Abstract
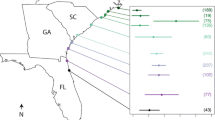
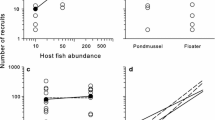
Drivers of large-scale variability in parasite prevalence are not well understood. For logistical reasons, explorations of spatial patterns in parasites are often performed as observational studies. However, to understand the mechanisms that underlie these spatial patterns, standardized and controlled comparisons are needed. Here, we examined spatial variability in infection of an important fishery species and ecosystem engineer, the oyster (Crassostrea virginica) by its pea crab parasite (Zaops ostreus) across 700 km of the southeastern USA coastline. To minimize the influence of host genetics on infection patterns, we obtained juvenile oysters from a homogeneous source stock and raised them in situ for 3 months at multiple sites with similar environmental characteristics. We found that prevalence of pea crab infection varied between 24 and 73 % across sites, but not systematically across latitude. Of all measured environmental variables, oyster recruitment correlated most strongly (and positively) with pea crab infection, explaining 92 % of the variability in infection across sites. Our data ostensibly suggest that regional processes driving variation in oyster recruitment similarly affect the recruitment of one of its common parasites.



Similar content being viewed by others
References
Beach NW (1969) The oyster crab, Pinnotheres ostreum Say, in the vicinity of Beaufort, North Carolina. Crustaceana 17:87–199
Blakeslee AMH, Altman I, Miller AW, Byers JE, Hamer CE, Ruiz GM (2012) Parasites and invasions: a biogeographic examination of parasites and hosts in native and introduced ranges. J Biogeogr 39:609–622
Brown HE, Childs JE, Diuk-Wasser MA, Fish D (2008) Ecological factors associated with West Nile virus transmission, northeastern United States. Emerg Infect Dis 14:1539–1545
Butman CA (1987) Larval settlement of soft-sediment invertebrates: the spatial scales of pattern explained by active habitat selection and the emerging role of hydrodynamical processes. Oceanogr Mar Biol 25:113–165
Byers JE, Blakeslee AMH, Linder E, Cooper AB, Maguire TJ (2008) Controls of spatial variation in the prevalence of trematode parasites infecting a marine snail. Ecology 89:439–451
Byers JE, Altman I, Grosse AM, Huspeni TC, Maerz JC (2011) Using parasitic trematode larvae to quantify an elusive vertebrate host. Conserv Biol 25:85–93
Christensen AM, McDermott JJ (1958) Life history and biology of the oyster crab, Pinnotheres ostreum Say. Biol Bull 114:146–179
Crossan J, Paterson S, Fenton A (2007) Host availability and the evolution of parasite life-history strategies. Evolution 61:675–684
de Montaudouin X, Lanceleur L (2011) Distribution of parasites in their second intermediate host, the cockle Cerastoderma edule: community heterogeneity and spatial scale. Mar Ecol Prog Ser 428:187–199
Dekshenieks MM, Hofmann EE, Powell EN (1993) Environmental effects on the growth and development of eastern oyster, Crassostrea virginica (Gmelin, 1791), larvae: a modeling study. J Shellfish Res 12:241–254
Fredensborg BL, Mouritsen KN, Poulin R (2006) Relating bird host distribution and spatial heterogeneity in trematode infections in an intertidal snail-from small to large scale. Mar Biol 149:275–283
Galtsoff PS (1964) The American oyster, Crassostrea virginica, Gmelin. United State Fish ad Wildlife Service, Fish Bull 64:1–480
Garamszegi LZ (2011) Climate change increases the risk of malaria in birds. Glob Change Biol 17:1751–1759
Harkonen L et al (2010) Predicting range expansion of an ectoparasite: the effect of spring and summer temperatures on deer ked Lipoptena cervi (Diptera: Hippoboscidae) performance along a latitudinal gradient. Ecography 33:906–912
Hatcher MJ, Taneyhill DE, Dunn AM, Tofts C (1999) Population dynamics under parasitic sex ratio distortion. Theor Popul Biol 56:11–28
Hawley DM, Altizer SM (2011) Disease ecology meets ecological immunology: understanding the links between organismal immunity and infection dynamics in natural populations. Funct Ecol 25:48–60
Hechinger RF, Lafferty KD (2005) Host diversity begets parasite diversity: bird final hosts and trematodes in snail intermediate hosts. Proc R Soc Lond B 272:1059–1066
Helmuth B et al (2006) Mosaic patterns of thermal stress in the rocky intertidal zone: implications for climate change. Ecol Monogr 76:461–479
Hudson PJ, Dobson AP, Newborn D (1998) Prevention of population cycles by parasite removal. Science 282:2256–2258
Linton TL (1968) Feasibility of raft culturing oysters in Georgia. In: Linton TL (ed) Proceedings of the Oyster Culture Workshop, July 11–13, 1967. University of Georgia and Georgia Game and Fisheries Commission, Marine Fisheries Division, Contribution Series Number 6, pp 69–73
Lopez JE, Gallinot LP, Wade MJ (2005) Spread of parasites in metapopulations: an experimental study of the effects of host migration rate and local host population size. Parasitology 130:323–332
Macnab V, Barber I (2012) Some (worms) like it hot: fish parasites grow faster in warmer water, and alter host thermal preferences. Glob Change Biol 18:1540–1548
Mercado-Silva N (2005) Condition index of the eastern oyster, Crassostrea virginica (Gmelin, 1791) in Sapelo Island Georgia: Effects of site, position on bed and pea crab parasitism. J Shellfish Res 24:121–126
Mouritsen KN, Jensen KT (1997) Parasite transmission between soft-bottom invertebrates: temperature mediated infection rates and mortality in Corophium volutator. Mar Ecol Prog Ser 151:123–134
Mouritsen KN, McKechnie S, Meenken E, Toynbee JL, Poulin R (2003) Spatial heterogeneity in parasite loads in the New Zealand cockle: the importance of host condition and density. J Marine Biol Assoc UK 83:307–310
O’Beirn FX, Walker RL (1999) Pea crab, Pinnotheres ostreum Say, 1817, in the eastern oyster, Crassostrea virginica (Gmelin, 1791): prevalence and apparent adverse effects on oyster gonad development. Veliger 42:17–20
Parks P (1968) A comparison of chemical composition of oysters (Crassostrea virginica) from different ecological habitats. In: Linton TL (ed) Proceedings of the Oyster Culture Workshop, July 11–13, 1967. University of Georgia and Georgia Game and Fisheries Commission, Marine Fisheries Division, Contribution Series Number 6, pp 147–173
Paull SH, Johnson PTJ (2011) High temperature enhances host pathology in a snail-trematode system: possible consequences of climate change for the emergence of disease. Freshw Biol 56:767–778
Perrin N, Christe P, Richner H (1996) On host life-history response to parasitism. Oikos 75:317–320
Poulin R, Mouritsen KN (2003) Large-scale determinants of trematode infections in intertidal gastropods. Mar Ecol Prog Ser 254:187–198
Raffel TR, Hoverman JT, Halstead NT, Michel PJ, Rohr JR (2010) Parasitism in a community context: trait-mediated interactions with competition and predation. Ecology 91:1900–1907
Sandifer PA (1972) Growth of young oyster crabs, Pinnotheres ostreum Say, reared in the laboratory. Chesapeake Sci 13:221–222
Sandoz M, Hopkins SH (1947) Early life history of the oyster crab, Pinnotheres ostreum (Say). Biol Bull 93:250–258
Smith NF (2001) Spatial heterogeneity in recruitment of larval trematodes to snail intermediate hosts. Oecologia 127:115–122
Smith NF (2007) Associations between shorebird abundance and parasites in the sand crab, Emerita analoga, along the California coast. J Parasitol 93:265–273
Stauber LA (1945) Pinnotheres ostreum, parasitic on the American oyster, Ostrea (Gryphaea) virginica. Biol Bull 88:269–291
Sutherst RW (2001) The vulnerability of animal and human health to parasites under global change. Int J Parasitol 31:933–948
Thieltges DW, Fredensborg BL, Poulin R (2009) Geographical variation in metacercarial infection levels in marine invertebrate hosts: parasite species character versus local factors. Mar Biol 156:983–990
Tinsley RC, York JE, Everard ALE, Stott LC, Chapple SJ, Tinsley MC (2011) Environmental constraints influencing survival of an African parasite in a north temperate habitat: effects of temperature on egg development. Parasitology 138:1029–1038
Torchin ME, Byers JE, Huspeni TC (2005) Differential parasitism of native and introduced snails: replacement of a parasite fauna. Biol Invasions 7:885–894
Tschirren B, Richner H (2006) Parasites shape the optimal investment in immunity. Proc R Soc Lond B 273:1773–1777
Wiedermann MM, Nordin A, Gunnarsson U, Nilsson MB, Ericson L (2007) Global change shifts vegetation and plant-parasite interactions in a boreal mire. Ecology 88:454–464
Wood CL, Byers JE, Cottingham KL, Altman I, Donahue MJ, Blakeslee AMH (2007) Parasites alter community structure. Proc Natl Acad Sci USA 104:9335–9339
Zamora-Vilchis I, Williams SE, Johnson CN (2012) Environmental temperature affects prevalence of blood parasites of birds on an elevation gradient: Implications for disease in a warming climate. Plos ONE 7:ARTN: e39208. doi:10.31371/journal.pone.0039208
Acknowledgments
We thank Luke Dodd, H. Garland, P. Langdon, Jenna Malek, E. Pettis, Walt Rogers, and Heidi Weiskel for help in the field and Tom McCrudden for spawning and growing our hatchery oyster spat. This work was financially supported by the National Science Foundation (NSF-OCE-0961853).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jeff Shima.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Byers, J.E., Rogers, T.L., Grabowski, J.H. et al. Host and parasite recruitment correlated at a regional scale. Oecologia 174, 731–738 (2014). https://doi.org/10.1007/s00442-013-2809-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-013-2809-2