Abstract
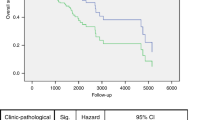
For TP53-mutated head and neck squamous cell carcinomas (HNSCCs), the codon and specific amino acid sequence change resulting from a patient’s mutation can be prognostic. Thus, developing a framework to predict patient survival for specific mutations in TP53 would be valuable. There are many bioinformatics and functional methods for predicting the phenotypic impact of genetic variation, but their overall clinical value remains unclear. Here, we assess the ability of 15 different methods to predict HNSCC patient survival from TP53 mutation, using TP53 mutation and clinical data from patients enrolled in E4393 by the Eastern Cooperative Oncology Group (ECOG), which investigated whether TP53 mutations in surgical margins were predictive of disease recurrence. These methods include: server-based computational tools SIFT, PolyPhen-2, and Align-GVGD; our in-house POSE and VEST algorithms; the rules devised in Poeta et al. with and without considerations for splice-site mutations; location of mutation in the DNA-bound TP53 protein structure; and a functional assay measuring WAF1 transactivation in TP53-mutated yeast. We assessed method performance using overall survival (OS) and progression-free survival (PFS) from 420 HNSCC patients, of whom 224 had TP53 mutations. Each mutation was categorized as “disruptive” or “non-disruptive”. For each method, we compared the outcome between the disruptive group vs. the non-disruptive group. The rules devised by Poeta et al. with or without our splice-site modification were observed to be superior to others. While the differences in OS (disruptive vs. non-disruptive) appear to be marginally significant (Poeta rules + splice rules, P = 0.089; Poeta rules, P = 0.053), both algorithms identified the disruptive group as having significantly worse PFS outcome (Poeta rules + splice rules, P = 0.011; Poeta rules, P = 0.027). In general, prognostic performance was low among assessed methods. Further studies are required to develop and validate methods that can predict functional and clinical significance of TP53 mutations in HNSCC patients.


Similar content being viewed by others

References
Adzhubei IA et al (2010) A method and server for predicting damaging missense mutations. Nat Meth 7: 248–249, http://www.nature.com/nmeth/journal/v7/n4/suppinfo/nmeth0410-248_S1.html
Ahmed AA et al (2010) Driver mutations in TP53 are ubiquitous in high grade serous carcinoma of the ovary. J Pathol 221:49–56. doi:10.1002/path.2696
Bartel F et al (2008) Both germ line and somatic genetics of the p53 pathway affect ovarian cancer incidence and survival. Clin Cancer Res 14:89–96. doi:10.1158/1078-0432.ccr-07-1192
Bisio A, Ciribilli Y, Fronza G, Inga A, Monti P (2014) TP53 mutants in the tower of babel of cancer progression. Hum Mutant n/a-n/a. doi:10.1002/humu.22514
Bonnet C et al (2008) Screening BRCA1 and BRCA2 unclassified variants for splicing mutations using reverse transcription PCR on patient RNA and an ex vivo assay based on a splicing reporter minigene. J Med Genet 45:438–446. doi:10.1136/jmg.2007.056895
Bradley PJ, MacLennan K, Brakenhoff RH, Leemans CR (2007) Status of primary tumour surgical margins in squamous head and neck cancer: prognostic implications. Current Opin Otolaryngol Head Neck Surg 15:74–81
Carter H et al (2009) Cancer-specific high-throughput annotation of somatic mutations: computational prediction of driver missense mutations. Cancer Res 69:6660–6667. doi:10.1158/0008-5472.can-09-1133
Carter H, Douville C, Stenson P, Cooper D, Karchin R (2013) Identifying mendelian disease genes with the variant effect scoring tool. BMC Genom 14:S3
Chan PA et al (2007) Interpreting missense variants: comparing computational methods in human disease genes CDKN2A, MLH1, MSH2, MECP2, and tyrosinase (TYR). Hum Mutat 28:683–693. doi:10.1002/humu.20492
Cox IDR (1970) Analysis of binary data. Methuen and Company Ltd, London, pp 61–62
Cox DR (1972) Regression models and life tables. JR stat soc B 34:187–220
Darcy KM et al (2008) Associations between p53 overexpression and multiple measures of clinical outcome in high-risk, early stage or suboptimally-resected, advanced stage epithelial ovarian cancers: a Gynecologic Oncology Group study. Gynecologic Oncology 111:487–495. doi:10.1016/j.ygyno.2008.08.020
de Graeff P et al. (2009) Modest effect of p53, EGFR and HER-2/neu on prognosis in epithelial ovarian cancer: a meta-analysis. Br J Cancer 101, pp. 149–159, http://www.nature.com/bjc/journal/v101/n1/suppinfo/6605112s1.html
Desmet FOO et al (2009) Human splicing finder: an online bioinformatics tool to predict splicing signals. Nucleic Acids Res 37:e67. doi:10.1093/nar/gkp215
Dohner H et al (1995) p53 gene deletion predicts for poor survival and non-response to therapy with purine analogs in chronic B-cell leukemias. Blood 85:1580–1589
Douville C et al (2013) CRAVAT: cancer-related analysis of variants toolkit. Bioinformatics 29:647–648. doi:10.1093/bioinformatics/btt017
El-Deiry WS et al (1993) WAF1, a potential mediator of p53 tumor suppression. Cell 75:817–825. doi:10.1016/0092-8674(93)90500-P
Ferris RL et al (2014) Phase I dendritic cell p53 peptide vaccine for head and neck cancer. Clin Cancer Res. doi:10.1158/1078-0432.ccr-13-2617
Gentile M, Bergman Jungeström M, Olsen KE, Söderkvist P, Wingren S (1999) p53 and survival in early onset breast cancer: analysis of gene mutations, loss of heterozygosity and protein accumulation. Eur J Cancer 35:1202–1207. doi:10.1016/S0959-8049(99)00121-5
Gonzalez D et al (2011) Mutational status of the TP53 gene as a predictor of response and survival in patients with chronic lymphocytic leukemia: results from the LRF CLL4 trial. J Clin Oncol 29:2223–2229. doi:10.1200/jco.2010.32.0838
Greaves M, Maley CC (2012) Clonal evolution in cancer. Nature 481: 306–313, http://www.nature.com/nature/journal/v481/n7381/abs/nature10762.html—supplementary-information
Hicks S, Wheeler DA, Plon SE, Kimmel M (2011) Prediction of missense mutation functionality depends on both the algorithm and sequence alignment employed. Hum Mutat 32:661–668. doi:10.1002/humu.21490
HØGdall EVS et al (2008) Distribution of p53 expression in tissue from 774 Danish ovarian tumour patients and its prognostic significance in ovarian carcinomas. APMIS 116:400–409. doi:10.1111/j.1600-0463.2008.00917.x
Integrated genomic analyses of ovarian carcinoma. (2011) Nature 474: pp. 609–615, http://www.nature.com/nature/journal/v474/n7353/abs/nature10166-f1.2.html—supplementary-information
Kaplan EL, Meier P (1958) Nonparametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Kato S et al (2003) Understanding the function, Äìstructure and function, Äìmutation relationships of p53 tumor suppressor protein by high-resolution missense mutation analysis. Proc Natl Acad Sci 100:8424–8429. doi:10.1073/pnas.1431692100
Keats JJ et al (2012) Clonal competition with alternating dominance in multiple myeloma. Blood 120:1067–1076. doi:10.1182/blood-2012-01-405985
Kumar P, Henikoff S, Ng PC (2009) Predicting the effects of coding non-synonymous variants on protein function using the SIFT algorithm. Nat Protoc 4:1073–1081
Lothe RA et al (1995) Deletion of 1p loci and microsatellite instability in colorectal polyps. Genes Chromosom Cancer 14:182–188
Ludwig JA, Weinstein JN (2005) Biomarkers in Cancer Staging, Prognosis and Treatment Selection. Nat Rev Cancer 5:845–856
Marcel V et al (2011) Biological functions of p53 isoforms through evolution: lessons from animal and cellular models. Cell Death Differ 18:1815–1824
Marusyk A, Almendro V, Polyak K (2012) Intra-tumour heterogeneity: a looking glass for cancer? Nat Rev Cancer 12:323–334
Masica DL, Sosnay PR, Cutting GR, Karchin R (2012) Phenotype-optimized sequence ensembles substantially improve prediction of disease-causing mutation in cystic fibrosis. Hum Mutat 33:1267–1274. doi:10.1002/humu.22110
Mehta CR, Patel NR, Tsiatis AA (1984) Exact significance testing to establish treatment equivalence with ordered categorical data. Biometrics, 819–825
Muller PAJ, Vousden KH (2013) p53 mutations in cancer. Nat Cell Biol 15:2–8
Newcomb EW et al (1998) Survival of patients with glioblastoma multiforme is not influenced by altered expression of P16, P53, EGFR, MDM2 or Bcl-2 genes. Brain Pathol 8:655–667. doi:10.1111/j.1750-3639.1998.tb00191.x
Olivier M (2013) In p53 in the Clinics. In: Hainaut P, Olivier M, Wiman KG (eds). Springer, New York, Ch. 8, pp. 127–146
Parsons DW et al (2011) The genetic landscape of the childhood cancer medulloblastoma. Science 331:435–439. doi:10.1126/science.1198056
Petitjean A et al (2007) Impact of mutant p53 functional properties on TP53 mutation patterns and tumor phenotype: lessons from recent developments in the IARC TP53 database. Hum Mutat 28:622–629. doi:10.1002/humu.20495
Peto R, Peto J (1972) Asymptotically efficient rank invariant test procedures. JR Stat Soc 135:185–206
Poeta ML et al (2007) TP53 Mutations and Survival in Squamous-Cell Carcinoma of the Head and Neck. N Engl J Med 357:2552–2561. doi:10.1056/NEJMoa073770
Powell B, Soong R, Iacopetta B, Seshadri R, Smith DR (2000) Prognostic significance of mutations to different structural and functional regions of the p53 gene in breast cancer. Clin Cancer Res 6:443–451
Robles AI, Harris CC (2010) Clinical outcomes and correlates of TP53 mutations and cancer. Cold Spring Harb Perspect Biol 2:a001016. doi:10.1101/cshperspect.a001016
Rossi D et al (2009) The Prognostic value of TP53 mutations in chronic lymphocytic leukemia is independent of Del17p13: implications for overall survival and chemorefractoriness. Clin Cancer Res 15:995–1004. doi:10.1158/1078-0432.ccr-08-1630
Shihab HA et al (2013) Predicting the functional, molecular, and phenotypic consequences of amino acid substitutions using hidden markov models. Hum Mutat 34:57–65. doi:10.1002/humu.22225
Simmons ML et al (2001) Analysis of complex relationships between age, p53, epidermal growth factor receptor, and survival in glioblastoma patients. Cancer Res 61:1122–1128
Soussi T, Kato S, Levy PP, Ishioka C (2005) Reassessment of the TP53 mutation database in human disease by data mining with a library of TP53 missense mutations. Hum Mutat 25:6–17. doi:10.1002/humu.20114
Steels E et al (2001) Role of p53 as a prognostic factor for survival in lung cancer: a systematic review of the literature with a meta-analysis. Eur Respir J 18:705–719
Tabor MP et al (2001) Persistence of genetically altered fields in head and neck cancer patients: biological and clinical implications. Clin Cancer Res 7:1523–1532
Tavtigian SV, Greenblatt MS, Lesueur F, Byrnes GB (2008) In silico analysis of missense substitutions using sequence-alignment based methods. Hum Mutat 29:1327–1336. doi:10.1002/humu.20892
Thusberg J, Olatubosun A, Vihinen M (2011) Performance of mutation pathogenicity prediction methods on missense variants. Hum Mutat 32:358–368. doi:10.1002/humu.21445
Uno H, Cai T, Pencina MJ, D’Agostino RB, Wei LJ (2011) On the C-statistics for evaluating overall adequacy of risk prediction procedures with censored survival data. Stat Med 30:1105–1117. doi:10.1002/sim.4154
Vogelstein B et al (2013) Cancer Genome Landscapes. Science 339:1546–1558. doi:10.1126/science.1235122
Weller M et al (2009) Molecular predictors of progression-free and overall survival in patients with newly diagnosed glioblastoma: a prospective translational study of the German Glioma network. J Clin Oncol 27:5743–5750. doi:10.1200/jco.2009.23.0805
Wong KK et al (2013) Poor survival with wild-type TP53 ovarian cancer? Gynecol Oncol 130:565–569. doi:10.1016/j.ygyno.2013.06.016
Yeo G, Burge CB (2004) Maximum entropy modeling of short sequence motifs with applications to RNA splicing signals. J Comput Biol 11:377–394
Zenz T et al (2010) TP53 Mutation and Survival in Chronic Lymphocytic Leukemia. J Clin Oncol 28:4473–4479. doi:10.1200/jco.2009.27.8762
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Masica, D.L., Li, S., Douville, C. et al. Predicting survival in head and neck squamous cell carcinoma from TP53 mutation. Hum Genet 134, 497–507 (2015). https://doi.org/10.1007/s00439-014-1470-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-014-1470-0