Abstract
Key message
Floret and seed traits are moderately correlated phenotypically in modern sunflower cultivars, but the underlying genetics are mostly independent. Seed traits in particular are governed in part by epistatic effects among quantitative trait loci.
Abstract
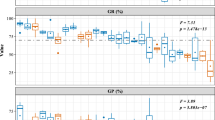
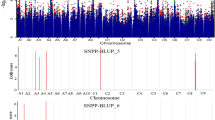
Seed size is an important quality component in marketing commercial sunflower (Helianthus annuus L.), particularly for the in-shell confectionery market, where long and broad seed types are preferred as a directly consumed snack food globally. Floret size is also important because corolla tube length was previously shown to be inversely correlated with pollinator visitation, impacting bee foraging potential and pollinator services to the plant. Commercial sunflower production benefits from pollinator visits, despite being self-compatible, and bees are required in hybrid seed production, where “female” and “male” inbred lines are crossed at field scale. Issues with pollination of long-seed confectionery sunflower suggest that there may be an unfavorable correlation between seed and floret traits; thus, our objective was to determine the strength of the correlation between seed and floret traits, and confirm any co-localization of seed and floret trait loci using genome-wide association analysis in the SAM diversity panel of sunflower. Our results indicate that phenotypic correlations between seed and floret traits are generally low to moderate, regardless of market class, a component of population substructure. Association mapping results mirror the correlations: while a few loci overlap, many loci for the two traits are not overlapping or even adjacent. The genetics of these traits, while modestly quantitative and influenced by epistatic effects, are not a barrier to simultaneous improvement of seed length and pollinator-friendly floret traits. We conclude that breeding for large seed size, which is required for the confectionery seed market, is possible without producing florets too long for efficient use by pollinators, which promotes bee foraging and associated pollination services.



Similar content being viewed by others
References
Bachlava E, Tang S, Pizarro G, Schuppert GF, Brunick RK, Draeger D, Leon A, Hahn V, Knapp SJ (2010) Pleiotropy of the branching locus (B) masks linked and unlinked quantitative trait loci affecting seed traits in sunflower. Theor Appl Genet 120:829–842. https://doi.org/10.1007/s00122-009-1212-1
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: Analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265. https://doi.org/10.1093/bioinformatics/bth457
Burke JM, Tang S, Knapp SJ, Rieseberg LH (2002) Genetic analysis of sunflower domestication. Genetics 161:1257–1267
Cakmak I, Song DS, Mixson TA, Serrano E, Clement ML, Savitski A, Johnson G, Giray T, Abramson CI, Barthell JF, Wells H (2010) Foraging response of turkish honey bee subspecies to flower color choices and reward consistency. J Insect Behav 23:100–116. https://doi.org/10.1007/s10905-009-9199-7
Cerrutti N, Pontet C (2016) Differential attractiveness of sunflower cultivars to the honeybee Apis mellifera L. Ocl 23:D204. https://doi.org/10.1051/ocl/2016005
Danecek P, Auton A, Abecasis G, Albers CA, Banks E, DePristo MA, Handsaker RE, Lunter G, Marth GT, Sherry ST, McVean G, Durbin R (2011) The variant call format and VCFtools. Bioinformatics 27:2156–2158. https://doi.org/10.1093/bioinformatics/btr330
DeBolt S, Scheible W-R, Schrick K, Auer M, Beisson F, Bischoff V, Bouvier-Navé P, Carroll A, Hematy K, Li Y, Milne J, Nair M, Schaller H, Zemla M, Somerville C (2009) Mutations in UDP-glucose: sterol glucosyltransferase in arabidopsis cause transparent testa phenotype and suberization defect in seeds. Plant Physiol 151:78–87. https://doi.org/10.1104/pp.109.140582
Degrandi-Hoffman G, Watkins JC (2000) The foraging activity of honey bees Apis mellifera and non-Apis bees on hybrid sunflowers (Helianthus annuus) and its influence on cross-pollination and seed set. J Apicult Res 39:37–45
Du Toit AP (1990) The importance of certain insects as pollinators of sunflower. S Afr J Plant Soil 7:159–162. https://doi.org/10.1080/02571862.1990.10634559
Gabriel SB, Schaffner SF, Nguyen H, Moore JM, Roy J, Blumenstiel B, Higgins J, DeFelice M, Lochner A, Faggart M, Liu-Cordero SN, Rotimi C, Adeyemo A, Cooper R, Ward R, Lander ES, Daly MJ, Altshuler D (2002) The structure of haplotype blocks in the human genome. Science 296:2225–2229. https://doi.org/10.1126/science.1069424
Gao X, Starmer J, Martin E (2008) A multiple testing correction method for genetic association studies using correlated single nucleotide polymorphisms. Genet Epidemiol 32:361–369
Garrison E, Marth G (2012) Haplotype-based variant detection from short-read sequencing. arXiv:1207.3907 [q-bio.GN]
Hao DC, Chen SL, Xiao PG, Liu M (2012) Application of high-throughput sequencing in medicinal plant transcriptome studies. Drug Dev Res 73:487–498
INRA (2018) INRA sunflower bioinformatics resources. https://www.heliagene.org. Accessed 7 Mar 2019
Kaczorowski RL, Juenger TE, Holtsford TP (2008) Heritability and correlation structure of nectar and floral morphology traits in Nicotiana alata. Evolution (NY) 62:1738–1750. https://doi.org/10.1111/j.1558-5646.2008.00400.x
Kamerling JP, Boons G-J, Lee YC, Suzuki A, Taniguchi N, and Voragen AGJ. (2007) Comprehensive glycoscience. From chemistry to systems biology. In: Kamerling JP (ed) Elsevier Science, Amsterdam, The Netherlands
Knauer AC, Schiestl FP (2015) Bees use honest floral signals as indicators of reward when visiting flowers. Ecol Lett 18:135–143. https://doi.org/10.1111/ele.12386
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25:1754–1760. https://doi.org/10.1093/bioinformatics/btp324
Li N, Li Y (2014) Ubiquitin-mediated control of seed size in plants. Front Plant Sci. https://doi.org/10.3389/fpls.2014.00332
Liu X, Huang M, Fan B, Buckler ES, Zhang Z (2016) Iterative usage of fixed and random effect models for powerful and efficient genome-wide association studies. PLoS Genet 12:24. https://doi.org/10.1371/journal.pgen.1005767
Mallinger R, Prasifka J (2017a) Benefits of insect pollination to confection sunflowers differ across plant genotypes. Crop Sci 57:3264. https://doi.org/10.2135/cropsci2017.03.0148
Mallinger RE, Prasifka JR (2017b) Bee visitation rates to cultivated sunflowers increase with the amount and accessibility of nectar sugars. J Appl Entomol 141:561–573. https://doi.org/10.1111/jen.12375
Mallinger RE, Bradshaw JD, Varenhorst A, Prasifka JR (2018) Native solitary bees provide economically significant pollination services to confection sunflowers across three states. J Econ Entomol 112:40–48
Mandel JR, Dechaine JM, Marek LF, Burke JM (2011) Genetic diversity and population structure in cultivated sunflower and a comparison to its wild progenitor, Helianthus annuus L. Theor Appl Genet 123:693–704. https://doi.org/10.1007/s00122-011-1619-3
Mandel JR, Nambeesan S, Bowers JE, Marek LF, Ebert D, Rieseberg LH, Knapp SJ, Burke JM (2013) Association mapping and the genomic consequences of selection in sunflower. PLoS Genet 9:1–13. https://doi.org/10.1371/journal.pgen.1003378
Mangeon A, Junqueira RM, Sachetto-Martins G (2010) Functional diversity of the plant glycine-rich proteins superfamily. Plant Signal Behav 5:99–104. https://doi.org/10.4161/psb.5.2.10336
Nambeesan SU, Mandel JR, Bowers JE, Marek LF, Ebert D, Corbi J, Rieseberg LH, Knapp SJ, and Burke JM. (2015) Association mapping in sunflower (Helianthus annuus L.) reveals independent control of apical vs. basal branching. BMC Plant Biol. https://doi.org/10.1186/s12870-015-0458-9
Nordström K, Dahlbom J, Pragadheesh VS, Ghosh S, Olsson A, Dyakova O, Suresh SK, Olsson SB (2017) In situ modeling of multimodal floral cues attracting wild pollinators across environments. Proc Nat Acad Sci USA 114:13218–13223. https://doi.org/10.1073/pnas.1714414114
Oz M, Karasu A, Cakmak I, Goksoy AT, Metin ZT (2009) Effects of honeybee (Apis mellifera) pollination on seed set in hybrid sunflower (Helianthus annuus L.). Afr J Biotechnol 8:1037–1043. https://doi.org/10.4314/AJB.V8I6.60012
Portlas Z, Tetlie J, Prischmann-Voldseth D, Hulke BS, Prasifka JR (2018) Variation in floret size explains differences in wild bee visitation to cultivated sunflowers. Plant Genet Resour. https://doi.org/10.1017/S1479262118000072
Prasifka JR, Mallinger RE, Portlas ZM, Hulke BS, Fugate KK, Paradis T, Hampton ME, Carter CJ (2018) Using nectar-related traits to enhance crop-pollinator interactions. Front Plant Sci 9:812. https://doi.org/10.3389/FPLS.2018.00812
R Core Team (2013) R: A language and environment for statistical computing. R Found. Stat. Comput. Vienna, Austria 0: ISBN:3-900051-07-0
SAS Institute (2013) The SAS system for Windows. Release 9.4. SAS Inst., Cary, NC
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Tang S, Leon A, Bridges WC, Knapp SJ (2006) Quantitative trait loci for genetically correlated seed traits are tightly linked to branching and pericarp loci in sunflower. Crop Sci 46:721–734
USDA-NASS. (2018). Census of Agriculture, Ag Census Web Maps
Yue B, Cai X, Yuan W, Vick B, Hu J (2009) Mapping quantitative trait loci (QTL) controlling seed morphology and disk diameter in sunflower (Helianthus annuus L.). Helia 32:17–36
Zuber H, Davidian J-C, Aubert G, Aimé D, Belghazi M, Lugan R, Heintz D, Wirtz M, Hell R, Thompson R, Gallardo K (2010) The seed composition of arabidopsis mutants for the group 3 sulfate transporters indicates a role in sulfate translocation within developing seeds. Plant Physiol 154:913–926. https://doi.org/10.1104/pp.110.162123
Zywiec M, Delibes M, Fedriani JM (2012) Microgeographical, inter-individual, and intra-individual variation in the flower characters of Iberian pear Pyrus bourgaeana (Rosaceae). Oecologia 169:713–722. https://doi.org/10.1007/s00442-011-2232-5
Acknowledgements
The authors acknowledge the assistance of Jonathan Tetlie, Brady Koehler, and Michael Grove in the maintenance and collection of field samples for this work. The assistance of Brian Smart and Cloe Pogoda to the development of figures is also greatly appreciated. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Funding
This work was supported by funds from the USDA-Agricultural Research Service (project number 3060-21000-043-00D).
Author information
Authors and Affiliations
Contributions
JRP and BSH developed the experimental design; JRP, BF, and ZMP conducted and validated the phenotypic experiments; QMG, SR, and BSH developed the statistical design, conducted the association mapping and epistasis work, and wrote the manuscript; and all authors contributed to final review and acceptance of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
This work did not involve human participants or research on animals.
Additional information
Communicated by Stefan Hohmann.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Reinert, S., Gao, Q., Ferguson, B. et al. Seed and floret size parameters of sunflower are determined by partially overlapping sets of quantitative trait loci with epistatic interactions. Mol Genet Genomics 295, 143–154 (2020). https://doi.org/10.1007/s00438-019-01610-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-019-01610-7