Abstract
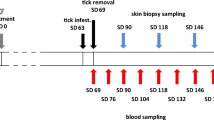
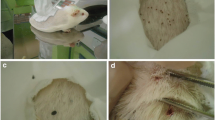
Specific pathogen-free dogs were experimentally infected with Borrelia burgdorferi sensu stricto using nymphal or adult female Ixodes scapularis ticks artificially infected with spirochetes by capillary feeding. The ticks were capillary fed B. burgdorferi isolate 610, previously isolated from a dog with Lyme disease and grown in BSK medium. This isolate induced clinical signs in the dogs similar to those for dogs infested with ticks naturally infected with B. burgdorferi. Adult ticks were more efficient than nymphs in transmitting spirochetes to the dogs. One of five dogs infested with nymphal ticks capillary fed B. burgdorferi was skin biopsy culture and serologically positive, and demonstrated lameness. In contrast, all five dogs infested with adult female ticks that had been capillary fed with B. burgdorferi were culture and serologically positive, with one dog developing lameness. The immunoblot profiles of dogs challenged with female ticks infected by capillary feeding (8 weeks post challenge) were similar to immunoblots (4 weeks post challenge) from dogs challenged with naturally infected females collected in the field. These studies demonstrated that B. burgdorferi cultured in BSK medium can be capillary fed to either nymphal or adult female ticks under laboratory controlled conditions for the purpose of transmitting the spirochete to dogs during the tick's blood meal. This tick infection system would be useful for a controlled and defined challenge of vaccinated and non-vaccinated dogs for proper evaluation of vaccine efficacy, which is difficult to achieve using field-collected ticks. Furthermore, this system may also be useful for investigation of the pathogenesis of Lyme disease, evaluation of the pathogenicity of new isolates of B. burgdorferi, or evaluation of antibiotic therapy.


Similar content being viewed by others
References
Appel, MJG, Allan S, Jacobson RH, Lauderdale TL, Chang YF, Shin SJ, Thomford JW, Todhunter RJ, Summers BA (1993) Experimental Lyme disease in dogs produces arthritis and persistent infection. J Infect Dis 167:651–664
Barbour AG (1984) Isolation and cultivation of Lyme disease spirochetes. Yale J Biol Med 57:521–525
Barbour AG, Tessier SL, Todd WJ (1983) Lyme disease spirochetes and ixodid tick spirochetes share a common surface antigenic determinant defined by a monoclonal antibody. Infect Immun 41:795–804
Barbour AG, Hayes SF, Heiland RA, Schrumpf ME, Tessier SL (1986) A Borrelia-specific monoclonal antibody binds to a flagellar epitope. Infect Immun 52:549–554
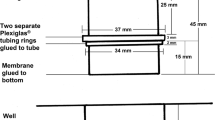
Broadwater AH, Sonenshine DE, Hynes WL, Ceraul SM, De Silva AM (2002) Glass capillary tube feeding: a method for infecting nymphal Ixodes scapularis (Acari: Ixodidae) with the Lyme disease spirochete Borrelia burgdorferi. J Med Entomol 39:285–292
Burgdorfer W (1957) Artificial feeding of ixodid ticks for studies on the transmission of disease agents. J Infect Dis 100:212–214
Burgdorfer W, Barbour AG, Hayes SF, Benach JL, Grunwaldt E, Davis JP (1982) Lyme disease—a tick-borne spirochetosis? Science 216:1317–1319
Burkot TR, Piesman J, Wirtz RA (1994) Quantitation of the Borrelia burgdorferi outer surface protein A in Ixodes scapularis: Fluctuations during the tick cycle, doubling times, and loss while feeding. J Infect Dis 170:883–889
Bushmich MS (1994) Lyme borreliosis in domestic animals. J Spirochet Tick Borne Dis 1:24–28
Chang Y-F, Novosel V, Chang C-F, Summers BA, Ma D-P, Chiang Y-W, Acree WM, Chu H-J, Shin S, Lein DH (2001) Experimental induction of chronic borreliosis in adult dogs exposed to Borrelia burgdorferi-infected ticks and treated with dexamethasone. Am J Vet Res 62:1104–1112
Chu YF, Chavez GJ, Blumer BM, Sebring RW, Wasmoen TL, Acree WM (1992) Immunogenicity and efficacy study of a commercial Borrelia burgdorferi bacterin. J Am Vet Med Assoc 201:403–411
Demaerschalck I, Messaoud AB, Kesel MD, Hoyois B, Lobet Y, Hoet P, Bigaignon G, Bollen A, Godfroid E (1995) Simultaneous presence of different Borrelia burgdorferi genospecies in biological fluids of Lyme disease patients. J Clin Microbiol 33:602–608
Fingerle V, Hauser U, Liegl G, Petko B, Preac-Mursic V, Wilske B (1995) Expression of outer surface proteins A and C of Borrelia burgdorferi in Ixodes ricinus. J Clin Microbiol 33:1867–1869
Gauthier DT, Mansfield LS (1999) Western immunoblot analysis for distinguishing vaccination and infection status with Borrelia burgdorferi (Lyme disease) in dogs. J Vet Diagn Invest 11:259–265
Gern L, Zhu Z, Aeschlimann A (1990) Development of Borrelia burgdorferi in Ixodes ricinus females during blood feeding. Ann Parasitol Hum Comp 65:89–93
Gern L, Toutoungi LN, Hu CM, Aeschlimann A (1991) Ixodes (Pholeoixodes) hexagonus, an efficient vector of Borrelia burgdorferi in the laboartory. Med Vet Entomol 5:431–435
Gern L, Schaible UE, Simon MM (1993) Mode of incoulation of the Lyme disease agent Borrelia burgdorferi influences infection and immune responses in inbred strains of mice. J Infect Dis 167:971–975
Gern L, Rais O, Capiau C, Hauser P, Lobet Y, Simoen E, Voet P, Pêtre J (1994) Immunization of mice by recombinant OspA preparations and protection against Borrelia burgdorferi infection by Ixodes ricinus tick bites. Immunol Lett 39:249–258
Greene RT, Levine JF, Breitschwerdt EB (1988) Clinical and serological evaluation of induced Borrelia burgdorferi infection in dogs. Am J Vet Res 49:752–757
Guttman DS, Wang PW, Wang IN, Bosler EM, Luft BJ, Dykhuizen DE (1996) Multiple infections of Ixodes scapularis ticks by Borrelia burgdorferi as revealed by single-strand conformation polymorphism analysis. J Clin Microbiol 34:652–656
Hu CM, Gern L, Aeschlimann A (1992) Changes in the protein profile and antigenicity of different Borrelia burgdorferi strains after reintroduction to Ixodes ricinus ticks. Parasite Immunol 14:415–427
Hu CM, Simon M, Kramer MD, Gern L (1996) Tick factors and in vitro cultivation influence the protein profile, antigenicity and pathogenicity of a cloned Borrelia garinii isolate from Ixodes ricinus hemolymph. Infection 24:251–257
Kornblatt AN, Urband PH, Steere AC (1985) Arthritis caused by Borrelia burgdorferi in dogs. J Am Vet Med Assoc 186:960–964
Kurtti TJ, Munderloh UG, Hughes CAN, Engstrom SM, Johnson RC. (1996) Resistance to tick-borne spirochete challenge induced by Borrelia burgdorferi strains that differ in expression of outer surface proteins. Infect Immun 64:4148–4153
Lacombe E, Rand PW, Smith RP (1993) Disparity of Borrelia burgdorferi infection rates of adult Ixodes dammini on deer and vegetation. J Infect Dis 167:1236–1238
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Levy SA, Dreesen DW (1992) Lyme borreliosis in dogs. Canine Pract 17:5–14
Levy SA, Magnarelli LA (1992) Relationship between development of antibodies to Borrelia burgdorferi in dogs and the subsequent development of limb/joint borreliosis. J Am Vet Med Assoc 200:344–347
Lissman BA (1990) Lyme disease in small animals. J Med Soc New Jersey 87:573–574
Lovrich SD, Callister SM, Lim LC, Schell RF (1993) Seroprotective groups among isolates of Borrelia burgdorferi. Infect Immun 61:4367–4374
Lovrich SD, Callister SM, DuChateau BK, Lim LC, Winfrey J, Day SP, Schell RF (1995) Abilities of OspA proteins from different seroprotective groups of Borrelia burgdorferi to protect hamsters from infection. Infect Immun 63:2113–2119
Macaluso KR, Sonenshine DE, Ceraul SM, Azad AF (2001) Infection and transovarial transmission of rickettsiae in Dermacentor variabilis ticks acquired by artificial feeding. Vector Borne Zoonotic Dis 1:45–53
Magnarelli LA, Anderson JF, Kaufman AF, Lieberman LL (1985) Borreliosis in dogs from southern Connecticut. J Am Vet Med Assoc 186:955–959
Magnarelli LA, Anderson JF, Schreier AB, Ficke CM (1987) Clinical and serologic studies of canine borreliosis. J Am Vet Med Assoc 191:1089–1094
Magnarelli LA, Anderson JF, Schreier AB (1990) Persistence of antibodies to Borrelia burgdorferi in dogs of New York and Connecticut. J Am Vet Med Assoc 196:1064–1068
Magnarelli LA, Andreadis TG, Stafford IKC, Holland CJ (1991) Rickettsiae and Borrelia burgdorferi in Ixodid ticks. J Clin Microbiol 29:2798–2804
Magnarelli LA, Flavell RA, Padula SJ, Anderson JF, Fikrig E (1997) Serologic diagnosis of canine and equine borreliosis: use of recombinant antigens in enzyme-linked immunosorbent assays. J Clin Microbiol 35:169–173
Marconi RT, Garon CF (1992) Development of polymerase chain reaction primer sets for diagnosis of Lyme disease and for species-specific identification of Lyme disease isolates by 16S rRNA signature nucleotide analysis. J Clin Microbiol 30:2830–2834
Nielsen A, Carr A (2002) Update on canine Lyme disease. Vet Med August:604–609
Pichon B, Godfroid E, Hoyois B, Bollen A, Rodhain F, Perez-Eid C (1995) Simultaneous infection of Ixodes ricinus nymphs by two Borrelia burgdorferi sensu lato species: possible implications for clinical manifestations. Emerg Infect Dis 1:89–90
Piesman J (1993) Standard system for infecting ticks (Acari: Ixodidae) with the Lyme disease spirochete, Borrelia burgdorferi. J Med Entomol 30:199–203
Piesman J, Sinsky RJ (1988) Ability of Ixodes scapularis, Dermacentor variabilis, and Amblyomma americanum (Acari: Ixodidae) to acquire, maintain, and transmit Lyme disease spirochetes (Borrelia burgdorferi). J Med Entomol 25:336–339
Piesman J, Mather TN, Sinsky RJ, Spielman A (1987) Duration of tick attachment and Borrelia burgdorferi transmission. J Clin Microbiol 25:557–558
Piesman J, Schneider BS, Zeidner NS (2001) Use of quantitative PCR to measure density of Borrelia burgdorferi in the midgut and salivary glands of feeding tick vectors. J Clin Microbiol 39:4145–4148
Pollack RJ, Telford III SR, Spielman A (1993) Standardization of medium for culturing Lyme disease spirochetes. J Clin Microbiol 31:1251–1255
Rechav Y, Zyzak M, Fielden LJ, Childs JE (1999) Comparison of methods for introducing and producing artificial infection of ixodid ticks (Acari: Ixodidae) with Ehrlichia chaffeensis. J Med Entomol 36:414–419
Roehrig JT, Piesman J, Hunt AR, Keen MG, Happ CM, Johnson BJB (1992) The hamster immune response to tick-transmitted Borrelia burgdorferi differs from the response to needle-inoculated, cultured organisms. J Immunol 149:3648–3653
Schaible UE, Gern L, Wallich R, Kramer MD, Prester M, Simon MM (1993) Distinct patterns of protective antibodies generated against Borrelia burgdorferi in mice experimentally infected with high and low doses of antigen. Immun Lett 36:219–226
Schwan TG, Burgdorfer W (1987) Antigenic changes of Borrelia burgdorferi as a result of in vitro cultivation. J Infect Dis 156:852–853
Schwan TG, Piesman J, Golde WT, Dolan MC, Rosa PA (1995) Induction of an outer surface protein on Borrelia burgdorferi during tick feeding. Proc Natl Acad Sci USA 92:2909–2913
Shaw SE, Day MJ, Birtles RJ, Breitschwerdt EB (2001) Tick-borne infectious diseases of dogs. Trends Parasitol 17:74–80
Shin SJ, Chang Y-F, Jacobson RH, Shaw E, Lauderdale T-L, Appel MJ, Lein DH (1993) Cross-reactivity between B. burgdorferi and other spirochetes affects specificity of serotests for detection of antibodies to the Lyme disease agent in dogs. Vet Microbiol 36:161–174
Straubinger RK, Chang Y-F, Jacobson RH, Appel MJG (1995) Sera from OspA-vaccinated dogs, but not those from tick-infected dogs, inhibit in vitro growth of Borrelia burgdorferi. J Clin Microbiol 33:2745–2751
Straubinger RK, Straubinger AF, Härter L, Jacobson RH, Chang Y-F, Summers BA, Erb HN, Appel MJG (1997) Borrelia burgdorferi migrates into joint capsules and causes an up-regulation of interleukin-8 in synovial membranes of dogs experimentally infected with ticks. Infect Immun 65:1273–1285
Straubinger RK, Summers BA, Chang YF, Appel MJG (1997) Persistence of Borrelia burgdorferi in experimentally infected dogs after antibiotic treatment. J Clin Microbiol 35:111–116
Straubinger RK, Straubinger AF, Summers BA, Jacobson RH (2000) Status of Borrelia burgdoferi infection after antibiotic treatment and the effects of corticosteroids: an experimental study. J Infect Dis 181:1069–1081
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedures and some applications. Proc Natl Acad Sci USA 76:4350–4354
Wasmoen TL, Sebring RW, Blumer BM, Chavez LG, Chu H, Acree W (1992) Examination of Koch's postulates for Borrelia burgdorferi as the causative agent of limb/joint dysfunction in dogs with borreliosis. J Am Vet Med Assoc 201:412–418
Xu Y, Kodner C, Coleman L, Johnson RC (1996) Correlation of plasmids with infectivity of Borrelia burgdorferi sensu stricto type strain B31. Infect Immun 64:3870–3876
Zung JL, Lewengrub S, Rudzinska MA, Spielman A, Telford SR, Piesman J (1989) Fine structural evidence for the penetration of the Lyme disease spirochete Borrelia burgdorferi through the gut and salivary tissues of Ixodes dammini. Can J Zool 67:1737–1748
Acknowledgements
This work is part of the M.S. dissertation of Jon Korshus. We thank Drs. Mark Mellencamp, Paul Runnels and Yu-Wei Chiang for advice and encouragement. The technical assistance of Jeffrey Crary, and Dr. Debra Dietrich was greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Korshus, J.B., Munderloh, U.G., Bey, R.F. et al. Experimental infection of dogs with Borrelia burgdorferi sensu stricto using Ixodes scapularis ticks artificially infected by capillary feeding. Med Microbiol Immunol 193, 27–34 (2004). https://doi.org/10.1007/s00430-003-0178-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-003-0178-x