Abstract
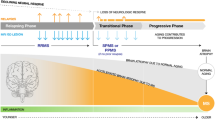
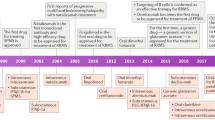
Increasing options are dictating the development of new algorithms to provide guidance in the treatment of people with multiple sclerosis (MS). There is a wealth of evidence on the safety and efficacy of interferon-beta and glatiramer acetate, which have been used in Europe and in the United States for more than 10 years. The spectrum of approved indications for these conventional disease modifying therapies includes the treatment of relapsing-remitting MS, secondary progressive MS, and the clinically isolated syndrome. Beyond these therapies we already have the recently introduced antibody natalizumab and, in some countries, the immunosuppressive agent mitoxantrone. Oral therapies are expected in the near future, with the sphingosin-1-phosphate receptor modulator fingolimod approved in the US and the EU and the purine nucleoside analogue cladribine in Australia and Russia. The evidence on all of these conventional and novel therapeutics is reviewed in this paper to provide an overview of the changing landscape of MS treatment.


Similar content being viewed by others
Notes
Evidence for Interferon Dose–Response: European-North American Comparative Efficacy.
Independent comparison of interferon.
International MS secondary Progressive Avonex Controlled Trial.
Betaferon efficacy yielding outcomes of a new dose.
Betaseron versus Copaxone in MS with triple-dose gadolinium and 3T MRI endpoints.
Rebif versus Glatiramer acetate in relapsing MS disease.
Controlled High-Risk Subjects Avonex Multiple Sclerosis Prevention Study.
Early treatment of Multiple Sclerosis.
Betaferon in newly emerging Multiple Sclerosis for initial treatment.
Early glatiramer acetate treatment in delaying conversion to clinically definite Multiple Sclerosis in subjects presenting with a clinically isolated syndrome.
Mitoxantrone in Multiple Sclerosis.
Fertility and Mitoxantrone in MS.
Natalizumab safety and efficacy in relapsing remitting Multiple Sclerosis.
Safety and efficacy of natalizumab in combination with Avonex.
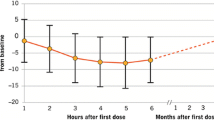
FTY720 research evaluating effects of daily oral therapy in Multiple Sclerosis.
Trial assessing injectable interferon versus FTY720 oral in RRMS.
Cladribine tablets treating MS orally.
References
Lublin FD, Reingold SC (1996) Defining the clinical course of multiple sclerosis: results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis. Neurology 46:907–911
Lassmann H, Bruck W, Lucchinetti CF (2007) The immunopathology of multiple sclerosis: an overview. Brain Pathol 17:210–218
Kuhlmann T, Lingfeld G, Bitsch A et al (2002) Acute axonal damage in multiple sclerosis is most extensive in early disease stages and decreases over time. Brain 125:2202–2212
De Stefano N, Narayanan S, Francis GS et al (2001) Evidence of axonal damage in the early stages of multiple sclerosis and its relevance to disability. Arch Neurol 58:65–70
Weinshenker BG, Bass B, Rice GP et al (1989) The natural history of multiple sclerosis: a geographically based study. 2. Predictive value of the early clinical course. Brain 112(Pt 6):1419–1428
Ebers GC (2001) Natural History of multiple sclerosis. J Neurol Neurosurg Psychiatry 71:ii16–ii19
Tintore M, Rovira A, Rio J et al (2006) Baseline MRI predicts future attacks and disability in clinically isolated syndromes. Neurology 67:968–972
Fisniku LK, Brex PA, Altmann DR et al (2008) Disability and T2 MRI lesions: a 20-year follow-up of patients with relapse onset of multiple sclerosis. Brain 131:808–817
Langer-Gould A, Popat RA, Huang SM et al (2006) Clinical and demographic predictors of long-term disability in patients with relapsing-remitting multiple sclerosis: a systematic review. Arch Neurol 63:1686–1691
McDonald WI, Compston A, Edan G et al (2001) Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol 50:121–127
Polman CH, Reingold SC, Edan G et al (2005) Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol 58:840–846
Dhib-Jalbut S, Marks S (2010) Interferon-beta mechanisms of action in multiple sclerosis. Neurology 74(Suppl 1):S17–S24
Racke MK, Lovett-Racke AE, Karandikar NJ (2010) The mechanism of action of glatiramer acetate treatment in multiple sclerosis. Neurology 74(Suppl 1):S25–S30
Linker RA, Lee D-H, Demir S et al (2010) Functional role of brain-derived neurotrophic factor in neuroprotective autoimmunity: therapeutic implications in a model of multiple sclerosis. Brain 133:2248–2263
Paty DW, Li DK (1993) Interferon beta-1b is effective in relapsing-remitting multiple sclerosis. II. MRI analysis results of a multicenter, randomized, double-blind, placebo-controlled trial. UBC MS/MRI Study Group and the IFNB Multiple Sclerosis Study Group. Neurology 43:662–667
The IFNB Multiple Sclerosis Study Group (1993) Interferon beta-1b is effective in relapsing-remitting multiple sclerosis. I. Clinical results of a multicenter, randomized, double-blind, placebo-controlled trial. The IFNB Multiple Sclerosis Study Group. Neurology 43:655–661
The IFNB Multiple Sclerosis Study Group (1995) Interferon beta-1b in the treatment of multiple sclerosis: final outcome of the randomized controlled trial. The IFNB Multiple Sclerosis Study Group and The University of British Columbia MS/MRI Analysis Group. Neurology 45:1277–1285
Goodin DS, Frohman EM, Garmany GP Jr et al (2002) Disease modifying therapies in multiple sclerosis: report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the MS Council for Clinical Practice Guidelines. Neurology 58:169–178
Wiendl H, Toyka KV, Rieckmann P et al (2008) Basic and escalating immunomodulatory treatments in multiple sclerosis: current therapeutic recommendations. J Neurol 255:1449–1463
Kappos L, European Study Group on Interferon beta-1b in Secondary progressive MS (1998) Placebo-controlled multicentre randomised trial of interferon beta-1b in treatment of secondary progressive multiple sclerosis. European Study Group on interferon beta-1b in secondary progressive MS. Lancet 352:1491–1497
Panitch H, Miller A, Paty D et al (2004) Interferon beta-1b in secondary progressive MS: results from a 3-year controlled study. Neurology 63:1788–1795
Jacobs LD, Cookfair DL, Rudick RA et al (1996) Intramuscular interferon beta-1a for disease progression in relapsing multiple sclerosis. The Multiple Sclerosis Collaborative Research Group (MSCRG). Ann Neurol 39:285–294
PRISMS Study Group (1998) Randomised double-blind placebo-controlled study of interferon beta-1a in relapsing/remitting multiple sclerosis. PRISMS (Prevention of Relapses and Disability by Interferon beta-1a Subcutaneously in Multiple Sclerosis) Study Group. Lancet 352:1498–1504
Li DK, Paty DW (1999) Magnetic resonance imaging results of the PRISMS trial: a randomized, double-blind, placebo-controlled study of interferon-beta1a in relapsing-remitting multiple sclerosis. Prevention of relapses and disability by interferon-beta1a subcutaneously in multiple sclerosis. Ann Neurol 46:197–206
Johnson KP, Brooks BR, Cohen JA et al (1995) Copolymer 1 reduces relapse rate and improves disability in relapsing-remitting multiple sclerosis: results of a phase III multicenter, double-blind placebo-controlled trial. The Copolymer 1 Multiple Sclerosis Study Group. Neurology 45:1268–1276
Comi G, Filippi M, Wolinsky JS (2001) European/Canadian multicenter, double-blind, randomized, placebo-controlled study of the effects of glatiramer acetate on magnetic resonance imaging–measured disease activity and burden in patients with relapsing multiple sclerosis. European/Canadian Glatiramer Acetate Study Group. Ann Neurol 49:290–297
Cohen JA, Cutter GR, Fischer JS et al (2002) Benefit of interferon beta-1a on MSFC progression in secondary progressive MS. Neurology 59:679–687
SPECTRIMS Study Group (2001) Randomized controlled trial of interferon-beta-1a in secondary progressive MS: clinical results. Neurology 56:1496–1504
Li DKB, Zhao GJ, Paty DW (2001) Randomized controlled trial of interferon-beta-1a in secondary progressive MS: MRI results. Neurology 56:1505–1513
Panitch H, Goodin DS, Francis G et al (2002) Randomized, comparative study of interferon beta-1a treatment regimens in MS: the EVIDENCE Trial. Neurology 59:1496–1506
Durelli L, Verdun E, Barbero P et al (2002) Every-other-day interferon beta-1b versus once-weekly interferon beta-1a for multiple sclerosis: results of a 2-year prospective randomised multicentre study (INCOMIN). Lancet 359:1453–1460
O’Connor P, Filippi M, Arnason B et al (2009) 250 microg or 500 microg interferon beta-1b versus 20 mg glatiramer acetate in relapsing-remitting multiple sclerosis: a prospective, randomised, multicentre study. Lancet Neurol 8:889–897
Cadavid D, Wolansky LJ, Skurnick J et al (2009) Efficacy of treatment of MS with IFNbeta-1b or glatiramer acetate by monthly brain MRI in the BECOME study. Neurology 72:1976–1983
Mikol DD, Barkhof F, Chang P et al (2008) Comparison of subcutaneous interferon beta-1a with glatiramer acetate in patients with relapsing multiple sclerosis (the REbif vs Glatiramer Acetate in Relapsing MS Disease [REGARD] study): a multicentre, randomised, parallel, open-label trial. Lancet Neurol 7:903–914
Reder AT, Ebers G, Cutter G, et al. (2010) Survival analysis 21 years after the initiation of the pivotal Interferon Beta-1b trial in patients with RRMS. European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), Gothenburg, Sweden:P 903
Jacobs LD, Beck RW, Simon JH et al (2000) Intramuscular interferon beta-1a therapy initiated during a first demyelinating event in multiple sclerosis. CHAMPS Study Group. N Engl J Med 343:898–904
Comi G, Filippi M, Barkhof F et al (2001) Effect of early interferon treatment on conversion to definite multiple sclerosis: a randomised study. Lancet 357:1576–1582
Kappos L, Polman CH, Freedman MS et al (2006) Treatment with interferon beta-1b delays conversion to clinically definite and McDonald MS in patients with clinically isolated syndromes. Neurology 67:1242–1249
Kappos L, Freedman MS, Polman CH et al (2007) Effect of early versus delayed interferon beta-1b treatment on disability after a first clinical event suggestive of multiple sclerosis: a 3-year follow-up analysis of the BENEFIT study. Lancet 370:389–397
Comi G, Martinelli V, Rodegher M et al (2009) Effect of glatiramer acetate on conversion to clinically definite multiple sclerosis in patients with clinically isolated syndrome (PreCISe study): a randomised, double-blind, placebo-controlled trial. Lancet 374:1503–1511
Kinkel RP, Kollman C, O’Connor P et al (2006) IM interferon beta-1a delays definite multiple sclerosis 5 years after a first demyelinating event. Neurology 66:678–684
Kinkel RP, Tanner JP, Simon J, et al. (2009) CHAMPIONS extension study: 10 year follow-up after a clinically isolated syndrome (CIS) in high risk patients. Neurology 72
Kappos L, Freedman MS, Polman CH et al (2009) Long-term effect of early treatment with interferon beta-1b after a first clinical event suggestive of multiple sclerosis: 5-year active treatment extension of the phase 3 BENEFIT trial. Lancet Neurol 8:987–997
Rojas JI, Romano M, Ciapponi A, et al. (2010) Interferon Beta for primary progressive multiple sclerosis. Cochrane Database Syst Rev:CD006643
Wolinsky JS, Narayana PA, O’Connor P et al (2007) Glatiramer acetate in primary progressive multiple sclerosis: Results of a multinational, multicenter, double-blind, placebo-controlled trial. Ann Neurol 61:14–24
Hawker K, O’Connor P, Freedman MS et al (2009) Rituximab in patients with primary progressive multiple sclerosis: results of a randomized double-blind placebo-controlled multicenter trial. Ann Neurol 66:460–471
Fidler JM, DeJoy SQ, Gibbons JJ Jr (1986) Selective immunomodulation by the antineoplastic agent mitoxantrone. I. Suppression of B lymphocyte function. J Immunol 137:727–732
Fidler JM, DeJoy SQ, Smith FR 3rd et al (1986) Selective immunomodulation by the antineoplastic agent mitoxantrone. II. Nonspecific adherent suppressor cells derived from mitoxantrone-treated mice. J Immunol 136:2747–2754
Chan A, Weilbach FX, Toyka KV et al (2005) Mitoxantrone induces cell death in peripheral blood leucocytes of multiple sclerosis patients. Clin Exp Immunol 139:152–158
Neuhaus O, Wiendl H, Kieseier BC et al (2005) Multiple sclerosis: mitoxantrone promotes differential effects on immunocompetent cells in vitro. J Neuroimmunol 168:128–137
Millefiorini E, Gasperini C, Pozzilli C et al (1997) Randomized placebo-controlled trial of mitoxantrone in relapsing-remitting multiple sclerosis: 24-month clinical and MRI outcome. J Neurol 244:153–159
van de Wyngaert FA, Beguin C, D’Hooghe MB et al (2001) A double-blind clinical trial of mitoxantrone versus methylprednisolone in relapsing, secondary progressive multiple sclerosis. Acta Neurol Belg 101:210–216
Edan G, Miller D, Clanet M et al (1997) Therapeutic effect of mitoxantrone combined with methylprednisolone in multiple sclerosis: a randomised multicentre study of active disease using MRI and clinical criteria. J Neurol Neurosurg Psychiatry 62:112–118
Hartung HP, Gonsette R, Konig N et al (2002) Mitoxantrone in progressive multiple sclerosis: a placebo-controlled, double-blind, randomised, multicentre trial. Lancet 360:2018–2025
Marriott JJ, Miyasaki JM, Gronseth G et al (2010) Evidence report: the efficacy and safety of mitoxantrone (Novantrone) in the treatment of multiple sclerosis: Report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Neurology 74:1463–1470
Kingwell E, Koch M, Leung B et al (2010) Cardiotoxicity and other adverse events associated with mitoxantrone treatment for MS. Neurology 74:1822–1826
Novantrone Prescribing Information (2009). http://www.novantrone.com/assets/pdf/novantrone_prescribing_info.pdf. Accessed 20 January 2011
Mistry AR, Felix CA, Whitmarsh RJ et al (2005) DNA topoisomerase II in therapy-related acute promyelocytic leukemia. N Engl J Med 352:1529–1538
Cocco E, Sardu C, Gallo P et al (2008) Frequency and risk factors of mitoxantrone-induced amenorrhea in multiple sclerosis: the FEMIMS study. Mult Scler 14:1225–1233
Cotte S, von Ahsen N, Kruse N et al (2009) ABC-transporter gene-polymorphisms are potential pharmacogenetic markers for mitoxantrone response in multiple sclerosis. Brain 132:2517–2530
Rieckmann P (2009) Concepts of induction and escalation therapy in multiple sclerosis. J Neurol Sci 277(Suppl 1):S42–S45
Le Page E, Comi G, Filippi M et al (2008) Comparison of two therapeutic strategies in aggressive relapsing-remitting MS: mitoxantrone as induction for 6 months followed by interferon-b-1b versus interferon-b-1b. A 3-Year Randomized Trial. American Academy of Neurology, 60th Annual Meeting, Chicago:S22.004
Arnold DL, Campagnolo D, Panitch H et al (2008) Glatiramer acetate after mitoxantrone induction improves MRI markers of lesion volume and permanent tissue injury in MS. J Neurol 255:1473–1478
Cocco E, Marchi P, Sardu C et al (2007) Mitoxantrone treatment in patients with early relapsing-remitting multiple sclerosis. Mult Scler 13:975–980
Le Page E, Leray E, Taurin G et al (2008) Mitoxantrone as induction treatment in aggressive relapsing remitting multiple sclerosis: treatment response factors in a 5 year follow-up observational study of 100 consecutive patients. J Neurol Neurosurg Psychiatry 79:52–56
Ramtahal J, Jacob A, Das K et al (2006) Sequential maintenance treatment with glatiramer acetate after mitoxantrone is safe and can limit exposure to immunosuppression in very active, relapsing remitting multiple sclerosis. J Neurol 253:1160–1164
Zaffaroni M, Rizzo A, Baldini SM et al (2008) Induction and add-on therapy with mitoxantrone and interferon beta in multiple sclerosis. Neurol Sci 29(Suppl 2):S230–S232
Kita M, Cohen JA, Fox RJ et al (2004) A phase II trial of mitoxantrone in patients with primary progressive multiple sclerosis. Neurology 62:A99
Rudick RA, Sandrock A (2004) Natalizumab: alpha 4-integrin antagonist selective adhesion molecule inhibitors for MS. Expert Rev Neurother 4:571–580
Kent SJ, Karlik SJ, Cannon C et al (1995) A monoclonal antibody to alpha 4 integrin suppresses and reverses active experimental allergic encephalomyelitis. J Neuroimmunol 58:1–10
Polman CH, O’Connor PW, Havrdova E et al (2006) A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N Engl J Med 354:899–910
Hutchinson M, Kappos L, Calabresi PA et al (2009) The efficacy of natalizumab in patients with relapsing multiple sclerosis: subgroup analyses of AFFIRM and SENTINEL. J Neurol 256:405–415
Rudick RA, Stuart WH, Calabresi PA et al (2006) Natalizumab plus interferon beta-1a for relapsing multiple sclerosis. N Engl J Med 354:911–923
O’Connor P, Polman C, Goodman A et al (2009) Efficacy and Safety of Natalizumab in the STRATA Study. In: American Academy of Neurology. Seattle, P06.127
Polman C, Goodman A, Kappos L et al (2010) Efficacy and Safety of Natalizumab in the STRATA Study. American Academy of Neurology, Toronto, ON, Canada, P06.173
Havrdova E, Galetta S, Hutchinson M et al (2009) Effect of natalizumab on clinical and radiological disease activity in multiple sclerosis: a retrospective analysis of the Natalizumab Safety and Efficacy in Relapsing-Remitting Multiple Sclerosis (AFFIRM) study. Lancet Neurol 8:254–260
Munschauer F, Giovannoni G, Lublin F et al (2009) Sustained improvement in physical disability with Natalizumab in patients with relapsing multiple sclerosis. American Academy of Neurology, Seattle, P06.131
Langer-Gould A, Atlas SW, Green AJ et al (2005) Progressive multifocal leukoencephalopathy in a patient treated with natalizumab. N Engl J Med 353:375–381
Kleinschmidt-DeMasters BK, Tyler KL (2005) Progressive multifocal leukoencephalopathy complicating treatment with natalizumab and interferon beta-1a for multiple sclerosis. N Engl J Med 353:369–374
Wenning W, Haghikia A, Laubenberger J et al (2009) Treatment of progressive multifocal leukoencephalopathy associated with natalizumab. N Engl J Med 361:1075–1080
(2011) UPDATE on Tysabri and PML. Sponsor and FDA provide information on cases and risks. http://www.nationalmssociety.org/news/news-detail/index.aspx?nid=2308. Accessed 04 March 2011
Chen Y, Bord E, Tompkins T et al (2009) Asymptomatic reactivation of JC virus in patients treated with natalizumab. N Engl J Med 361:1067–1074
Jilek S, Jaquiery E, Hirsch HH et al (2010) Immune responses to JC virus in patients with multiple sclerosis treated with natalizumab: a cross-sectional and longitudinal study. Lancet Neurol 9:264–272
Warnke C, Smolianov V, Dehmel T et al (2010) CD34+ progenitor cells mobilized by natalizumab are not a relevant reservoir for JC virus. Mult Scler 17(2):151–156
del Pilar Martin M, Cravens PD, Winger R et al (2008) Decrease in the numbers of dendritic cells and CD4+ T cells in cerebral perivascular spaces due to natalizumab. Arch Neurol 65:1596–1603
Clifford DB, De Luca A, Simpson DM et al (2010) Natalizumab-associated progressive multifocal leukoencephalopathy in patients with multiple sclerosis: lessons from 28 cases. Lancet Neurol 9:438–446
Tan CS, Koralnik IJ (2010) Progressive multifocal leukoencephalopathy and other disorders caused by JC virus: clinical features and pathogenesis. Lancet Neurol 9:425–437
Boster A, Hreha S, Berger JR et al (2009) Progressive multifocal leukoencephalopathy and relapsing-remitting multiple sclerosis: a comparative study. Arch Neurol 66:593–599
Bozic C, Cristiano LM, Hyde R et al (2010) Utilisation and safety of natalizumab in patients with relapsing multiple sclerosis. European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), Gothenburg, Sweden, p 893
Sunyaev SR, Lugovskoy A, Simon K et al (2009) Adaptive mutations in the JC virus protein capsid are associated with progressive multifocal leukoencephalopathy (PML). PLoS Genet 5:e1000368
Tan CS, Chen Y, Viscidi RP et al (2010) Discrepant findings in immune responses to JC virus in patients receiving natalizumab. Lancet Neurol 9:565-566; author reply 566-567
Sadiq SA, Puccio LM, Brydon EW (2010) JCV detection in multiple sclerosis patients treated with natalizumab. J Neurol 257:954–958
Haghikia A, Pappas D, Pula B et al (2009) Assessment of a possible bioenergetic marker of cellular immunocompetence in MS-patients undergoing immunotherapy: longitudinal analyses. Neurology 72 (Suppl3): P09.102
Gorelik L, Lerner M, Bixler S et al (2010) Anti-JC virus antibodies: implications for PML risk stratification. Ann Neurol 68:295–303
Kerbrat Lecuyer A, Le Page E, Leray E et al (2010) Assessment of disease activity within 6 months after natalizumab discontinuation: an observational study of 28 consecutive relapsing–remitting multiple sclerosis patients. European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), Gothenburg, Sweden:P394
Khatri BO, Man S, Giovannoni G et al (2009) Effect of plasma exchange in accelerating natalizumab clearance and restoring leukocyte function. Neurology 72:402–409
Linda H, von Heijne A, Major EO et al (2009) Progressive multifocal leukoencephalopathy after natalizumab monotherapy. N Engl J Med 361:1081–1087
Elphick GF, Querbes W, Jordan JA et al (2004) The human polyomavirus, JCV, uses serotonin receptors to infect cells. Science 306:1380–1383
Brickelmaier M, Lugovskoy A, Kartikeyan R et al (2009) Identification and characterization of mefloquine efficacy against JC virus in vitro. Antimicrob Agents Chemother 53:1840–1849
Kappos L, Bates D, Hartung HP et al (2007) Natalizumab treatment for multiple sclerosis: recommendations for patient selection and monitoring. Lancet Neurol 6:431–441
Vermersch P, Foley J, Gold R et al (2010) Overview of clinical outcomes in cases of natalizumab-associated progressive multifocal leukoencephalopathy. European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), Gothenburg, Sweden, p 112
Tremlett HL, Oger J (2003) Interrupted therapy: stopping and switching of the beta-interferons prescribed for MS. Neurology 61:551–554
Devonshire V, Lapierre Y, Macdonell R et al (2011) The Global Adherence Project (GAP): a multicenter observational study on adherence to disease-modifying therapies in patients with relapsing-remitting multiple sclerosis. Eur J Neurol 18:69–77
Patti F (2010) Optimizing the benefit of multiple sclerosis therapy: the importance of treatment adherence. Patient Prefer Adherence 4:1–9
Fujita T, Inoue K, Yamamoto S et al (1994) Fungal metabolites. Part 11. A potent immunosuppressive activity found in Isaria sinclairii metabolite. J Antibiot Tokyo 47:208–215
Brinkmann V (2009) FTY720 (fingolimod) in Multiple Sclerosis: therapeutic effects in the immune and the central nervous system. Br J Pharmacol 158:1173–1182
Kappos L, Antel J, Comi G et al (2006) Oral fingolimod (FTY720) for relapsing multiple sclerosis. N Engl J Med 355:1124–1140
O’Connor P, Comi G, Montalban X et al (2009) Oral fingolimod (FTY720) in multiple sclerosis: two-year results of a phase II extension study. Neurology 72:73–79
Comi G, O’Connor P, Montalban X et al (2010) Phase II study of oral fingolimod (FTY720) in multiple sclerosis: 3-year results. Mult Scler 16:197–207
Kappos L, Radue EW, O’Connor P et al (2010) A placebo-controlled trial of oral fingolimod in relapsing multiple sclerosis. N Engl J Med 362:387–401
Cohen JA, Barkhof F, Comi G et al (2010) Oral fingolimod or intramuscular interferon for relapsing multiple sclerosis. N Engl J Med 362:402–415
Cohen JA, Kappos L, Pelletier J et al (2010) Oral fingolimod (FTY720) in relapsing-remitting multiple sclerosis: safety findings from TRANSFORMS and FREEDOMS trials. Consortium of Multiple Sclerosis Centers (CMSC), San Antonio, p S22
Saab G, Almony A, Blinder KJ et al (2008) Reversible cystoid macular edema secondary to fingolimod in a renal transplant recipient. Arch Ophthalmol 126:140–141
Khatri B, Barkhof F, Comi G et al (2010) 24-Month efficacy and safety outcomes from the TRANSFORMS extension study of oral fingolimod (FTY720) in patients with relapsing-remitting multiple sclerosis. American Academy of Neurology, Toronto, P03.125
Carson DA, Kaye J, Seegmiller JE (1977) Lymphospecific toxicity in adenosine deaminase deficiency and purine nucleoside phosphorylase deficiency: possible role of nucleoside kinase(s). Proc Natl Acad Sci USA 74:5677–5681
Carson DA, Wasson DB, Taetle R et al (1983) Specific toxicity of 2-chlorodeoxyadenosine toward resting and proliferating human lymphocytes. Blood 62:737–743
Guarnaccia JB, Rinder H, Smith B (2008) Preferential effects of cladribine on lymphocyte subpopulations. Mult Scler 14:S45 (P55)
Beutler E, Sipe JC, Romine JS et al (1996) The treatment of chronic progressive multiple sclerosis with cladribine. Proc Natl Acad Sci USA 93:1716–1720
Romine JS, Sipe JC, Koziol JA et al (1999) A double-blind, placebo-controlled, randomized trial of cladribine in relapsing-remitting multiple sclerosis. Proc Assoc Am Physicians 111:35–44
Rice GP, Filippi M, Comi G (2000) Cladribine and progressive MS: clinical and MRI outcomes of a multicenter controlled trial. Cladribine MRI Study Group. Neurology 54:1145–1155
Giovannoni G, Comi G, Cook S et al (2010) A placebo-controlled trial of oral cladribine for relapsing multiple sclerosis. N Engl J Med 362:416–426
Hartung HP, Aktas O, Kieseier B et al (2010) Development of oral cladribine for the treatment of multiple sclerosis. J Neurol 257:163–170
Refusal of the marketing authorisation for Movectro (cladribine) (2011) http://www.ema.europa.eu/docs/en_GB/document_library/Summary_of_opinion_-_Initial_authorisation/human/001197/WC500101072.pdf. Accessed 24 January 2011
Acknowledgments
This review includes data presented by the authors during an expert meeting sponsored by Bayer Schering Pharma AG (Berlin, Germany) in Berlin, Germany on June 26, 2010. Editorial and medical writing support was provided by Physicians World Europe GmbH, (Mannheim, Germany), funded by Bayer Schering Pharma AG (Berlin, Germany). Bayer Schering Pharma AG had no role in data collection, data interpretation or writing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duddy, M., Haghikia, A., Cocco, E. et al. Managing MS in a changing treatment landscape. J Neurol 258, 728–739 (2011). https://doi.org/10.1007/s00415-011-6009-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-011-6009-x