Abstract
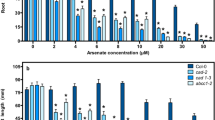
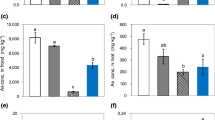
Several species of fern from the Pteris genus are able to accumulate extremely high concentrations of arsenic (As) in the fronds. We have conducted short-term unidirectional As influx and translocation experiments with 73As-radiolabeled arsenate, and found that the concentration-dependent influx of arsenate into roots was significantly larger in two of these As-hyperaccumulating species, Pteris vittata (L.) and Pteris cretica cv. Mayii (L.), than in Nephrolepis exaltata (L.), a non-accumulating fern. The arsenate influx could be described by Michaelis-Menten kinetics and the kinetic parameter K m was found to be lower in the Pteris species, indicating higher affinity of the transport protein for arsenate. Quantitative analysis of kinetic parameters showed that phosphate inhibited arsenate influx in a directly competitive manner, consistent with the hypothesis that arsenate enters plant roots on a phosphate-transport protein. The significantly augmented translocation of arsenic to the shoots that was seen in these As hyperaccumulator species is proposed to be due to a combination of the increased root influx and also decreased sequestration of As in the roots, as a larger fraction of As could be extracted from roots of the Pteris species than from roots of N. exaltata. This leaves a larger pool of mobile As available for translocation to the shoot, probably predominantly as arsenite.





Similar content being viewed by others
Abbreviations
- As V :
-
Arsenate
- As III :
-
Arsenite
- K m :
-
Michaelis-Menten constant
- P i :
-
Phosphate
- V max :
-
Maximum rate of an enzyme-catalyzed reaction
References
Ganje TJ, Rains DW (1982) Arsenic. In: Page A, Miller R, Keeney D (eds) Methods of soil analysis. Part 2: chemical and microbiological properties, 2nd edn. Soil Science Society of America, Madison, WI, pp 385–402
Hartley-Whitaker J, Ainsworth G, Vooijs R, Bookum WT, Schat H, Meharg AA (2001) Phytochelatins are involved in differential arsenate tolerance in Holcus lanatus. Plant Physiol 126:299–306
Kochian LV (2000) Molecular physiology of mineral nutrient acquisition, transport and utilization. In: Buchanan D, Gruissem W, Jones R (eds) Biochemistry and molecular biology of plants. American Society of Plant Physiologists, Rockville, MD, pp 1204–1249
Lasat MM, Baker AJM, Kochian LV (1996) Physiological characterization of root Zn2+ absorption and translocation to shoots in Zn hyperaccumulator and nonaccumulator species of Thlaspi. Plant Physiol 112:1715–1722
Lasat MM, Baker AJM, Kochian LV (1998) Altered Zn compartmentation in the root symplasm and stimulated Zn absorption into the leaf as mechanisms involved in Zn hyperaccumulation in Thlaspi caerulescens. Plant Physiol 118:875–883
Liu Z, Shen J, Carbrey JM, Mukhopadhyay R, Agre P, Rosen BP (2002) Arsenite transport by mammalian aquaglyceroporins AQP7 and AQP9. Proc Natl Acad Sci USA 99:6053–6058
Ma LQ, Komar KM, Tu C, Zhang W, Cai Y (2001) A fern that hyperaccumulates arsenic. Nature 409:579
Meharg AA (2003) Variation in arsenic accumulation—hyperaccumulation in ferns and their allies. New Phytol 157:25–31
Meharg AA, Jardine L (2003) Arsenite transport into paddy rice (Oryza sativa) roots. New Phytol 157:39–44
Meharg AA, Macnair MR (1990) An altered phosphate uptake system in arsenate tolerant Holcus lanatus L. New Phytol 116:431–441
Meharg AA, Macnair MR (1992) Suppression of the high affinity phosphate uptake system: a mechanism of arsenate tolerance in Holcus lanatus L. J Exp Bot 43:519–524
Meharg AA, Naylor J, Macnair MR (1994) Phosphorus nutrition of arsenate-tolerant and nontolerant phenotypes of velvetgrass. J Environ Qual 23:234–238
National Research Council (1977) Committee of medical biological effects of environmental pollutants: arsenic. National Academy of Sciences, Washington DC
National Research Council (1999) Arsenic in drinking water. National Academy Press, Washington DC
Pickering IJ, Prince RC, George MJ, Smith RD, George GN, Salt DE (2000) Reduction and coordination of arsenic in Indian mustard. Plant Physiol 122:1171–1178
Roy SJ, Cuin TA, Leigh RA (2003) Nanolitre-scale assays to determine the activities of enzymes in individual plant cells. Plant J 34:555–564
Segel, IH (1976) Biochemical calculations, 2nd edn. Wiley, New York
Tu C, Ma LQ, Bondada B (2002) Arsenic accumulation in the hyperaccumulator Chinese Brake and its utilization potential for phytoremedication. J Environ Qual 31:1671–1675
Wang J, Zhao F-J, Meharg AA, Raab A, Feldmann J, McGrath SP (2002) Mechanisms of arsenic hyperaccumulation in Pteris vittata. Uptake kinetics, interactions with phosphate, and arsenic speciation. Plant Physiol 130:1552–1561
Wysocki R, Chery CC, Wawrzycka D, Van Hulle M, Cornelis R, Thevelein JM, Tamas MJ (2001) The glycerol channel Fps1p mediates the uptake of arsenite and antimonite in Saccharomyces cerevisiae. Mol Microbiol 40:1391–1401
Zhao F-J, Dunham SJ, McGrath SP (2002) Arsenic hyperaccumulation by different fern species. New Phytol 156:27–31
Zhao F-J, Wang J, Barker JHA, Schat H, Bleeker PM, McGrath SP (2003) The role of phytochelatins in arsenic tolerance in the hyperaccumulator Pteris vittata. New Phytol 159:403–410
Acknowledgements
The authors thank Drs. Jon Hart and Stuart Roy for helpful discussions on analysis of kinetic data and Jon Shaff for excellent technical assistance. The project described was supported by grant number 5 R44 ES011065-03 from the National Institute of Environmental Health Sciences (NIEHS), NIH, USA. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIEHS, NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Poynton, C.Y., Huang, J.W., Blaylock, M.J. et al. Mechanisms of arsenic hyperaccumulation in Pteris species: root As influx and translocation. Planta 219, 1080–1088 (2004). https://doi.org/10.1007/s00425-004-1304-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-004-1304-8