Abstract
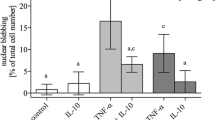
Pro-inflammatory cytokines induce meniscal matrix degradation and inhibition of endogenous repair mechanisms, but the pathogenic mechanisms behind this are mostly unknown. Therefore, we investigated details of interleukin-1 (IL-1α)-induced aggrecan turnover in mature meniscal tissue explants. Fibro-cartilagenous disks (3 mm diameter × 1 mm thickness) were isolated from the central, weight-bearing region of menisci from 2-year-old cattle. After 3 or 6 days of IL-1α-treatment, GAG loss (DMMB assay), biosynthetic activity ([35SO4]-sulfate and [3H]-proline incorporation), gene expression (quantitative RT-PCR) and the abundance (zymography, Western blot) of matrix-degrading enzymes and specific aggrecan products were determined. Meniscal fibrocartilage had a 4-fold lower GAG content (per wet weight) than adjacent articular cartilage, and expressed MMPs-1, -2, -3 and ADAMTS4 constitutively, whereas ADAMTS5 m-RNA was essentially undetectable. Significant IL-1 effects were a decrease in biosynthetic activity, an increase in GAG release and in the expression/abundance of MMP-2, MMP-3 and ADAMTS4. Fresh tissue contained aggrecan core protein products similar to those previously described for bovine articular cartilage of this age. IL-1 induced the release of aggrecanase-generated CS-substituted products including both high (>250 kDa) and low molecular weight (about 75 kDa) species. TIMP-3 (but not TIMP-1 and -2 or a broad spectrum MMP inhibitor) inhibited IL-1-dependent GAG loss. In addition, IL-1 induced the release of preformed pools of three known G1-bearing products. We conclude that aggrecanases are responsible for IL-1-stimulated GAG release from meniscal explants, and that IL-1 also stimulates release of G1-bearing products, by a process possibly involving hyaluronan fragmentation.




Similar content being viewed by others
References
Bluteau G, Conrozier T, Mathieu P, Vignon E, Herbage D, Mallein-Gerin F (2001) Matrix metalloproteinase-1 ,-3, -13 and aggrecanase-1 and -2 are differentially expressed in experimental osteoarthritis. Biochim Biophys Acta 1526:147–158
Carlson CS, Guilak F, Vail TP, Gardin JF, Kraus VB (2002) Synovial fluid biomarker levels predict articular cartilage damage following complete medial meniscectomy in the canine knee. J Orthop Res 20:92–100
Embry Flory JJ, Fosang AJ, Knudson W (2006) The accumulation of intracellular ITEGE and DIPEN neoepitopes in bovine articular chondrocytes is mediated by CD44 internalization of hyaluronan. Arthritis Rheum 54:443–454
Gao G, Westling J, Thompson VP, Howell TD, Gottschall PE, Sandy JD (2002) Activation of the proteolytic activity of ADAMTS4 (aggrecanase-1) by C-terminal truncation. J Biol Chem 277:11034–11041
Gendron C, Kashiwagi M, Hughes C, Caterson B, Nagase H (2003) TIMP-3 inhibits aggrecanase-mediated glycosaminoglycan release from cartilage explants stimulated by catabolic factors. FEBS Lett 555:431–436
Hennerbichler A, Moutos FT, Hennerbichler D, Weinberg JB, Guilak F (2007) Interleukin-1 and tumor necrosis alpha inhibit repair of the porcine meniscus in vitro. Osteoarthr Cartil 15:1053–1060
Kashiwagi M, Tortorella M, Nagase H, Brew K (2001) TIMP-3 is a potent inhibitor of aggrecanase 1 (ADAM-TS4) and aggrecanase 2 (ADAM-TS5). J Biol Chem 276:12501–12504
Knudson W, Loeser RF (2002) CD44 and integrin matrix receptors participate in cartilage homeostasis. Cell Mol Life Sci 59:36–44
Kurz B, Jin M, Patwari P, Cheng DM, Lark MW, Grodzinsky AJ (2001) Biosynthetic response and mechanical properties of articular cartilage after injurious compression. J Orthop Res 19:1140–1146
Loening AM, James IE, Levenston ME, Badger AM, Frank EH, Kurz B, Nuttall ME, Hung HH, Blake SM, Grodzinsky AJ, Lark MW (2000) Injurious mechanical compression of bovine articular cartilage induces chondrocyte apoptosis. Arch Biochem Biophys 381:205–212
Lohmander LS, Neame PJ, Sandy JD (1993) The structure of aggrecan fragments in human synovial fluid. Evidence that aggrecanase mediates cartilage degradation in inflammatory joint disease, joint injury, and osteoarthritis. Arthritis Rheum 36:1214–1222
Maehara H, Suzuki K, Sasaki T, Oshita H, Wada E, Inoue T, Shimizu K (2007) G1-G2 aggrecan product that can be generated by M-calpain on truncation at Ala709-Ala710 is present abundantly in human articular cartilage. J Biochem 141:469–477
Manicourt DH, Lefebvre V (1993) An assay for matrix metalloproteinases and other proteases acting on proteoglycans, casein, or gelatin. Anal Biochem 215:171–179
MacPherson LJ, Bayburt EK, Capparelli MP, Carroll BJ, Goldstein R, Justice MR, Zhu L, Hu S, Melton RA, Fryer L, Goldberg RL, Doughty JR, Spirito S, Blancuzzi V, Wilson D, O'Byrne EM, Ganu V, Parker DT (1997) Discovery of CGS 27023A, a non-peptidic, potent, and orally active stromelysin inhibitor that blocks cartilage degradation in rabbits. J Med Chem 40:2525–2532
Oshita H, Sandy JD, Suzuki K, Akaike A, Bai Y, Sasaki T, Shimizu K (2004) Mature bovine articular cartilage contains abundant aggrecan that is C-terminally truncated at Ala719-Ala720, a site which is readily cleaved by m-calpain. Biochem J 382:253–259
Patwari P, Gao G, Lee JH, Grodzinsky AJ, Sandy JD (2005) Analysis of ADAMTS4 and MT4-MMP indicates that both are involved in aggrecanolysis in interleukin-1-treated bovine cartilage. Osteoarthr Cartil 13:269–277
Rupp S, Seil R, Kohn D (2002) Meniscus lesions. Orthopade 31:812–828
Sandy JD (2006) A contentious issue finds some clarity: on the independent and complementary roles of aggrecanase activity and MMP activity in human joint aggrecanolysis. Osteoarthr Cartil 14:95–100
Sandy JD, Verscharen C (2001) Analysis of aggrecan in human knee cartilage and synovial fluid indicates that aggrecanase (ADAMTS) activity is responsible for the catabolic turnover and loss of whole aggrecan whereas other protease activity is required for C-terminal processing in vivo. Biochem J 358:615–626
Sandy JD, Flannery CR, Neame PJ, Lohmander LS (1992) The structure of aggrecan fragments in human synovial fluid. Evidence for the involvement in osteoarthritis of a novel proteinase which cleaves the Glu 373-Ala374 bond of the interglobular domain. J Clin Invest 89:1512–1516
Sandy JD, Plaas AH, Koob TJ (1995) Pathways of aggrecan processing in joint tissues. Implications for disease mechanism and monitoring. Acta Orthop Scand Suppl 266:26–32
Sandy JD, Thompson V, Doege K, Verscharen C (2000) The intermediates of aggrecanase-dependent cleavage of aggrecan in rat chondrosarcoma cells treated with interleukin-1. Biochem J 351:161–166
Schlaak JF, Pfers I, Meyer Zum Buschenfelde KH, Marker-Hermann E (1996) Different cytokine profiles in the synovial fluid of patients with osteoarthritis, rheumatoid arthritis and seronegative spondylarthropathies. Clin Exp Rheumatol 14:155–162
Shin SJ, Fermor B, Weinberg JB, Pisetsky DS, Guilak F (2003) Regulation of matrix turnover in meniscal explants: role of mechanical stress, interleukin-1, and nitric oxide. J Appl Physiol 95:308–313
Sugimoto K, Iizawa T, Harada H, Yamada K, Katsumata M, Takahashi M (2004) Cartilage degradation independent of MMP/aggrecanases. Osteoarthr Cartil 12:1006–1014
Wilson CG, Vanderploeg EJ, Zuo F, Sandy JD, Levenston ME (2009) Aggrecanolysis and in vitro matrix degradation in the immature bovine meniscus: mechanisms and functional implications. Arthritis Res Therapy epub doi:10.1186/ar2862
Wilusz RE, Weinberg JB, Guilak F, McNulty AL (2008) Inhibition of integrative repair of the meniscus following acute exposure to interleukin-1 in vitro. J Orthop Res 26:504–512
Acknowledgements
We thank Rita Kirsch, Elsbeth Schulz, Frank Lichte and Marianne Ahler for their technical support. We also thank the Hofschlachterei Muhs (Krummbek) and NFZ Norddeutsche Fleischzentrale GmbH for the utilization of the knee joints.
Note
The amino acid residue numbers used in this manuscript are the bovine aggrecan sequence starting with N-terminal 1MTTL (NCBI accession no. P13608).
Author information
Authors and Affiliations
Corresponding author
Additional information
Angelika K. Lemke and John D. Sandy contributed equally to this paper.
The study was funded by the Endo-Stiftung, Stiftung des Gemeinnützigen Vereins der ENDO-Klinik e.V., Hamburg.
Rights and permissions
About this article
Cite this article
Lemke, A.K., Sandy, J.D., Voigt, H. et al. Interleukin-1α treatment of meniscal explants stimulates the production and release of aggrecanase-generated, GAG-substituted aggrecan products and also the release of pre-formed, aggrecanase-generated G1 and m-calpain-generated G1-G2. Cell Tissue Res 340, 179–188 (2010). https://doi.org/10.1007/s00441-010-0941-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-010-0941-4