Abstract
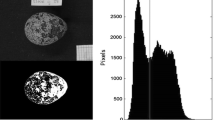
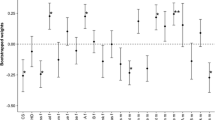
Many small passerine birds worldwide lay white eggs speckled with red, brown and black protoporphyrin pigment spots (maculation). Unlike some patterns of avian eggshell pigmentation which clearly serve a crypsis or signalling function, the ubiquity of maculation among passerines suggests that its origins lie in another function, not specific to any particular ecological or behavioural group. Elsewhere, we have presented evidence that protoporphyrin pigments serve a structural function related to eggshell thickness and calcium availability: eggshell maculation in the great tit Parus major increases with decreasing soil calcium levels, pigments demarcate thinner areas of shell, and both the pigment intensity and distribution are related to shell thickness. Here we show that maculation also affects the rate of water loss from the egg during incubation (≈ Mass Loss per Day or MLD, which is critical to egg viability), but not that of unincubated eggs. We also demonstrate, both by observation and experiment, that the effect of female incubation behaviour on MLD compensates in some way for variation in egg characteristics, and that differences between females in the degree of such compensation are related to differences in clutch maculation. Our results suggest that, while a principal function of maculation in this species may be to strengthen the eggshell, it may also reduce eggshell permeability when large amounts of pigment are used, and that this necessitates a behavioural adjustment from the female during incubation. We discuss these findings and make further testable predictions from our model.





Similar content being viewed by others
References
Ar A, Rahn H (1978) Interdependence of gas conductance, incubation length and weight of the avian egg. In: Piiper J (ed) Respiratory function in birds, adult and embryonic. Springer, Berlin Heidelberg New York, pp 227–236
Ar A, Paganelli CV, Reeves RB, Greene DG, Rahn H (1974) The avian egg: water vapour conductance, shell thickness, and functional pore area. Condor 76:153–158
Ar A, Rahn H, Paganelli CV (1979) The avian egg: mass and strength. Condor 81:321–327
Bakken GS, Vanderbilt VC, Buttemer WA, Dawson WR (1978) Avian eggs: thermoregulatory value of very high near-infrared reflectance. Science 200:321–323
Blanco G, Bertellotti M (2002) Differential predation by mammals and birds: implications for egg-colour polymorphism in a nomadic breeding seabird. Biol J Linn Soc 75:137–146
Davies NB, Brooke MD (1989a) An experimental-study of co-evolution between the Cuckoo, Cuculus-Canorus, and its hosts. 1. Host egg discrimination. J Anim Ecol 58:207–224
Davies NB, Brooke MD (1989b) An experimental-study of co-evolution between the Cuckoo, Cuculus-Canorus, and its hosts. 2. Host egg markings, chick discrimination and general discussion. J Anim Ecol 58:225–236
Dawkins HC, Field DRB (1978) C.F.I. Occasional Paper No. 1. A Long-Term Surveillance System for British Woodland Vegetation. Commonwealth Forestry Institute, University of Oxford, Oxford
Farmer AM (1995) Soil chemistry change in a lowland English deciduous woodland. Water Air Soil pollut 85:677–682
Gosler AG (1993). The Great Tit. Hamlyn species guides. Paul Hamlyn, London
Gosler AG, Barnett PR, Reynolds SJ (2000) Inheritance and variation in eggshell patterning in the great tit Parus major. Proc R Soc Lond B Biol Sci 267:2469–2473
Gosler AG, Higham JP, Reynolds SJ (2005) Why are birds’ eggs speckled? Ecol Lett 8:1105–1113
Graveland J, Berends AE (1997) Timing of the calcium intake and effect of calcium deficiency on behaviour and egg laying in captive great tits, Parus major. Physiol Zool 70:74–84
Graveland J, Drent RH (1997) Calcium availability limits breeding success of passerines on poor soils. J Anim Ecol 66:279–288
Graveland J, Vanderwal R, Van Balen JH, Van Noordwijk AJ (1994) Poor reproduction in forest passerines from decline of snail abundance on acidified soils. Nature 368:446–448
Hoyt DF (1979) Practical methods of estimating volume and fresh weight of birds eggs. Auk 96:73–77
Kennedy GY, Vevers HG (1976) A survey of avian eggshell pigments. Compu Biochem Physiol 55B:117–123
Lack D (1968) Ecological adaptations for breeding in birds. Methuen, London
Mand R, Tilgar V, Leivits A (2000) Reproductive response of Great Tits, Parus major, in a naturally base-poor forest habitat to calcium supplementation. Can J Zool-Rev Can Zool 78:689–695
Mikhailov KE (1997) Avian eggshells: an atlas of scanning electron micrographs., BOC occasional publications: 3 British Ornithologists’ Club
Moreno J, Osorno JL (2003) Avian egg colour and sexual selection: does eggshell pigmentation reflect female condition and genetic quality? Ecol Lett 6:803–806
Moreno J, Osorno JL, Morales J, Merino S, Tomás G (2004) Egg colouration and male parental effort in the pied flycatcher Ficedula hypoleuca. J Avian Biol 35:300–304
Newton AV (1896) A dictionary of birds. A. C. Black, London
Perrins CM (1979) British Tits. Collins, London
Perrins CM (1996) Eggs, egg formation and the timing of breeding. Ibis 138:2–15
Rahn H, Ar A (1974) The avian egg: incubation time and water loss. Condor 76:147–152
Sanchez JM, Corbacho C, del Viejo AM, Parejo D (2004) Colony-site tenacity and egg color crypsis in the Gull-billed Tern. Waterbirds 27:21–30
Seebohm H 1896 Eggs of British birds. Pawson & Brailsford, Sheffield
Sibley CG, Monroe BL Jr (1990) Distribution and taxonomy of the birds of the world. Yale University Press, New Haven
Soler JJ, Moreno J, Aviles JM, Møller AP (2005) Blue and green egg-color intensity is associated with parental effort and mating system in passerines: support for the sexual selection hypothesis. Evolution 59:636–644
Solomon SE (1987) Egg shell pigmentation. In: Wells RG, Belyarin CG (eds) Egg quality—current problems and recent advances. Butterworths, London, pp 147–157
Solomon SE (1997) Egg and eggshell quality. Iowa State University Press, Ames
Tazawa H, Whittow GC (2000) Incubation physiology. In: Sturkie PD, Whittow GC (eds) Avian physiology. Academic Press, London
Tilgar V, Mand R, Leivits A (1999) Effect of calcium availability and habitat quality on reproduction in Pied Flycatcher Ficedula hypoleuca and Great Tit Paras major. J Avian Biol 30:383–391
Tyler C (1969) Avian egg shells: their structure and chararcteristics. In: Felts WJL, Harrison RJ (eds) International review of general and experimental zoology. Academic Press, New York
Van Noordwijk AJ, Keizer LCP, Van Balen JH, Scharloo W (1981) Genetic variation in egg dimensions in natural populations of the Great Tit. Genetica 55:221–232
Weimar V, Schmidt K-H (1998) Untersuchungen zur Eiqualitat bei der Kohlmeise (Parus major) in Abhangigkeit von der Bodenbeschaffenheit. Journal fur Ornithologie 139:3–9
Acknowledgments
This work was carried out under English Nature licences to AGG—2002: 20020828 and 20012058, and 2003: 20030589, 20030184 and 20031149. Robin McCleery made the CFI calcium data available to us. We are grateful to Ben Sheldon, Dany Garant, Tom Pizzari and three anonymous referees for their constructive criticism of earlier drafts, and Stuart Semple, Markku Orell and one anonymous referee for helpful comments on the present one. JPH was supported by a BBSRC studentship, AGG by the Edward Grey Institute, University of Oxford.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Markku Orell.
Rights and permissions
About this article
Cite this article
Higham, J.P., Gosler, A.G. Speckled eggs: water-loss and incubation behaviour in the great tit Parus major . Oecologia 149, 561–570 (2006). https://doi.org/10.1007/s00442-006-0484-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-006-0484-2