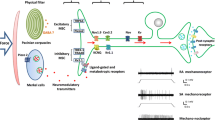
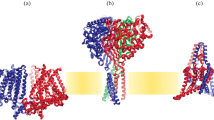
Abstract
Our ability to sense mechanical cues from our environment depend on the capacity of molecular sensor capable of converting mechanical energy into biochemical or electrical signals. This process, termed mechanotransduction, relies on the activity of mechanosensitive ion channels (MSCs) that are expressed in most tissues, including cells of the inner and outer ear, sensory and sympathetic neurons, and vascular cells. However, the precise role these channels play in the physiology of the cells and organs, where they are expressed is not completely understood. In this review, we will explore some of the recent findings on the role of MSCs to our sense of mechanical pain.




Similar content being viewed by others
References
Basu D, Haswell ES (2017) Plant mechanosensitive ion channels: an ocean of possibilities. Curr Opin Plant Biol 40:43–48. https://doi.org/10.1016/j.pbi.2017.07.002
Beaulieu-Laroche L, Christin M, Donoghue A, Agosti F, Yousefpour N, Petitjean H, Davidova A, Stanton C, Khan U, Dietz C, Faure E, Fatima T, MacPherson A, Mouchbahani-Constance S, Bisson DG, Haglund L, Ouellet JA, Stone LS, Samson J, Smith MJ, Ask K, Ribeiro-da-Silva A, Blunck R, Poole K, Bourinet E, Sharif-Naeini R (2020) TACAN is an ion channel involved in sensing mechanical pain. Cell. https://doi.org/10.1016/j.cell.2020.01.033
Castellanos A, Pujol-Coma A, Andres-Bilbe A, Negm A, Callejo G, Soto D, Noel J, Comes N, Gasull X (2020) TRESK background K(+) channel deletion selectively uncovers enhanced mechanical and cold sensitivity. J Physiol 598(5):1017–1038. https://doi.org/10.1113/JP279203
Caterina MJ, Leffler A, Malmberg AB, Martin WJ, Trafton J, Petersen-Zeitz KR, Koltzenburg M, Basbaum AI, Julius D (2000) Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 288(5464):306–313. https://doi.org/10.1126/science.288.5464.306
Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389(6653):816–824. https://doi.org/10.1038/39807
Chalfie M (2009) Neurosensory mechanotransduction. Nat Rev Mol Cell Biol 10(1):44–52. https://doi.org/10.1038/nrm2595
Chesler AT, Szczot M, Bharucha-Goebel D, Ceko M, Donkervoort S, Laubacher C, Hayes LH, Alter K, Zampieri C, Stanley C, Innes AM, Mah JK, Grosmann CM, Bradley N, Nguyen D, Foley AR, Le Pichon CE, Bonnemann CG (2016) The role of PIEZO2 in human mechanosensation. N Engl J Med 375(14):1355–1364. https://doi.org/10.1056/NEJMoa1602812
Cho H, Koo JY, Kim S, Park SP, Yang Y, Oh U (2006) A novel mechanosensitive channel identified in sensory neurons. Eur J Neurosci 23(10):2543–2550. https://doi.org/10.1111/j.1460-9568.2006.04802.x
Cho H, Shin J, Shin CY, Lee SY, Oh U (2002) Mechanosensitive ion channels in cultured sensory neurons of neonatal rats. J Neurosci 22(4):1238–1247
Christensen AP, Corey DP (2007) TRP channels in mechanosensation: direct or indirect activation? Nat Rev Neurosci 8(7):510–521. https://doi.org/10.1038/nrn2149
Coste B, Mathur J, Schmidt M, Earley TJ, Ranade S, Petrus MJ, Dubin AE, Patapoutian A (2010) Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science 330(6000):55–60. https://doi.org/10.1126/science.1193270
Di Castro A, Drew LJ, Wood JN, Cesare P (2006) Modulation of sensory neuron mechanotransduction by PKC- and nerve growth factor-dependent pathways. Proc Natl Acad Sci USA 103(12):4699–4704. https://doi.org/10.1073/pnas.0508005103
Drew LJ, Rohrer DK, Price MP, Blaver KE, Cockayne DA, Cesare P, Wood JN (2004) Acid-sensing ion channels ASIC2 and ASIC3 do not contribute to mechanically activated currents in mammalian sensory neurones. J Physiol 556(Pt 3):691–710. https://doi.org/10.1113/jphysiol.2003.058693
Drew LJ, Rugiero F, Cesare P, Gale JE, Abrahamsen B, Bowden S, Heinzmann S, Robinson M, Brust A, Colless B, Lewis RJ, Wood JN (2007) High-threshold mechanosensitive ion channels blocked by a novel conopeptide mediate pressure-evoked pain. PLoS ONE 2(6):e515. https://doi.org/10.1371/journal.pone.0000515
Drew LJ, Wood JN, Cesare P (2002) Distinct mechanosensitive properties of capsaicin-sensitive and -insensitive sensory neurons. J Neurosci 22(12):228
Dubin AE, Patapoutian A (2010) Nociceptors: the sensors of the pain pathway. J Clin Investig 120(11):3760–3772. https://doi.org/10.1172/JCI42843
Dubin AE, Schmidt M, Mathur J, Petrus MJ, Xiao B, Coste B, Patapoutian A (2012a) Inflammatory signals enhance piezo2-mediated mechanosensitive currents. Cell Rep 2(3):511–517. https://doi.org/10.1016/j.celrep.2012.07.014
Dubin AE, Schmidt M, Mathur J, Petrus MJ, Xiao B, Coste B, Patapoutian A (2012b) Inflammatory signals enhance Piezo2-mediated mechanosensitive currents. Cell Rep. https://doi.org/10.1016/j.celrep.2012.07.014
French AS, Torkkeli PH (2004) Mechanotransduction in spider slit sensilla. Can J Physiol Pharmacol 82(8–9):541–548. https://doi.org/10.1139/y04-031
French AS, Torkkeli PH (2007) Mechanosensitive ion channels of spiders: mechanical coupling, electrophysiology, and synaptic modulation. Curr Top Membr 59:1–20. https://doi.org/10.1016/S1063-5823(06)59001-5
French AS, Torkkeli PH, Seyfarth EA (2002) From stress and strain to spikes: mechanotransduction in spider slit sensilla. J Comp Phys A Neuroethol Sens Neural Behav Physiol 188(10):739–752. https://doi.org/10.1007/s00359-002-0363-1
Gingl E, Burger AM, Barth FG (2006) Intracellular recording from a spider vibration receptor. J Comp Phys A Neuroethol Sens Neural Behav Physiol 192(5):551–558. https://doi.org/10.1007/s00359-005-0092-3
Goodman MB (2006) Mechanosensation. WormBook. https://doi.org/10.1895/wormbook.1.62.1
Hamilton ES, Schlegel AM, Haswell ES (2015) United in diversity: mechanosensitive ion channels in plants. Annu Rev Plant Biol 66:113–137. https://doi.org/10.1146/annurev-arplant-043014-114700
Hao J, Delmas P (2010) Multiple desensitization mechanisms of mechanotransducer channels shape firing of mechanosensory neurons. J Neurosci 30(40):13384–13395. https://doi.org/10.1523/jneurosci.2926-10.2010
Hao J, Delmas P (2011) Recording of mechanosensitive currents using piezoelectrically driven mechanostimulator. Nat Protoc 6(7):979–990. https://doi.org/10.1038/nprot.2011.343
Hao J, Padilla F, Dandonneau M, Lavebratt C, Lesage F, Noel J, Delmas P (2013) Kv1.1 channels act as mechanical brake in the senses of touch and pain. Neuron 77(5):899–914. https://doi.org/10.1016/j.neuron.2012.12.035
He BH, Christin M, Mouchbahani-Constance S, Davidova A, Sharif-Naeini R (2017) Mechanosensitive ion channels in articular nociceptors drive mechanical allodynia in osteoarthritis. Osteoarthr Cartil 25(12):2091–2099. https://doi.org/10.1016/j.joca.2017.08.012
Hoger U, Torkkeli PH, Seyfarth EA, French AS (1997) Ionic selectivity of mechanically activated channels in spider mechanoreceptor neurons. J Neurophysiol 78(4):2079–2085. https://doi.org/10.1152/jn.1997.78.4.2079
Hu J, Lewin GR (2006) Mechanosensitive currents in the neurites of cultured mouse sensory neurones. J Physiol 577(Pt 3):815–828. https://doi.org/10.1113/jphysiol.2006.117648
Hu J, Milenkovic N, Lewin GR (2006) The high threshold mechanotransducer: a status report. Pain 120(1–2):3–7. https://doi.org/10.1016/j.pain.2005.11.002
Jaffe MJ, Leopold AC, Staples RC (2002) Thigmo responses in plants and fungi. Am J Bot 89(3):375–382. https://doi.org/10.3732/ajb.89.3.375
Juusola M, French AS (1995) Recording from cuticular mechanoreceptors during mechanical stimulation. Pflugers Arch 431(1):125–128. https://doi.org/10.1007/bf00374385
Juusola M, Seyfarth EA, French AS (1994) Sodium-dependent receptor current in a new mechanoreceptor preparation. J Neurophysiol 72(6):3026–3028. https://doi.org/10.1152/jn.1994.72.6.3026
Kim SE, Coste B, Chadha A, Cook B, Patapoutian A (2012) The role of Drosophila Piezo in mechanical nociception. Nature 483(7388):209–212. https://doi.org/10.1038/nature10801
Kloda A, Martinac B (2002) Common evolutionary origins of mechanosensitive ion channels in Archaea, Bacteria and cell-walled Eukarya. Archaea 1(1):35–44. https://doi.org/10.1155/2002/419261
Kung C (2005) A possible unifying principle for mechanosensation. Nature 436(7051):647–654. https://doi.org/10.1038/nature03896
Le Dain AC, Saint N, Kloda A, Ghazi A, Martinac B (1998) Mechanosensitive ion channels of the archaeon Haloferax volcanii. J Biol Chem 273(20):12116–12119. https://doi.org/10.1074/jbc.273.20.12116
Levina N, Totemeyer S, Stokes NR, Louis P, Jones MA, Booth IR (1999) Protection of Escherichia coli cells against extreme turgor by activation of MscS and MscL mechanosensitive channels: identification of genes required for MscS activity. EMBO J 18(7):1730–1737. https://doi.org/10.1093/emboj/18.7.1730
Lewin GR, Moshourab R (2004) Mechanosensation and pain. J Neurobiol 61(1):30–44. https://doi.org/10.1002/neu.20078
Martinac B, Buechner M, Delcour AH, Adler J, Kung C (1987) Pressure-sensitive ion channel in Escherichia coli. Proc Natl Acad Sci USA 84(8):2297–2301. https://doi.org/10.1073/pnas.84.8.2297
McCarter GC, Reichling DB, Levine JD (1999) Mechanical transduction by rat dorsal root ganglion neurons in vitro. Neurosci Lett 273(3):179–182
McIver SB (1975) Structure of cuticular mechanoreceptors of arthropods. Annu Rev Entomol 20:381–397. https://doi.org/10.1146/annurev.en.20.010175.002121
McQueen DS, Iggo A, Birrell GJ, Grubb BD (1991) Effects of paracetamol and aspirin on neural activity of joint mechanonociceptors in adjuvant arthritis. Br J Pharmacol 104(1):178–182
Murthy SE, Loud MC, Daou I, Marshall KL, Schwaller F, Kuhnemund J, Francisco AG, Keenan WT, Dubin AE, Lewin GR, Patapoutian A (2018) The mechanosensitive ion channel Piezo2 mediates sensitivity to mechanical pain in mice. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aat9897
Neogi T, Guermazi A, Roemer F, Nevitt MC, Scholz J, Arendt-Nielsen L, Woolf C, Niu J, Bradley LA, Quinn E, Law LF (2016) Association of joint inflammation with pain sensitization in knee osteoarthritis: the multicenter osteoarthritis study. Arthritis Rheumatol 68(3):654–661. https://doi.org/10.1002/art.39488
O'Hagan R, Chalfie M, Goodman MB (2005) The MEC-4 DEG/ENaC channel of Caenorhabditis elegans touch receptor neurons transduces mechanical signals. Nat Neurosci 8(1):43–50. https://doi.org/10.1038/nn1362
Okun A, Liu P, Davis P, Ren J, Remeniuk B, Brion T, Ossipov MH, Xie J, Dussor GO, King T, Porreca F (2012) Afferent drive elicits ongoing pain in a model of advanced osteoarthritis. Pain 153(4):924–933. https://doi.org/10.1016/j.pain.2012.01.022
Schaible HG (2014) Nociceptive neurons detect cytokines in arthritis. Arthritis Res Therapy 16(5):470
Sharif-Naeini R (2015) Contribution of mechanosensitive ion channels to somatosensation. Prog Mol Biol Transl Sci 131:53–71. https://doi.org/10.1016/bs.pmbts.2014.11.011
Shieh CC, Jarvis MF, Lee CH, Perner RJ (2006) P2X receptor ligands and pain. Expert Opin Ther Pat 16(8):1113–1127. https://doi.org/10.1517/13543776.16.8.1113
Szczot M, Liljencrantz J, Ghitani N, Barik A, Lam R, Thompson JH, Bharucha-Goebel D, Saade D, Necaise A, Donkervoort S, Foley AR, Gordon T, Case L, Bushnell MC, Bonnemann CG, Chesler AT (2018) PIEZO2 mediates injury-induced tactile pain in mice and humans. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aat9892
Vollrath MA, Kwan KY, Corey DP (2007) The micromachinery of mechanotransduction in hair cells. Annu Rev Neurosci 30:339–365. https://doi.org/10.1146/annurev.neuro.29.051605.112917
Woo SH, Lukacs V, de Nooij JC, Zaytseva D, Criddle CR, Francisco A, Jessell TM, Wilkinson KA, Patapoutian A (2015) Piezo2 is the principal mechanotransduction channel for proprioception. Nat Neurosci 18(12):1756–1762. https://doi.org/10.1038/nn.4162
Zhang M, Wang Y, Geng J, Zhou S, Xiao B (2019) Mechanically activated piezo channels mediate touch and suppress acute mechanical pain response in mice. Cell Rep 26(6):1419–1431. https://doi.org/10.1016/j.celrep.2019.01.056(e1414)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sharif-Naeini, R. Role of mechanosensitive ion channels in the sensation of pain. J Neural Transm 127, 407–414 (2020). https://doi.org/10.1007/s00702-020-02182-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-020-02182-2