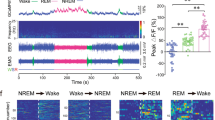
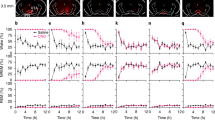
Abstract
The midbrain dopamine system via the dorsal and ventral striatum regulates a wide range of behaviors. To dissect the role of dopaminergic projections to the dorsal striatum (nigrostriatal projection) and ventral striatum (mesolimbic projection) in sleep–wake behavior, we selectively chemogenetically stimulated nigrostriatal or mesolimbic projections and examined the resulting effects on sleep in rats. Stimulation of nigrostriatal pathways increased sleep and EEG delta power, while stimulation of mesolimbic pathways decreased sleep and reduced cortical EEG power. These results indicate that midbrain dopamine signaling in the dorsal or ventral striatum promotes sleep or wake, respectively.







Similar content being viewed by others
References
Belaid H, Adrien J, Laffrat E, Tande D, Karachi C, Grabli D, Arnulf I, Clark SD, Drouot X, Hirsch EC, Francois C (2014) Sleep disorders in Parkinsonian macaques: effects of L-dopa treatment and pedunculopontine nucleus lesion. J Neurosci 34(27):9124–9133. https://doi.org/10.1523/JNEUROSCI.0181-14.2014
Bjorklund A, Dunnett SB (2007) Dopamine neuron systems in the brain: an update. Trends Neurosci 30(5):194–202. https://doi.org/10.1016/j.tins.2007.03.006
Chen MC, Ferrari L, Sacchet MD, Foland-Ross LC, Qiu MH, Gotlib IH, Fuller PM, Arrigoni E, Lu J (2015) Identification of a direct GABAergic pallidocortical pathway in rodents. Eur J Neurosci 41(6):748–759. https://doi.org/10.1111/ejn.12822
Cooper AJ, Stanford IM (2001) Dopamine D2 receptor mediated presynaptic inhibition of striatopallidal GABA(A) IPSCs in vitro. Neuropharmacology 41(1):62–71
Dimpfel W (2008) Pharmacological modulation of dopaminergic brain activity and its reflection in spectral frequencies of the rat electropharmacogram. Neuropsychobiology 58(3–4):178–186. https://doi.org/10.1159/000191124
Eban-Rothschild A, Rothschild G, Giardino WJ, Jones JR, de Lecea L (2016) VTA dopaminergic neurons regulate ethologically relevant sleep–wake behaviors. Nat Neurosci 19(10):1356–1366. https://doi.org/10.1038/nn.4377
Feenstra MG, Botterblom MH, Mastenbroek S (2000) Dopamine and noradrenaline efflux in the prefrontal cortex in the light and dark period: effects of novelty and handling and comparison to the nucleus accumbens. Neuroscience 100(4):741–748
Fifel K, Vezoli J, Dzahini K, Claustrat B, Leviel V, Kennedy H, Procyk E, Dkhissi-Benyahya O, Gronfier C, Cooper HM (2014) Alteration of daily and circadian rhythms following dopamine depletion in MPTP treated non-human primates. PLoS One 9(1):e86240. https://doi.org/10.1371/journal.pone.0086240
Fuller PM, Sherman D, Pedersen NP, Saper CB, Lu J (2011) Reassessment of the structural basis of the ascending arousal system. J Comp Neurol 519(5):933–956. https://doi.org/10.1002/cne.22559
Gerashchenko D, Blanco-Centurion CA, Miller JD, Shiromani PJ (2006) Insomnia following hypocretin2-saporin lesions of the substantia nigra. Neuroscience 137(1):29–36. https://doi.org/10.1016/j.neuroscience.2005.08.088
Grillner S, Robertson B (2016) The basal ganglia over 500 million years. Curr Biol 26(20):R1088–R1100. https://doi.org/10.1016/j.cub.2016.06.041
Guo CN, Machado NL, Zhan SQ, Yang XF, Yang WJ, Lu J (2016) Identification of cholinergic pallidocortical neurons. CNS Neurosci Ther 22(10):863–865. https://doi.org/10.1111/cns.12602
Hirayama M, Nakamura T, Hori N, Koike Y, Sobue G (2008) The video images of sleep attacks in Parkinson’s disease. Mov Disord 23(2):288–290. https://doi.org/10.1002/mds.21830
Isaac SO, Berridge CW (2003) Wake-promoting actions of dopamine D1 and D2 receptor stimulation. J Pharmacol Exp Ther 307(1):386–394. https://doi.org/10.1124/jpet.103.053918
Jones BE, Bobillier P, Pin C, Jouvet M (1973) The effect of lesions of catecholamine-containing neurons upon monoamine content of the brain and EEG and behavioral waking in the cat. Brain Res 58(1):157–177. https://doi.org/10.1016/0006-8993(73)90830-5
Lai YY, Shalita T, Hajnik T, Wu JP, Kuo JS, Chia LG, Siegel JM (1999) Neurotoxic N-methyl-d-aspartate lesion of the ventral midbrain and mesopontine junction alters sleep–wake organization. Neuroscience 90(2):469–483
Lai YY, Hsieh KC, Nguyen D, Peever J, Siegel JM (2008) Neurotoxic lesions at the ventral mesopontine junction change sleep time and muscle activity during sleep: an animal model of motor disorders in sleep. Neuroscience 154(2):431–443. https://doi.org/10.1016/j.neuroscience.2008.03.085
Laloux C, Derambure P, Houdayer E, Jacquesson JM, Bordet R, Destee A, Monaca C (2008a) Effect of dopaminergic substances on sleep/wakefulness in saline- and MPTP-treated mice. J Sleep Res 17(1):101–110. https://doi.org/10.1111/j.1365-2869.2008.00625.x
Laloux C, Derambure P, Kreisler A, Houdayer E, Brueziere S, Bordet R, Destee A, Monaca C (2008b) MPTP-treated mice: long-lasting loss of nigral TH-ir neurons but not paradoxical sleep alterations. Exp Brain Res 186(4):635–642. https://doi.org/10.1007/s00221-008-1268-1
Lipford MC, Silber MH (2012) Long-term use of pramipexole in the management of restless legs syndrome. Sleep Med 13(10):1280–1285. https://doi.org/10.1016/j.sleep.2012.08.004
Lu J, Greco MA, Shiromani P, Saper CB (2000) Effect of lesions of the ventrolateral preoptic nucleus on NREM and REM sleep. J Neurosci 20(10):3830–3842
Lu J, Zhang YH, Chou TC, Gaus SE, Elmquist JK, Shiromani P, Saper CB (2001) Contrasting effects of ibotenate lesions of the paraventricular nucleus and subparaventricular zone on sleep–wake cycle and temperature regulation. J Neurosci 21(13):4864–4874
Lu J, Jhou TC, Saper CB (2006) Identification of wake-active dopaminergic neurons in the ventral periaqueductal gray matter. J Neurosci 26(1):193–202. https://doi.org/10.1523/JNEUROSCI.2244-05.2006
Malenka RC, Neslter EJ, Hyman ES (2009) Widely projecting systems: monoamines, acetylcholine, and orexin. In: Sydor A, Brown RY (eds) Molecular neuropharmacology: a foundation for clinical neuroscience, 2nd edn. McGraw-Hill Medical, New York, pp 147–148
Nambu A (2008) Seven problems on the basal ganglia. Curr Opin Neurobiol 18(6):595–604. https://doi.org/10.1016/j.conb.2008.11.001
Oishi Y, Suzuki Y, Takahashi K, Yonezawa T, Kanda T, Takata Y, Cherasse Y, Lazarus M (2017) Activation of ventral tegmental area dopamine neurons produces wakefulness through dopamine D2-like receptors in mice. Brain Struct Funct. https://doi.org/10.1007/s00429-017-1365-7
Ongini E, Bonizzoni E, Ferri N, Milani S, Trampus M (1993) Differential effects of dopamine D-1 and D-2 receptor antagonist antipsychotics on sleep–wake patterns in the rat. J Pharmacol Exp Ther 266(2):726–731
Paus S, Brecht HM, Koster J, Seeger G, Klockgether T, Wullner U (2003) Sleep attacks, daytime sleepiness, and dopamine agonists in Parkinson’s disease. Mov Disord 18(6):659–667. https://doi.org/10.1002/mds.10417
Paxinos G, Watson C (2009) The rat brain. Stereotaxic coordinates, 6th edn. Elsevier Inc, Amsterdam
Qiu MH, Vetrivelan R, Fuller PM, Lu J (2010) Basal ganglia control of sleep–wake behavior and cortical activation. Eur J Neurosci 31(3):499–507. https://doi.org/10.1111/j.1460-9568.2009.07062.x
Qiu MH, Chen MC, Fuller PM, Lu J (2016a) Stimulation of the pontine parabrachial nucleus promotes wakefulness via extra-thalamic forebrain circuit nodes. Curr Biol 26(17):2301–2312. https://doi.org/10.1016/j.cub.2016.07.054
Qiu MH, Chen MC, Wu J, Nelson D, Lu J (2016b) Deep brain stimulation in the globus pallidus externa promotes sleep. Neuroscience 322:115–120. https://doi.org/10.1016/j.neuroscience.2016.02.032
Qiu MH, Yao QL, Vetrivelan R, Chen MC, Lu J (2016c) Nigrostriatal dopamine acting on globus pallidus regulates sleep. Cereb Cortex 26(4):1430–1439. https://doi.org/10.1093/cercor/bhu241
Qu WM, Huang ZL, Xu XH, Matsumoto N, Urade Y (2008) Dopaminergic D1 and D2 receptors are essential for the arousal effect of modafinil. J Neurosci 28(34):8462–8469. https://doi.org/10.1523/JNEUROSCI.1819-08.2008
Querejeta E, Delgado A, Valdiosera R, Erlij D, Aceves J (2001) Intrapallidal D2 dopamine receptors control globus pallidus neuron activity in the rat. Neurosci Lett 300(2):79–82
Schultz W (2007) Multiple dopamine functions at different time courses. Annu Rev Neurosci 30:259–288. https://doi.org/10.1146/annurev.neuro.28.061604.135722
Smith AD, Olson RJ, Justice JB Jr (1992) Quantitative microdialysis of dopamine in the striatum: effect of circadian variation. J Neurosci Methods 44(1):33–41
Taylor SR, Badurek S, Dileone RJ, Nashmi R, Minichiello L, Picciotto MR (2014) GABAergic and glutamatergic efferents of the mouse ventral tegmental area. J Comp Neurol 522(14):3308–3334. https://doi.org/10.1002/cne.23603
Towne C, Raoul C, Schneider BL, Aebischer P (2008) Systemic AAV6 delivery mediating RNA interference against SOD1: neuromuscular transduction does not alter disease progression in fALS mice. Mol Ther 16(6):1018–1025. https://doi.org/10.1038/mt.2008.73
Towne C, Schneider BL, Kieran D, Redmond DE Jr, Aebischer P (2010) Efficient transduction of non-human primate motor neurons after intramuscular delivery of recombinant AAV serotype 6. Gene Ther 17(1):141–146. https://doi.org/10.1038/gt.2009.119
Videnovic A, Noble C, Reid KJ, Peng J, Turek FW, Marconi A, Rademaker AW, Simuni T, Zadikoff C, Zee PC (2014) Circadian melatonin rhythm and excessive daytime sleepiness in Parkinson disease. JAMA Neurol 78:89. https://doi.org/10.1001/jamaneurol.2013.6239
Zhang JP, Xu Q, Yuan XS, Cherasse Y, Schiffmann SN, de Kerchove DA, Qu WM, Urade Y, Lazarus M, Huang ZL, Li RX (2013) Projections of nucleus accumbens adenosine A2A receptor neurons in the mouse brain and their implications in mediating sleep–wake regulation. Front Neuroanat 7:43. https://doi.org/10.3389/fnana.2013.00043
Acknowledgements
This work was supported by National Natural Science Foundation of China 31171049 and 81771430 (M. H. Q.), the Scientific Research Foundation for Returned Overseas Chinese Scholars, State Education Ministry (M. H. Q.), and NIH NS061841 and NS095986 (J. L.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qiu, MH., Zhong, ZG., Chen, M.C. et al. Nigrostriatal and mesolimbic control of sleep–wake behavior in rat. Brain Struct Funct 224, 2525–2535 (2019). https://doi.org/10.1007/s00429-019-01921-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-019-01921-w