Abstract
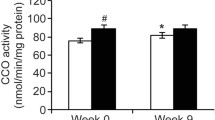
The purpose of this study was to assess the physiological adaptations in physically fit individuals to a period of intensified training. Ten trained males cycled outdoors ~170 km day−1 on 19 out of 21 days. Expired gas was collected on days 1 and 21 during maximal graded exercise and used for the determination of gross efficiency and whole body substrate use. Muscle biopsies were obtained before and after exercise on days 2 and 22 for the determination of mtDNA/gDNA ratio, gene expression, metabolic enzyme activity and glycogen use. Muscle glycogen before and after exercise, fat oxidation, and gross efficiency increased, carbohydrate oxidation decreased (p < 0.05), and VO2max did not change over the 21 days of training. Citrate synthase (CS), β-hydroxyacyl CoA dehydrogenase (β-HAD) and cytochrome c oxidase (COX) enzyme activity did not change with training. CS and β-HAD mRNA did not change with acute exercise or training. COX (subunit IV) mRNA increased with acute exercise (p < 0.05) but did not change over the 21 days. PGC-1α mRNA increased with acute exercise, but did not increase to the same degree on day 22 as it did on day 2 (p < 0.05). UCP3 mRNA decreased with training (p < 0.05). Acute exercise caused an increase in mitofusin2 (MFN2) mRNA (p < 0.05) and a trend for an increase in mtDNA/gDNA ratio (p = 0.057). However, training did not affect MFN2 mRNA or mtDNA/gDNA ratio. In response to 3,211 km of cycling, changes in substrate use and gross efficiency appear to be more profound than mitochondrial adaptations in trained individuals.



Similar content being viewed by others
References
Bach D, Pich S, Soriano FX et al (2003) Mitofusin-2 determines mitochondrial network architecture and mitochondrial metabolism. A novel regulatory mechanism altered in obesity. J Biol Chem 278:17190–17197. doi:10.1074/jbc.M212754200
Bezaire V, Seifert EL, Harper ME (2007) Uncoupling protein-3: clues in an ongoing mitochondrial mystery. FASEB J 21:312–324. doi:10.1096/fj.06-6966rev
Cadefau J, Green HJ, Cusso R, Ball-Burnett M, Jamieson G (1994) Coupling of muscle phosphorylation potential to glycolysis during work after short-term training. J Appl Physiol 76:2586–2593
Cartoni R, Leger B, Hock MB et al (2005) Mitofusins 1/2 and ERRalpha expression are increased in human skeletal muscle after physical exercise. J Physiol 567:349–358. doi:10.1113/jphysiol.2005.092031
Chesley A, Heigenhauser GJ, Spriet LL (1996) Regulation of muscle glycogen phosphorylase activity following short-term endurance training. Am J Physiol 270:E328–E335
Claypool SM (2009) Cardiolipin, a critical determinant of mitochondrial carrier protein assembly and function. Biochim Biophys Acta 1788:2059–2068. doi:10.1016/j.bbamem.2009.04.020
Duhamel TA, Stewart RD, Tupling AR, Ouyang J, Green HJ (2007) Muscle sarcoplasmic reticulum calcium regulation in humans during consecutive days of exercise and recovery. J Appl Physiol 103:1212–1220. doi:10.1152/japplphysiol.00437.2007
Dumke CL, Mark Davis J, Angela Murphy E et al (2009) Successive bouts of cycling stimulates genes associated with mitochondrial biogenesis. Eur J Appl Physiol 107:419–427. doi:10.1007/s00421-009-1143-1
Green HJ, Bombardier E, Duhamel TA, Stewart RD, Tupling AR, Ouyang J (2008a) Metabolic, enzymatic, and transporter responses in human muscle during three consecutive days of exercise and recovery. Am J Physiol Regul Integr Comp Physiol 295:R1238–R1250. doi:10.1152/ajpregu.00171.2008
Green HJ, Duhamel TA, Stewart RD, Tupling AR, Ouyang J (2008b) Dissociation between changes in muscle Na+ –K+ -ATPase isoform abundance and activity with consecutive days of exercise and recovery. Am J Physiol Endocrinol Metab 294:E761–E767. doi:10.1152/ajpendo.00751.2007
Henderson GC, Fattor JA, Horning MA et al (2007) Lipolysis and fatty acid metabolism in men and women during the postexercise recovery period. J Physiol 584:963–981. doi:10.1113/jphysiol.2007.137331
Hermansen L, Hultman E, Saltin B (1967) Muscle glycogen during prolonged severe exercise. Acta Physiol Scand 71:129–139
Himms-Hagen J, Harper ME (2001) Physiological role of UCP3 may be export of fatty acids from mitochondria when fatty acid oxidation predominates: an hypothesis. Exp Biol Med (Maywood) 226:78–84
Holloszy JO (1967) Biochemical adaptations in muscle. Effects of exercise on mitochondrial oxygen uptake and respiratory enzyme activity in skeletal muscle. J Biol Chem 242:2278–2282
Holloszy JO (1975) Adaptation of skeletal muscle to endurance exercise. Med Sci Sports 7:155–164
Hurley BF, Hagberg JM, Allen WK et al (1984) Effect of training on blood lactate levels during submaximal exercise. J Appl Physiol 56:1260–1264
Hurley BF, Nemeth PM, Martin WH 3rd, Hagberg JM, Dalsky GP, Holloszy JO (1986) Muscle triglyceride utilization during exercise: effect of training. J Appl Physiol 60:562–567
Jemiolo B, Trappe S (2004) Single muscle fiber gene expression in human skeletal muscle: validation of internal control with exercise. Biochem Biophys Res Commun 320:1043–1050. doi:10.1016/j.bbrc.2004.05.223
Jeukendrup AE, Wallis GA (2005) Measurement of substrate oxidation during exercise by means of gas exchange measurements. Int J Sports Med 26(Suppl 1):S28–S37. doi:10.1055/s-2004-830512
Lawn RM, Efstratiadis A, O’Connell C, Maniatis T (1980) The nucleotide sequence of the human beta-globin gene. Cell 21:647–651
Lin J, Wu PH, Tarr PT et al (2004) Defects in adaptive energy metabolism with CNS-linked hyperactivity in PGC-1alpha null mice. Cell 119:121–135. doi:10.1016/j.cell.2004.09.013
Liu X, Weaver D, Shirihai O, Hajnoczky G (2009) Mitochondrial ‘kiss-and-run’: interplay between mitochondrial motility and fusion–fission dynamics. EMBO J 28:3074–3089. doi:10.1038/emboj.2009.255
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2[-Delta Delta C(T)] Method. Methods 25:402–408. doi:10.1006/meth.2001.1262
Lucia A, Earnest C, Arribas C (2003) The tour de France: a physiological review. Scand J Med Sci Sports 13:275–283
Marcuello A, Gonzalez-Alonso J, Calbet JA, Damsgaard R, Lopez-Perez MJ, Diez-Sanchez C (2005) Skeletal muscle mitochondrial DNA content in exercising humans. J Appl Physiol 99:1372–1377. doi:10.1152/japplphysiol.00289.2005
Menshikova EV, Ritov VB, Toledo FG, Ferrell RE, Goodpaster BH, Kelley DE (2005) Effects of weight loss and physical activity on skeletal muscle mitochondrial function in obesity. Am J Physiol Endocrinol Metab 288:E818–E825. doi:10.1152/ajpendo.00322.2004
Menshikova EV, Ritov VB, Ferrell RE, Azuma K, Goodpaster BH, Kelley DE (2007) Characteristics of skeletal muscle mitochondrial biogenesis induced by moderate-intensity exercise and weight loss in obesity. J Appl Physiol 103:21–27. doi:10.1152/japplphysiol.01228.2006
Miller FJ, Rosenfeldt FL, Zhang C, Linnane AW, Nagley P (2003) Precise determination of mitochondrial DNA copy number in human skeletal and cardiac muscle by a PCR-based assay: lack of change of copy number with age. Nucleic Acids Res 31:e61
Moseley L, Jeukendrup AE (2001) The reliability of cycling efficiency. Med Sci Sports Exerc 33:621–627
Ono T, Isobe K, Nakada K, Hayashi JI (2001) Human cells are protected from mitochondrial dysfunction by complementation of DNA products in fused mitochondria. Nat Genet 28:272–275. doi:10.1038/90116
Phillips SM, Green HJ, Tarnopolsky MA, Heigenhauser GJ, Grant SM (1996) Progressive effect of endurance training on metabolic adaptations in working skeletal muscle. Am J Physiol 270:E265–E272
Pilegaard H, Saltin B, Neufer PD (2003) Exercise induces transient transcriptional activation of the PGC-1alpha gene in human skeletal muscle. J Physiol 546:851–858 PHY_034850[pii]
Schmutz S, Dapp C, Wittwer M, Vogt M, Hoppeler H, Fluck M (2006) Endurance training modulates the muscular transcriptome response to acute exercise. Pflugers Arch 451:678–687. doi:10.1007/s00424-005-1497-0
Schrauwen P, Hesselink MK, Vaartjes I et al (2002) Effect of acute exercise on uncoupling protein 3 is a fat metabolism-mediated effect. Am J Physiol Endocrinol Metab 282:E11–E17
Schrauwen P, Russell AP, Moonen-Kornips E, Boon N, Hesselink MK (2005) Effect of 2 weeks of endurance training on uncoupling protein 3 content in untrained human subjects. Acta Physiol Scand 183:273–280. doi:10.1111/j.1365-201X.2004.01393.x
Siri WE (1993) Body composition from fluid spaces and density: analysis of methods. 1961. Nutrition 9:480–491 discussion 480, 492
Szuhai K, Ouweland J, Dirks R et al (2001) Simultaneous A8344G heteroplasmy and mitochondrial DNA copy number quantification in myoclonus epilepsy and ragged-red fibers (MERRF) syndrome by a multiplex molecular beacon based real-time fluorescence PCR. Nucleic Acids Res 29:E13
Tunstall RJ, Mehan KA, Wadley GD et al (2002) Exercise training increases lipid metabolism gene expression in human skeletal muscle. Am J Physiol Endocrinol Metab 283:E66–E72. doi:10.1152/ajpendo.00475.2001
Wang S, Subramaniam A, Cawthorne MA, Clapham JC (2003) Increased fatty acid oxidation in transgenic mice overexpressing UCP3 in skeletal muscle. Diabetes Obes Metab 5:295–301 273[pii]
Wende AR, Schaeffer PJ, Parker GJ et al (2007) A role for the transcriptional coactivator PGC-1alpha in muscle refueling. J Biol Chem 282:36642–36651. doi:10.1074/jbc.M707006200
Wojtaszewski JF, MacDonald C, Nielsen JN et al (2003) Regulation of 5′AMP-activated protein kinase activity and substrate utilization in exercising human skeletal muscle. Am J Physiol Endocrinol Metab 284:E813–E822. doi:10.1152/ajpendo.00436.2002
Yeo WK, Paton CD, Garnham AP, Burke LM, Carey AL, Hawley JA (2008) Skeletal muscle adaptation and performance responses to once a day versus twice every second day endurance training regimens. J Appl Physiol 105:1462–1470. doi:10.1152/japplphysiol.90882.2008
Acknowledgments
The views and conclusions contained herein are those of the authors and should not be interpreted as necessarily representing the official policies or endorsements, either expressed or implied, of Air Force Research laboratory or the U.S. government. Sponsored by Air Force Research Laboratory under agreement number FA5650-06-2-6740 and the University of Montana Grant program. We would like to acknowledge the assistance of the Molecular Biology Core Facility of The University of Montana supported in part by National Center for Research Resources Grants P20RR017670 and P20RR015583.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Susan A. Ward.
Rights and permissions
About this article
Cite this article
Slivka, D.R., Dumke, C.L., Hailes, W.S. et al. Substrate use and biochemical response to a 3,211-km bicycle tour in trained cyclists. Eur J Appl Physiol 112, 1621–1630 (2012). https://doi.org/10.1007/s00421-011-2129-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-011-2129-3