Abstract
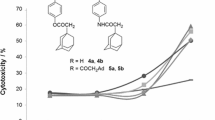
The cytotoxic effects of novel racemic and optically active constrained N-phosphonoalkyl bicyclic β-amino acids were tested against a panel of human tumor cell lines. All of the compounds investigated exhibited different concentration-dependent antiproliferative effects against the HT-29, MDA-MB-231, HepG2 and HeLa cell lines after 24 h treatment. The most sensitive cells were the HeLa cells at various concentrations of the four compounds tested. The aminophosphonate 3 exerted the most pronounced antiproliferative effect against the HeLa cells (inhibition of the cell vitality up to 70% at 0.5 mg/ml) and was not toxic to the normal Lep3 cells at lower concentration. Furthermore, the N-phosphonophenyl derivatives 1 and 2 displayed antiproliferative effect against mainly the MDA-MB-231 tumour cells at higher concentration.







Similar content being viewed by others
References
Akkari R, Calmès M, Martinez J (2004a) Toward Diels–Alder reactions on a solid support using polymer bound N-substituted 3-hydroxy-4,4-dimethyl-2-pyrrolidinone acrylate derivatives. Eur J Org Chem 11:2441–2450
Akkari R, Calmès M, Escale F, Iapichella J, Rolland M, Martinez J (2004b) (R)- or (S)-4-(3-hydroxy-4,4-dimethyl-2-oxopyrrolidin-1-yl)benzoic acid as a new chiral auxiliary for solid phase asymmetric Diels–Alder reactions. Tetrahedron Asymmetr 15:2515–2525
Arad D, Kende AS, Lan J (2002) Regiospecific synthesis of a bridgehead-functionalized bicyclo[2.2.2]octenone. Tetrahedron Lett 43:5237–5239
Calmès M, Didierjean C, Martinez J, Songis O (2005) (R)-(4-(Benzyloxycarbonylphenyl)-3-hydroxy-4,4-dimethyl-2-pyrrolidinone)acrylate derivative as a chiral dienophile for the synthesis of enantiopure 2-aminocyclohexane carboxylic acids. Tetrahedron Asymmetr 15:2173–2178
Carruthers W (1990) Cycloaddition reaction in organic synthesis, tetrahedron organic chemistry, series vol 8. Pergamon Press, Elmsford, NY
Fringuelli F, Tatichi A (2002) The Diels–Alder reaction: selected practical methods. Wiley, New York
Fülöp F (2001) The chemistry of 2-aminocycloalkanecarboxylic acids. Chem Rev 101:2181–2204
Gademann K, Hintermann T, Scheiber JV (1999) Beta-peptides: twisting and turning. Current Med Chem 6:905–925
Hirschmann R, Smith AB III, Taylor CM, Benkovic PA, Taylor SD, Yager KM, Sprengler PA, Benkovic SJ (1994) Peptide synthesis catalyzed by an antibody containing a binding site for variable amino acids. Science 265:234–237
Kabachnik M, Minaeva L, Beletskaya I (2009) Synthesis of novel α-aminophosphonates containing adamantyl fragment. Synthesis 14:2357–2360
Kafarski P, Lejczak B (2000) Aminophosphonic and aminophosphinic acids. In: Chemistry and biological activity. Wiley, New york, p 407
Kafarski P, Lejczak B (2001) Aminophosphonic acids of potential medical importance. Curr Med Chem Anti-Cancer Agents 1:301–312
Naydenova E, Todorov P, Topashka-Ancheva M, Momekov G, Yordanova T, Konstantinov S, Troev K (2008) Novel N-(phosphonomethyl) glycine derivatives. Design, characterization and biological activity. Eur J Med Chem 43(6):1199–1205
Naydenova E, Todorov P, Troev K (2010) Resent synthesis of aminophosphonic acids as potential biological importance. Amino Acids 38(1):23–30
Orsini F, Sello G, Sisti M (2010) Aminophosphonic acids and derivatives. Synthesis and biological applications. Curr Med Chem 17:264–289
Park K-H, Kurth MJ (2002) Cyclic amino acid derivatives. Tetrahedron 58:8629–8659
Reynolds RC, Johnson CA, Piper JR, Sirotnak FM (2001) Synthesis and antifolate evaluation of the aminopterin analogue with a bicyclo. Eur J Med Chem 36:237–242
Roberts JD, Moreland WT, Frazer W (1953) Synthesis of some 4-substituted bicyclo[2.2.2]octane carboxylic acids. J Am Chem Soc 75:637–640
Rozenfeld R, Iturrioz X, Okada M, Maigret B, Llorents-Cortes C (2003) Contribution of molecular modeling and site-directed mutagenesis to the identification of a new residue, glutamate 215, involved in the exopeptidase specificity of aminopeptidase A. Biochemistry 42:14785–14793
Smith PW, Trivedi N, Howes PD, Sollis SL, Rahim G, Bethell RC, Lynn S (1999) Synthesis of a tetrasubstituted bicyclo [2.2.2] octane as a potential inhibitor of influenza virus sialidase. Bioorg Med Chem Lett 9:611–614
Songis O, Didierjean C, Laurent C, Martinez J, Calmès M (2007) Asymmetric Diels–Alder cycloaddition of 1-aminocyclohexadiene to chiral acrylate: synthesis of enantiopure bridgehead-aminobicyclo[2.2.2]octane-2-carboxylic acid derivatives. Eur J Org Chem 3166–3172
Todorov PT, Pavlov ND, Shivachev BL, Petrova RN, Martinez J, Naydenova ED, Calmes M (2011) Synthesis of new racemic and optically active N-phosphonylalkyl bicyclic β-amino acids via Kabachnik–Fields reaction as potential biologically active compounds. Heteroatom Chem. doi:10.1002/hc.20759
Troev KD (2006) Chemistry and application of H-phosphonates. Elsevier, Amsterdam
Wang B, Miao ZW, Wang J, Chen RY, Zhang XD (2008) Synthesis and biological evaluation of novel naphthoquinone fused cyclic aminoalkylphosphonates and aminoalkylphosphonic monoester. Amino Acids 35:463–468
Widianti T, Hiraga Y, Kojima S, Abe M (2010) Novel cyclic β-aminophosphonate derivatives as efficient organocatalysts for the asymmetric Michael addition reactions of ketones to nitrostyrenes. Tetrahedron Asymmetr 21:1861–1868
Wynn CM, Vaughan WR (1968) Stereochemistry of pyrrolidine addition to bicy-clo[2.2.2]oct-2-ene-2-carbonitrile. J Org Chem 33:2371–2374
Zand R, Sellinger OZ, Water R, Harris R (1974) α-Aminocyclic and bicyclic alkane carboxylic acids: differential effects on selected amino acids of rat brain cortex. J Neurochem 23:1201–1206
Acknowledgments
The study was supported by Grant DPOSTDOC 02/3 of the National Research Fund, Sofia, Bulgaria.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Todorov, P.T., Wesselinova, D.W., Pavlov, N.D. et al. Cytotoxic activity of new racemic and optically active N-phosphonoalkyl bicyclic β-amino acids against human malignant cell lines. Amino Acids 43, 1445–1450 (2012). https://doi.org/10.1007/s00726-012-1217-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-012-1217-y