Abstract
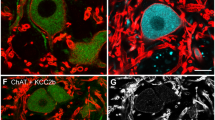
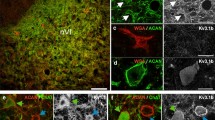
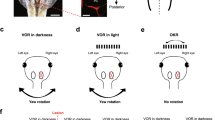
Medial rectus motoneurons are innervated by two main pontine inputs. The specific function of each of these two inputs remains to be fully understood. Indeed, selective partial deafferentation of medial rectus motoneurons, performed by the lesion of either the vestibular or the abducens input, initially induces similar changes in motoneuronal discharge. However, at longer time periods, the responses to both lesions are dissimilar. Alterations on eye movements and motoneuronal discharge induced by vestibular input transection recover completely 2 months post-lesion, whereas changes induced by abducens internuclear lesion are more drastic and permanent. Functional recovery could be due to some kind of plastic process, such as reactive synaptogenesis, developed by the remaining intact input, which would occupy the vacant synaptic spaces left after lesion. Herein, by means of confocal microscopy, immunocytochemistry and retrograde labeling, we attempt to elucidate the possible plastic processes that take place after partial deafferentation of medial rectus motoneuron. 48 h post-injury, both vestibular and abducens internuclear lesions produced a reduced synaptic coverage on these motoneurons. However, 96 h after vestibular lesion, there was a partial recovery in the number of synaptic contacts. This suggests that there was reactive synaptogenesis. This recovery was preceded by an increase in somatic neurotrophin content, suggesting a role of these molecules in presynaptic axonal sprouting. The rise in synaptic coverage might be due to terminal sprouting performed by the remaining main input, i.e., abducens internuclear neurons. The present results may improve the understanding of this apparently redundant input system.




Similar content being viewed by others
References
Atwal JK, Massie B, Miller FD, Kaplan DR (2000) The TrkB-Shc site signals neuronal survival and local axon growth via MEK and PI3-kinase. Neuron 27:265–277. https://doi.org/10.1016/S0896-6273(00)00035-0
Aw ST, Chen L, Todd MJ, Barnett MH, Halmagyi GM (2017) Vestibulo-ocular reflex deficits with medial longitudinal fasciculus lesions. J Neurol 264:2119–2129. https://doi.org/10.1007/s00415-017-8607-8
Baker R, Highstein SM (1978) Vestibular projections to interno rectus subdivision of oculomotor nucleus. J Neurophysiol 41:1629–1646. https://doi.org/10.1152/jn.1978.41.6.1629
Bäurle J, Gorver BG, Grüsser-Cornehls U (1992) Plasticity of GABAergic terminals in Deiters’ nucleus of weaver mutant and normal mice: a quantitative light microscope study. Brain Res 591:305–318. https://doi.org/10.1016/0006-8993(92)91712-N
Benítez-Temiño B, Morcuende S, Mentis GZ, de la Cruz RR, Pastor AM (2004) Expression of Trk receptors in the oculomotor system of the adult cat. J Comp Neurol 473:538–552. https://doi.org/10.1002/cne.20095
Biefang DC (1978) The course of direct projections from the abducens nucleus to the contralateral interno rectus subdivision of the oculomotor nucleus in the cat. Brain Res 145:277–289. https://doi.org/10.1016/0006-8993(78)90862-4
Bohlen MO, Warren S, May PJ (2017) A central mesencephalic reticular formation projection to medial rectus motoneurons supplying singly and multiply innervated extraocular muscle fibers. J Comp Neurol 525:2000–2018. https://doi.org/10.1002/cne.24187
Büttner-Ennever JA (2006) The extraocular motor nuclei: organization and functional neuroanatomy. In: Büttner-Ennever JA (ed) Neuroanatomy of the oculomotor system, progress in brain research, vol 151. Elveiser, Amsterdam, pp 95–126. https://doi.org/10.1016/s0079-6123(05)51004-5
Carpenter MB, Carleton SC (1983) Comparison of vestibular and abducens internuclear projections to the interno rectus subdivision of the oculomotor nucleus in the monkey. Brain Res 274:144–149. https://doi.org/10.1016/0006-8993(83)90530-9
Choi SY, Kim HJ, Kim JS (2017) Impaired vestibular responses in internuclear ophthalmoplegia: association and dissociation. Neurology 89:2476–2480. https://doi.org/10.1212/WNL.0000000000004745
Cohen-Cory S, Fraser SE (1995) Effects of brain-derived neurotrophic factor on optic axon branching and remodeling in vivo. Nature 378:192–196. https://doi.org/10.1038/378192a0
Cotman CW, Nadler JV (1978) Reactive synaptogenesis in the hippocampus. In: Cotman CW (ed) Neuronal plasticity. Raven, New York, pp 227–272
Cotman CW, Nieto-Sampedro M (1984) Cell biology of synaptic plasticity. Science 225:1287–1294. https://doi.org/10.1126/science.6382610
Cotman CW, Nieto-Sampedro M, Harris EW (1981) Synapse replacement in the nervous system of adult vertebrates. Physiol Rev 61:684–784. https://doi.org/10.1152/physrev.1981.61.3.684
Davis-López de Carrizosa MA, Morado-Díaz CJ, Tena JJ, Benítez-Temiño B, Pecero ML, Morcuende SR, de la Cruz RR, Pastor AM (2009) Complementary actions of BDNF and neurotrophin-3 on the firing patterns and synaptic composition of motoneurons. J Neurosci 29:575–587. https://doi.org/10.1523/JNEUROSCI.5312-08.2009
Davis-López de Carrizosa MA, Morado-Díaz CJ, Morcuende S, de la Cruz RR, Pastor AM (2010) Nerve growth factor regulates the firing patterns and synaptic composition of motoneurons. J Neurosci 30:8308–8319. https://doi.org/10.1523/JNEUROSCI.0719-10.2010
de la Cruz RR, Pastor AM, Martínez-Guijarro FJ, López-García C, Delgado-García JM (1992) Role of GABA in the extraocular motor nuclei of the cat: a postembedding immunocytochemical study. Neuroscience 5:911–929. https://doi.org/10.1016/0306-4522(92)90529-B
de la Cruz RR, Pastor AM, Martínez-Guijarro FJ, López-García C, Delgado-García JM (1998) Localization of parvalbumin, calretinin, and calbindin D-28k in identified extraocular motoneurons and internuclear neurons of the cat. J Comp Neurol 390:377–391. https://doi.org/10.1002/(SICI)1096-9861(19980119)390:3%3c377:AID-CNE6%3e3.0.CO;2-Z
Delgado-García JM, del Pozo F, Baker R (1986) Behavior of neurons in the abducens nucleus of the alert cat-II. Internuclear neurons. Neuroscience 17:953–973. https://doi.org/10.1016/0306-4522(86)90073-4
Dinocourt C, Gallagher SE, Thompson SM (2006) Injury-induced axonal sprouting in the hippocampus is initiated by activation of trkB receptors. Eur J Neurosci 24:1857–1866. https://doi.org/10.1111/j.1460-9568.2006.05067.x
Endo T, Spenger C, Tominaga T, Brene S, Olson L (2007) Cortical sensory map rearrangement after spinal cord injury: fMRI responses linked to Nogo signalling. Brain 130:2951–2961. https://doi.org/10.1093/brain/awm237
Federoff HJ, Grabczyk E, Fishman MC (1988) Dual regulation of GAP-43 gene expression by nerve growth factor and glucocorticoids. J Biol Biochem 263:19290–19295
Ferri CC, Ghasemlou N, Bisby MA, Kawaja MD (2002) Nerve growth factor alters p75 neurotrophin receptor-induced effects in mouse facial motoneurons following axotomy. Brain Res 950:180–185. https://doi.org/10.1016/S0006-8993(02)03035-4
Furuya N, Markham CH (1981) Arborization of axons in oculomotor nucleus identified by vestibular stimulation and intra-axonal injection of horseradish peroxidase. Exp Brain Res 43:289–303. https://doi.org/10.1007/BF00238370
Gallo G, Letourneau PC (1998) Localized sources of neurotrophins initiate axin collateral sprouting. J Neurosci 18:5403–5414. https://doi.org/10.1523/JNEUROSCI.18-14-05403.1998
Gamlin PD, Gnadt JW, Mays L (1989) Lidocaine-induced unilateral internuclear ophthalmoplegia: effects on convergence and conjugate eye movements. J Neurophysiol 62:82–95
Geremia NM, Pettersson LME, Hasmatali JC, Hryciw T, Danielsen N, Schreyer DJ, Valerie MK, Verge A (2010) Endogenous BDNF regulates induction of intrinsic neuronal growth programs in injured sensory neurons. Exp Neurol 223:128–142. https://doi.org/10.1016/j.expneurol.2009.07.022
Gomez-Pinilla F, Ying Z, Roy RR, Hodgson J, Edgerton VR (2004) Afferent input modulates neurotrophins and synaptic plasticity in the spinal cord. J Neurophysiol 92:3423–3432. https://doi.org/10.1152/jn.00432.2004
González MJ, Pombal MA, Rodicio MC, Anadón R (1998) Internuclear neurons of the ocular motor system of the larval sea lamprey. J Comp Neurol 401:1–15
Grinnell AD, Rheuben MB (1979) The physiology, pharmacology and trophic effectiveness of synapses formed by autonomic preganglionic nerves on frog skeletal muscle. J Physiol 289:219–240. https://doi.org/10.1113/jphysiol.1979.sp012734
Hernández RG, Benítez-Temiño B, Morado-Díaz CJ, Davis-López de Carrizosa MA, de la Cruz RR, Pastor AM (2017a) Effects of selective deafferentation on the discharge characteristics of medial rectus motoneurons. J Neurosci 37:9172–9188. https://doi.org/10.1523/JNEUROSCI.1391-17.2017
Hernández RG, Silva-Hucha S, Morcuende S, de la Cruz RR, Pastor AM, Benítez-Temiño B (2017b) Extraocular motor system exhibits a higher expression of neurotrophins when compared with other brainstem motor systems. Front Neurosci 11:399. https://doi.org/10.3389/fnins.2017.00399
Highstein SM, Baker R (1978) Excitatory termination of abducens internuclear neurons on medial rectus motoneurons: relationship to syndrome of internuclear ophthalmoplegia. J Neurophysiol 41:1647–1661. https://doi.org/10.1152/jn.1978.41.6.1647
Highstein SM, Holstein GR (2006) The anatomy of the vestibular nuclei. In: Büttner-Ennever JA (ed) Neuroanatomy of the oculomotor system, progress in brain research, vol 151. Elsevier, Amsterdam, pp 157–203. https://doi.org/10.1016/s0079-6123(05)51006-9
Him A, Dutia MB (2001) Intrinsic excitability changes in vestibular nucleus neurons after unilateral deafferentation. Brain Res 908:58–66. https://doi.org/10.1016/S0006-8993(01)02600-2
Inoue A, Sanes JR (1997) Lamina-specific connectivity in the brain: regulation by N-cadherin, neurotrophins and glycoconjugates. Science 276:1428–1431. https://doi.org/10.1126/science.276.5317.1428
Izawa Y, Sugiuchi Y, Shinoda Y (1999) Neural organization from the superior colliculus to motoneurons in the horizontal oculomotor system of the cat. J Neurophysiol 81:2597–2611
Jakeman LB, Wei P, Guan Z, Stokes BT (1998) Brain-derived neurotrophic factor stimulates hindlimb stepping and sprouting of cholinergic fibers after spinal cord injury. Exp Neurol 154:170–184. https://doi.org/10.1006/exnr.1998.6924
Johnson RA, Okragly AJ, Haak-Frendscho M, Mitchell GS (2000) Cervical dorsal rhizotomy increases brain-derived neurotrophic factor and neurotrophin-3 expression in the ventral spinal cord. J Neurosci 20:1–5. https://doi.org/10.1523/JNEUROSCI.20-10-j0005.2000
Keast JR, Kepper ME (2001) Differential regulation of TrkA and p75 in noradrenergic pelvic autonomic ganglion cells after deafferentation of their cholinergic neighbours. Eur J Neurosci 13:211–220. https://doi.org/10.1111/j.1460-9568.2001.01374.x
Krassioukov AV, Weaver LC (1996) Morphological changes in sympathetic preganglionic neurons after spinal cord injury in rats. Neuroscience 1:211–225. https://doi.org/10.1016/0306-4522(95)00294-S
Lee KS, Stanford EJ, Cotman CW, Lynch GS (1977) Ultrastructural evidence for bouton proliferation in the partially deafferented dentate gyrus of the adult rat. Exp Brain Res 29:475–485. https://doi.org/10.1007/BF00236185
Lindsay RM (1994) Trophic protection of motor neurons: clinical potential in motor neuron diseases. J Neurol 242:S8–S11. https://doi.org/10.1007/BF00939232
Lund RD, Lund JS (1971) Modifications of synaptic patterns in the superior colliculus of the rat during development and following deafferentation. Vision Res Suppl 3:281–298. https://doi.org/10.1016/0042-6989(71)90046-0
Masliah E, Fagan AM, Terry RD, DeTeresa R, Mallory M, Gage FH (1991) Reactive synaptogenesis assessed by synaptophysin immunoreactivity is associated with GAP-43 in the dentate gyrus of the adult rat. Exp Neurol 113:131–142. https://doi.org/10.1016/0014-4886(91)90169-D
Matthews DA, Cotman C, Lynch G (1976) An electron microscopic study of lesion-induced synaptogenesis in the dentate gyrus of the adult rat. II. Reappearance of morphologically normal synaptic contacts. Brain Res 115:23–41. https://doi.org/10.1016/0006-8993(76)90820-9
McCrea RA, Baker R (1985) Anatomical connections of the nucleus prepositus of the cat. J Comp Neurol 237:377–407
Mendell LM, Sassoon EM, Wall PD (1978) Properties of synaptic linkage form long-ranging afferents onto dorsal horn neurons in normal and deafferented cats. J Physiol 285:299–310. https://doi.org/10.1113/jphysiol.1978.sp012572
Mohiuddin L, Fernandez K, Tomlinson DR, Fernyhough P (1995) Nerve growth factor and neurotrophin-3 enhance neurite outgrowth and up-regulate the levels of messenger RNA for growth-associated protein GAP-43 and Tal a-tubulin in cultured adult rat sensory neurons. Neurosci Lett 185:20–23. https://doi.org/10.1016/0304-3940(94)11215-5
Morcuende S, Matarredona ER, Benítez-Temiño B, Muñoz-Hernández R, Pastor AM, de la Cruz RR (2011) Differential regulation of the expression of neurotrophin receptors in rat extraocular motoneurons after lesion. J Comp Neurol 519:2335–2352. https://doi.org/10.1002/cne.22630
Morcuende S, Muñoz-Hernández R, Benítez-Temiño B, Pastor AM, de la Cruz RR (2013) Neuroprotective effects of NGF, BDNF, NT-3 and GDNF on axotomized extraocular motoneurons in neonatal rats. Neuroscience 250:31–48. https://doi.org/10.1016/j.neuroscience.2013.06.050
Nadler JV, Perry BW, Cotman CW (1980a) Selective reinnervation of hippocampal area CA1 and the fascia dentata after destruction of CA3-CA4 afferents with kainic acid. Brain Res 182:1–9. https://doi.org/10.1016/0006-8993(80)90825-2
Nadler JV, Perry BW, Gentry C, Cotman CW (1980b) Loss and reacquisition of hippocampal synapses after selective destruction of CA3-CA4 afferents with kainic acid. Brain Res 191:387–403. https://doi.org/10.1016/0006-8993(80)91289-5
Nguyen LT, Spencer RF (1999) Abducens internuclear and ascending tract of Deiters inputs to medial rectus motoneurons in the cat oculomotor nucleus: neurotransmitters. J Comp Neurol 411:73–86. https://doi.org/10.1002/(SICI)1096-9861(19990816)411:1%3c73:AID-CNE6%3e3.0.CO;2-7
Nguyen LT, Baker R, Spencer RF (1999) Abducens internuclear and ascending tract of Deiters inputs to medial rectus motoneurons in the cat oculomotor nucleus: synaptic organization. J Comp Neurol 405:141–159. https://doi.org/10.1002/(SICI)1096-9861(19990308)405:2%3c141:AID-CNE1%3e3.0.CO;2-%23
Nja A, Purves D (1978) The effects of nerve growth factor and its antiserum on synapses in the superior cervical ganglion of the guinea pig. J Physiol 277:53–75. https://doi.org/10.1113/jphysiol.1978.sp012260
Paxinos G, Watson C (2009) The rat brain in stereotaxic coordinates. Elsevier, Amsterdam
Perrone-Bizzozero NI, Neve RL, Irwin N, Lewis S, Fischer I, Benowitz LI (1991) Post-transcriptional regulation of GAP-43 mRNA levels during neuronal differentiation and nerve regeneration. Mol Cell Neurosci 2:402–409. https://doi.org/10.1016/1044-7431(91)90027-L
Perrone-Bizzozero NI, Cansino VV, Kohn DT (1993) Posttranscriptional regulation of GAP-43 gene expression in PC12 cells through protein kinase C-dependent stabilization of the mRNA. J Cell Biol 120:1263–1270. https://doi.org/10.1083/jcb.120.5.1263
Puzdrowski RL, Leonard RB (1994) Vestibulo-oculomotor connections in an elasmobranch fish, the Atlantic stingray, Dasyatis sabina. J Comp Neurol 339:587–597
Reisine H, Highstein SM (1979) The ascending tract of Deiters’ conveys a head velocity signal to medial rectus motoneurons. Brain Res 170:172–176. https://doi.org/10.1016/0006-8993(79)90949-1
Reisine H, Strassman A, Highstein SM (1981) Eye position and eye velocity signals are conveyed to medial rectus motoneurons in the alert cat by the ascending tract of Deiters’. Brain Res 211:153–157. https://doi.org/10.1016/0006-8993(81)90075-5
Rotstein DL, Chen H, Wilton AS, Kwong JC, Marrie RA, Gozdyra P, Krysko KM, Kopp A, Copes R, Tu K (2018) Temporal trends in multiple sclerosis prevalence and incidence in a large population. Neurology 90:e1435–e1441. https://doi.org/10.1212/wnl.0000000000005331
Ruit KG, Snider WD (1991) Administration or deprivation of nerve growth factor during development permanently alters neuronal geometry. J Comp neurol 314:106–131. https://doi.org/10.1002/cne.903140110
Sanna MD, Ghelardini C, Galeotti N (2017) HuD-mediated distinct BDNF regulatory pathways promote regeneration after nerve injury. Brain Res 1659:55–63. https://doi.org/10.1016/j.brainres.2017.01.019
Scott AL, Borisoff JF, Ramer MS (2005) Deafferentation and neurotrophin-mediated intraspinal sprouting: a central role for the p75 neurotrophin receptor. Eur J Neurosci 21:81–92. https://doi.org/10.1111/j.1460-9568.2004.03838.x
Sedivec MJ, Ovelmen-Levitt J, Karp R, Mendell LM (1983) Increase in nociceptive input to spinocervical tract neurons following chronic partial deafferentation. J Neurosci 3:1511–1519. https://doi.org/10.1523/JNEUROSCI.03-07-01511.1983
State FA, Dessouky HI (1977) Effect of length of the distal stump of transected nerve upon the rate of degeneration of taste buds. Acta Anat 98:353–360. https://doi.org/10.1159/000144812
Straka H, Baker R (2013) Vestibular blueprint in early vertebrates. Front Neural Circuits 7:182. https://doi.org/10.3389/fncir.2013.00182
Tessler A, Glazer E, Artymyshyn R, Murray M, Goldberger ME (1980) Recovery of substance P in the cat spinal cord after unilateral lumbosacral deafferentation. Brain Res 191:459–470. https://doi.org/10.1016/0006-8993(80)91294-9
Walls GL (1962) The evolutionay history of eye movements. Vision Res 2:69–80
Zhang Y, Gamlin PD, Mays LE (1991) Antidromic identification of midbrain near response cells projecting to the oculomotor nucleus. Exp Brain Res 84:525–528
Zhang B, Goldberger ME, Wu LF, Murray M (1995) Plasticity of complex terminals in lamina II in partially deafferented spinal cord: the cat spared root preparation. Exp Neurol 132:186–193. https://doi.org/10.1016/0014-4886(95)90024-1
Acknowledgements
This work was supported by MICINN-FEDER Grant BFU2015-64515-P and PGC2018-094654-B-I00. RGH was a fellowship holder of the Universidad de Sevilla. Confocal microscopy was carried out at the Central Services of the Universidad de Sevilla (CITIUS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare to have no conflict of interests.
Ethical approval
All experimental procedures were in compliance with the European Union Directive on the protection of animals used for scientific purposes (2010/63/EU), and the Spanish legislation (RD 53/2013/BOE 34/11370-421).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hernández, R.G., Djebari, S., Vélez-Ortiz, J.M. et al. Short-term plasticity after partial deafferentation in the oculomotor system. Brain Struct Funct 224, 2717–2731 (2019). https://doi.org/10.1007/s00429-019-01929-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-019-01929-2