailability of glutamine and glutamine-related amino acids such as glycine, arginine, and asparagine in the fetal compartment. All together, these findings show that l-glutamine supplementation enhances amino acid availability in the fetus and prevents alcohol-induced fetal growth restriction.

Similar content being viewed by others
Abbreviations
- FAS:
-
Fetal alcohol syndrome
- FASD:
-
Fetal alcohol spectrum disorders
- GD:
-
Gestational day
- IUGR:
-
Intra uterine growth restriction
References
Abel EL, Dintcheff BA (1978) Effects of prenatal alcohol exposure on growth and development in rats. J Pharmacol Exp Ther 207(3):916–921
Als H, Tronick E, Adamson L, Brazelton TB (1976) The behavior of the full-term but underweight newborn infant. Developmental medicine and child neurology 18(5):590–602
Assaad H, Zhou L, Carroll RJ, Wu G (2014) Rapid publication-ready MS-Word tables for one-way ANOVA. Springerplus 3: 474
Assaad H, Hou YQ, Zhou L, Carroll RJ, Wu G (2015) Rapid publication-ready MS-Word tables for two-way ANOVA. Springerplus 4: 33
Barker DJ (1994) Maternal and fetal origins of coronary heart disease. J Royal Coll Phys Lond 28(6):544–551
Caetano R, Ramisetty-Mikler S, Floyd LR, McGrath C (2006) The epidemiology of drinking among women of child-bearing age. Alcohol Clin Exp Res 30(6):1023–1030
Coles CD, Brown RT, Smith IE, Platzman KA, Erickson S, Falek A (1991) Effects of prenatal alcohol exposure at school age. I. Physical and cognitive development. Neurotoxicol Teratol 13(4):357–367
Cooper C, Westlake S, Harvey N, Dennison E (2009) Developmental origins of osteoporotic fracture. Adv Exp Med Biol 639:217–236. doi:10.1007/978-1-4020-8749-3_16
Cornelius MD, Goldshmidt L, Taylor PM, Day NL (1999) Prenatal alcohol use among teenagers: effects on neonatal outcomes. Alcohol Clin Experiment Res 23(7):1238–1244
Coster J, McCauley R, Hall J (2004) Glutamine: metabolism and application in nutrition support. Asia Pac J Clin Nutr 13(1):25–31
Day NL, Richardson G, Robles N, Sambamoorthi U, Taylor P, Scher M, Stoffer D, Jasperse D, Cornelius M (1990) Effect of prenatal alcohol exposure on growth and morphology of offspring at 8 months of age. Pediatrics 85(5):748–752
Day NL, Zuo Y, Richardson GA, Goldschmidt L, Larkby CA, Cornelius MD (1999) Prenatal alcohol use and offspring size at 10 years of age. Alcohol Clin Experiment Res 23(5):863–869
Day NL, Leech SL, Richardson GA, Cornelius MD, Robles N, Larkby C (2002) Prenatal alcohol exposure predicts continued deficits in offspring size at 14 years of age. Alcohol Clin Experiment Res 26(10):1584–1591. doi:10.1097/01.ALC.0000034036.75248.D9
Dobbing J, Sands J (1979) Comparative aspects of the brain growth spurt. Early Hum Dev 3(1):79–83
Ehrenkranz RA, Das A, Wrage LA, Poindexter BB, Higgins RD, Stoll BJ, Oh W (2011) Early nutrition mediates the influence of severity of illness on extremely LBW infants. Pediatr Res 69(6):522–529. doi:10.1203/PDR.0b013e318217f4f1
Garlick PJ (2001) Assessment of the safety of glutamine and other amino acids. J Nutr 131(9 Suppl):2556S–2561S
Hales CN, Ozanne SE (2003) For debate: Fetal and early postnatal growth restriction lead to diabetes, the metabolic syndrome and renal failure. Diabetologia 46(7):1013–1019. doi:10.1007/s00125-003-1131-7
Haynes TE, Li P, Li X, Shimotori K, Sato H, Flynn NE, Wang J, Knabe DA, Wu G (2009) L-Glutamine or L-alanyl-L-glutamine prevents oxidant- or endotoxin-induced death of neonatal enterocytes. Amino Acids 37(1):131–142. doi:10.1007/s00726-009-0243-x
Hutchinson EA, De Luca CR, Doyle LW, Roberts G, Anderson PJ (2013) School-age outcomes of extremely preterm or extremely low birth weight children. Pediatrics 131(4):e1053–e1061. doi:10.1542/peds.2012-2311
Karl PI, Kwun R, Slonim A, Fisher SE (1995) Ethanol elevates fetal serum glutamate levels in the rat. Alcohol Clin Experiment Res 19(1):177–181
Kwon H, Spencer TE, Bazer FW, Wu G (2003) Developmental changes of amino acids in ovine fetal fluids. Biol Reprod 68(5):1813–1820. doi:10.1095/biolreprod.102.012971
Lassala A, Bazer FW, Cudd TA, Datta S, Keisler DH, Satterfield MC, Spencer TE, Wu G (2011) Parenteral administration of L-arginine enhances fetal survival and growth in sheep carrying multiple fetuses. J Nutr 141(5):849–855. doi:10.3945/jn.111.138172
Lucas JS, Inskip HM, Godfrey KM, Foreman CT, Warner JO, Gregson RK, Clough JB (2004) Small size at birth and greater postnatal weight gain: relationships to diminished infant lung function. Am J Respir Crit Care Med 170(5):534–540. doi:10.1164/rccm.200311-1583OC
Marquis SM, Leichter J, Lee M (1984) Plasma amino acids and glucose levels in the rat fetus and dam after chronic maternal alcohol consumption. Biol Neonate 46(1):36–43
Mates JM, Perez-Gomez C, Nunez de Castro I, Asenjo M, Marquez J (2002a) Glutamine and its relationship with intracellular redox status, oxidative stress and cell proliferation/death. Int J Biochem Cell Biol 34(5):439–458
Mates JM, Perez-Gomez C, Nunez de Castro I, Asenjo M, Marquez J (2002b) Glutamine and its relationship with intracellular redox status, oxidative stress and cell proliferation/death. Int J Biochem Cell Biol 34(5):439–458
Ouellette EM, Rosett HL, Rosman NP, Weiner L (1977) Adverse effects on offspring of maternal alcohol abuse during pregnancy. N Engl J Med 297(10):528–530. doi:10.1056/NEJM197709082971003
Padmanabhan R, Ibrahim A, Bener A (2002) Effect of maternal methionine pre-treatment on alcohol-induced exencephaly and axial skeletal dysmorphogenesis in mouse fetuses. Drug Alcohol Depend 65(3):263–281
Poindexter BB, Ehrenkranz RA, Stoll BJ, Koch MA, Wright LL, Oh W, Papile LA, Bauer CR, Carlo WA, Donovan EF, Fanaroff AA, Korones SB, Laptook AR, Shankaran S, Stevenson DK, Tyson JE, Lemons JA (2003) Effect of parenteral glutamine supplementation on plasma amino acid concentrations in extremely low-birth-weight infants. Am J Clin Nutr 77(3):737–743
Probyn ME, Zanini S, Ward LC, Bertram JF, Moritz KM (2012) A rodent model of low- to moderate-dose ethanol consumption during pregnancy: patterns of ethanol consumption and effects on fetal and offspring growth. Reprod Fertil Dev 24(6):859–870. doi:10.1071/RD11200
Ramadoss J, Hogan HA, Given JC, West JR, Cudd TA (2006) Binge alcohol exposure during all three trimesters alters bone strength and growth in fetal sheep. Alcohol 38(3):185–192
Ramadoss J, Wu G, Cudd TA (2008) Chronic binge ethanol-mediated acidemia reduces availability of glutamine and related amino acids in maternal plasma of pregnant sheep. Alcohol 42(8):657–666. doi:10.1016/j.alcohol.2008.08.008
Rezaei R, Knabe DA, Tekwe CD, Dahanayaka S, Ficken MD, Fielder SE, Eide SJ, Lovering SL, Wu G (2013) Dietary supplementation with monosodium glutamate is safe and improves growth performance in postweaning pigs. Amino Acids 44:911–923. doi:10.1007/s00726-012-1420-x
Riley EP, Infante MA, Warren KR (2011) Fetal alcohol spectrum disorders: an overview. Neuropsychol Rev 21(2):73–80. doi:10.1007/s11065-011-9166-x
Rosett HL, Weiner L, Lee A, Zuckerman B, Dooling E, Oppenheimer E (1983) Patterns of alcohol consumption and fetal development. Obstet Gynecol 61(5):539–546
Sawant OB, Lunde ER, Washburn SE, Chen WJ, Goodlett CR, Cudd TA (2013a) Different patterns of regional Purkinje cell loss in the cerebellar vermis as a function of the timing of prenatal ethanol exposure in an ovine model. Neurotox Teratol 35:7–13. doi:10.1016/j.ntt.2012.11.001
Sawant OB, Ramadoss J, Hogan HA, Washburn SE (2013b) The role of acidemia in maternal binge alcohol-induced alterations in fetal bone functional properties. Alcohol Clin Experiment Res 37(9):1476–1482. doi:10.1111/acer.12118
Sawant OB, Ramadoss J, Hankins GD, Wu G, Washburn SE (2014) Effects of L-glutamine supplementation on maternal and fetal hemodynamics in gestating ewes exposed to alcohol. Amino Acids 46:1981–1996. doi:10.1007/s00726-014-1751-x
Schenker S, Becker HC, Randall CL, Phillips DK, Baskin GS, Henderson GI (1990) Fetal alcohol syndrome: current status of pathogenesis. Alcohol Clin Exp Res 14(5):635–647
Scott MN, Taylor HG, Fristad MA, Klein N, Espy KA, Minich N, Hack M (2012) Behavior disorders in extremely preterm/extremely low birth weight children in kindergarten. J Dev Behav Pediatr 33(3):202–213. doi:10.1097/DBP.0b013e3182475287
Smith IE, Coles CD, Lancaster J, Fernhoff PM, Falek A (1986) The effect of volume and duration of prenatal ethanol exposure on neonatal physical and behavioral development. Neurobehav Toxicol Teratol 8(4):375–381
Spohr HL, Willms J, Steinhausen HC (1993) Prenatal alcohol exposure and long-term developmental consequences. Lancet 341(8850):907–910
Streissguth AP, Martin DC, Martin JC, Barr HM (1981) The Seattle longitudinal prospective study on alcohol and pregnancy. Neurobehav Toxicol Teratol 3(2):223–233
Subramanian MG (1992) Lactation and prolactin release in foster dams suckling prenatally ethanol exposed pups. Alcohol Clin Exp Res 16(5):891–894
Subramanian K, Naik VD, Sathishkumar K, Sawant OB, Washburn SE, Wu G, Yallampalli C, Saade GR, Hankins GD, Ramadoss J (2014a) Interactive effects of in vitro binge-like alcohol and ATP on umbilical endothelial nitric oxide synthase post-translational modifications and redox modulation. Reprod Toxicol 43:94–101. doi:10.1016/j.reprotox.2013.11.006
Subramanian K, Naik VD, Sathishkumar K, Yallampalli C, Saade GR, Hankins GD, Ramadoss J (2014b) Chronic binge alcohol exposure during pregnancy impairs rat maternal uterine vascular function. Alcohol Clin Exp Res 38(7):1832–1838. doi:10.1111/acer.12431
Susser E, Neugebauer R, Hoek HW, Brown AS, Lin S, Labovitz D, Gorman JM (1996) Schizophrenia after prenatal famine. Further evidence. Arch Gen Psychiatry 53(1):25–31
Tagare A, Chaudhari S, Kadam S, Vaidya U, Pandit A, Sayyad MG (2013) Mortality and morbidity in extremely low birth weight (ELBW) infants in a neonatal intensive care unit. Indian J Pediatr 80(1):16–20. doi:10.1007/s12098-012-0818-5
van den Berg A, van Elburg RM, Westerbeek EA, van der Linde EG, Knol J, Twisk JW, Fetter WP (2007) The effect of glutamine-enriched enteral nutrition on intestinal microflora in very low birth weight infants: a randomized controlled trial. Clin Nutr 26(4):430–439. doi:10.1016/j.clnu.2007.03.002
Vohr BR, Wright LL, Dusick AM, Mele L, Verter J, Steichen JJ, Simon NP, Wilson DC, Broyles S, Bauer CR, Delaney-Black V, Yolton KA, Fleisher BE, Papile LA, Kaplan MD (2000) Neurodevelopmental and functional outcomes of extremely low birth weight infants in the National Institute of Child Health and Human Development Neonatal Research Network, 1993–1994. Pediatrics 105(6):1216–1226
Wang J, Chen L, Li P, Li X, Zhou H, Wang F, Li D, Yin Y, Wu G (2008) Gene expression is altered in piglet small intestine by weaning and dietary glutamine supplementation. J Nutr 138(6):1025–1032
Wang Y, Tao YX, Cai W, Tang QY, Feng Y, Wu J (2010) Protective effect of parenteral glutamine supplementation on hepatic function in very low birth weight infants. Clin Nutr 29(3):307–311. doi:10.1016/j.clnu.2010.03.009
Warren KR, Calhoun FJ, May PA, Viljoen DL, Li TK, Tanaka H, Marinicheva GS, Robinson LK, Mundle G (2001) Fetal alcohol syndrome: an international perspective. Alcohol Clin Exp Res 25 (5 Suppl): 202S–206S
Washburn SE, Sawant OB, Lunde ER, Wu G, Cudd TA (2013) Acute alcohol exposure, acidemia or glutamine administration impacts amino acid homeostasis in ovine maternal and fetal plasma. Amino Acids 45:543–554. doi:10.1007/s00726-012-1453-1
Washburn SE, Ramadoss J, Chen WJ, Cudd TA (2014) Effects of all three trimester moderate binge alcohol exposure on the foetal hippocampal formation and olfactory bulb. Brain injury doi:10.3109/02699052.2014.947629
Weinberg J (1985) Effects of ethanol and maternal nutritional status on fetal development. Alcohol Clin Exp Res 9:49–55
Woodall SM, Johnston BM, Breier BH, Gluckman PD (1996) Chronic maternal undernutrition in the rat leads to delayed postnatal growth and elevated blood pressure of offspring. Pediatr Res 40(3):438–443. doi:10.1203/00006450-199609000-00012
Wu G (2009) Amino acids: metabolism, functions, and nutrition. Amino Acids 37(1):1–17. doi:10.1007/s00726-009-0269-0
Wu G (2010) Functional amino acids in growth, reproduction, and health. Adv Nutr 1:31–37. doi:10.3945/an.110.1008
Wu G (2013) Functional amino acids in nutrition and health. Amino Acids 45:407–411
Wu G (2014) Dietary requirements of synthesizable amino acids by animals: a paradigm shift in protein nutrition. J Anim Sci Biotechnol 5:34
Wu G, Meier SA, Knabe DA (1996) Dietary glutamine supplementation prevents jejunal atrophy in weaned pigs. J Nutr 126(10):2578–2584
Wu G, Bazer FW, Cudd TA, Meininger CJ, Spencer TE (2004a) Maternal nutrition and fetal development. J Nutr 134(9):2169–2172
Wu G, Fang YZ, Yang S, Lupton JR, Turner ND (2004b) Glutathione metabolism and its implications for health. J Nutr 134(3):489–492
Wu G, Bazer FW, Burghardt RC, Johnson GA, Kim SW, Knabe DA, Li P, Li X, McKnight JR, Satterfield MC, Spencer TE (2011) Proline and hydroxyproline metabolism: implications for animal and human nutrition. Amino Acids 40(4):1053–1063. doi:10.1007/s00726-010-0715-z
Wu G, Bazer FW, Dai ZL, Li DF, Wang JJ, Wu ZL (2014) Amino acid nutrition in animals: protein synthesis and beyond. Annu Rev Anim Biosci 2:387–417
Acknowledgments
We would like to acknowledge the important role of the late Dr. Timothy Cudd in starting this study and making significant contributions. We would like to thank Emilie Lunde, Brittney Kramer, Colt Sharpton and Sudath Dahanayaka for their assistance in this study. This research was supported by NIAAA grant AA010940 and AA18166-2 (SW).
Conflict of interest
The authors have no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sawant, O.B., Wu, G. & Washburn, S.E. Maternal l-glutamine supplementation prevents prenatal alcohol exposure-induced fetal growth restriction in an ovine model. Amino Acids 47, 1183–1192 (2015). https://doi.org/10.1007/s00726-015-1945-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-015-1945-x