Abstract
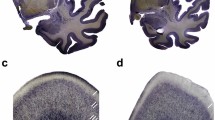
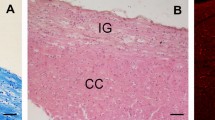
Von Economo neurons (VENs) are large spindle-shaped neurons localized to anterior cingulate cortex (ACC) and fronto-insular cortex (FI). VENs appear late in development in humans, are a recent phylogenetic specialization, and are selectively destroyed in frontotemporal dementia, a disease which profoundly disrupts social functioning and self-awareness. Agenesis of the corpus callosum (AgCC) is a congenital disorder that can have significant effects on social and emotional behaviors, including alexithymia, difficulty intuiting the emotional states of others, and deficits in self- and social-awareness that can impair humor, comprehension of non-literal or affective language, and social judgment. To test the hypothesis that VEN number is selectively reduced in AgCC, we used stereology to obtain unbiased estimates of total neuron number and VEN number in postmortem brain specimens of four normal adult controls, two adults with isolated callosal dysgenesis, and one adult whose corpus callosum and ACC were severely atrophied due to a non-fatal cerebral arterial infarction. The partial agenesis case had approximately half as many VENs as did the four normal controls, both in ACC and FI. In the complete agenesis case the VENs were almost entirely absent. The percentage of neurons in FI that are VENs was reduced in callosal agenesis, but was actually slightly above normal in the stroke patient. These results indicate that the VEN population is selectively reduced in AgCC, but that the VENs do not depend on having an intact corpus callosum. We conclude that in agenesis of the corpus callosum the reduction in the number of VENs is not the direct result of the failure of this structure to develop, but may instead be another consequence of the genetic disruption that caused the agenesis. The reduction of the VEN population could help to explain some of the social and emotional deficits that are seen in this disorder.






Similar content being viewed by others
References
Allman JM, Hakeem A, Watson KK (2002) Two phylogenetic specializations in the human brain. Neuroscientist 8:335–346
Allman JM, Watson KK, Tetreault NA, Hakeem AY (2005) Intuition and autism: a possible role for Von Economo neurons. Trends Cogn Sci 9:367–373. doi:10.1016/j.tics.2005.06.008
Bartels A, Zeki S (2000) The neural basis of romantic love. Neuroreport 11:3829–3834. doi:10.1097/00001756-200011270-00046
Berthoz S, Armony JL, Blair RJ, Dolan RJ (2002) An fMRI study of intentional and unintentional (embarrassing) violations of social norms. Brain 125:1696–1708. doi:10.1093/brain/awf190
Brown WS, Paul LK (2000) Cognitive and psychosocial deficits in agenesis of the corpus callosum with normal intelligence. Cognit Neuropsychiatry 5:135–157. doi:10.1080/135468000395781
Brown WS, Paul LK, Symington M, Dietrich R (2005) Comprehension of humor in primary agenesis of the corpus callosum. Neuropsychologia 43:906–916. doi:10.1016/j.neuropsychologia.2004.09.008
Chiarello C (1980) A house divided? Cognitive functioning with callosal agenesis. Brain Lang 11:128–158. doi:10.1016/0093-934X(80)90116-9
David AS, Wacharasindhu A, Lishman WA (1993) Severe psychiatric disturbance and abnormalities of the corpus callosum: review and case series. J Neurol Neurosurg Psychiatry 56:85–93
Dennis M (1981) Language in congenitally acallosal brain. Brain Lang 12:33–53. doi:10.1016/0093-934X(81)90004-3
Devinsky O, Laff R (2003) Callosal lesions and behavior: history and modern concepts. Epilepsy Behav 4:607–617. doi:10.1016/j.yebeh.2003.08.029
Duan X, Chang JH, Ge S et al (2007) Disrupted-in-schizophrenia 1 regulates integration of newly generated neurons in the adult brain. Cell 130:1146–1158. doi:10.1016/j.cell.2007.07.010
Ettlinger G (1977) Agenesis of the corpus callosum. In: Vinken PJ, Bruyn GW (eds) Handbook of clinical neurology, vol 30. Elsevier, Amsterdam, pp 285–297
Fischer M, Ryan SB, Dobyns WB (1992) Mechanisms of interhemispheric transfer and patterns of cognitive function in acallosal patients of normal intelligence. Arch Neurol 49:271–277
Gilliam F, Wyllie E, Kotagal P, Geckler C, Rusyniak G (1996) Parental assessment of functional outcome after corpus callosotomy. Epilepsia 37:753–757. doi:10.1111/j.1528-1157.1996.tb00647.x
Gott PS, Saul RE (1978) Agenesis of the corpus callosum: limits of functional compensation. Neurology 28:1272–1279
Gundersen HJ, Jensen EB, Kieû K, Nielsen J (1999) The efficiency of systematic sampling in stereology—reconsidered. J Microsc 193:199–211. doi:10.1046/j.1365-2818.1999.00457.x
Hakeem AY, Sherwood CC, Bonar CJ, Hof PR, Allman JM (2008) Von Economo neurons in the elephant brain. Anat Rec (in press)
Hof PR, Van der Gucht E (2007) Structure of the cerebral cortex of the humpback whale, Megaptera novaeangliae (Cetacea, Mysticeti, Balaenopteridae). Anat Rec 290:1–31. doi:10.1002/ar.20407
Hoppe KB, Bogen JE (1977) Alexithymia in 12 commissurotomized patients. Psychother Psychosom 28:148–155
Jeeves MA (1994) Callosal agenesis—a natural split brain: overview. In: Lassonde M, Jeeves MA (eds) Callosal agenesis—a natural split brain?. Plenum Press, New York, pp 285–299
Jeeves MA, Temple CM (1987) A further study of language function in callosal agenesis. Brain Lang 32:325–335. doi:10.1016/0093-934X(87)90131-3
Jeret JS, Serur D, Wisniewski KE, Lubin RA (1987) Clinicopathological findings associated with agenesis of the corpus callosum. Brain Dev 9:255–264
Karama S, Lecours AR, Leroux J, Bourgouin P, Beaudoin G, Joubert S et al (2002) Areas of brain activation in males and females during viewing of erotic film excerpts. Hum Brain Mapp 16:1–13. doi:10.1002/hbm.10014
Lassonde M, Sauerwein C (1997) Neuropsychological outcome of corpus callosotomy in children and adolescents. J Neurosurg Sci 41:67–73. doi:10.1097/00006123-199707000-00014
Loeser JD, Alvord EC (1968) Agenesis of the corpus callosum. Brain 91:553–570. doi:10.1093/brain/91.3.553
Lough S, Kipps CM, Treise C, Watson P, Blair JR, Hodges JR (2006) Social reasoning, emotion and empathy in frontotemporal dementia. Neuropsychologia 44:950–958. doi:10.1016/j.neuropsychologia.2005.08.009
Meyer BU, Roricht S, Niehaus L (1998) Morphology of acallosal brains as assessed by MRI in six patients leading a normal daily life. J Neurol 245:106–110. doi:10.1007/s004150050187
Miller BL, Seeley WW, Mychack P, Rosen HJ, Mena I, Boone K (2001) Neuroanatomy of the self: evidence from patients with frontotemporal dementia. Neurology 57:817–821
Nimchinsky EA, Gilissen E, Allman JM, Perl DP, Erwin JM, Hof PR (1999) A neuronal morphologic type unique to humans and great apes. Proc Natl Acad Sci USA 96:5268–5273. doi:10.1073/pnas.96.9.5268
Nimchinsky EA, Vogt BA, Morrison JH, Hof PR (1995) Spindle neurons of the human anterior cingulate cortex. J Comp Neurol 355:27–37. doi:10.1002/cne.903550106
O’Brien G (1994) The behavioral and developmental consequences of corpus callosum agenesis and Aicardi Syndrome. In: Lassonde M, Jeeves MA (eds) Callosal agenesis: a natural split brain?. Plenum, New York, pp 235–246
Paul LK, Brown WS, Adolphs R, Tyszka JM, Richards LJ, Mukherjee P et al (2007) Agenesis of the corpus callosum: genetic, developmental and functional aspects of connectivity. Nat Rev Neurosci 8:287–299. doi:10.1038/nrn2107
Paul LK, Schieffer B, Brown WS (2004) Social processing deficits in agenesis of the corpus callosum: narratives from the thematic appreciation test. Arch Clin Neuropsychol 19:215–225. doi:10.1016/S0887-6177(03)00024-6
Paul LK, Van Lancker-Sidtis D, Schieffer B, Dietrich R, Brown WS (2003) Communicative deficits in agenesis of the corpus callosum: nonliteral language and affective prosody. Brain Lang 85:313–324. doi:10.1016/S0093-934X(03)00062-2
Rakic P (1984) Emergence of neuronal and glial cell lineages in primate brain. In: Black IB (ed) Cellular and molecular biology of neural development. Plenum Press, New York
Rankin KP, Gorno-Tempini ML, Allison SC, Stanley CM, Glenn S, Weiner MW et al (2006) Structural anatomy of empathy in neurodegenerative disease. Brain 129:2945–2956. doi:10.1093/brain/awl254
Rose JE, Woolsey CN (1948) Structure and relations of limbic cortex and anterior thalamic nuclei in rabbit and cat. J Comp Neurol 89:279–347. doi:10.1002/cne.900890307
Rose JE, Woolsey CN (1958) Cortical connections and functional organization of thalamic auditory system of cat. In: Harlow HF, Woolsey CN (eds) Biological and biochemical bases of behavior. University of Wisconsin Press, Madison, pp 127–150
Samuelsen GB, Larsen KB, Bogdanovic N, Laursen H, Graem N, Larsen JF et al (2003) The changing number of cells in the human fetal forebrain and its subdivisions: a stereological analysis. Cereb Cortex 13:115–122. doi:10.1093/cercor/13.2.115
Sanders RJ (1989) Sentence comprehension following agenesis of the corpus callosum. Brain Lang 37:59–72. doi:10.1016/0093-934X(89)90101-6
Sauerwein HC, Nolin P, Lassonde M (1994) Cognitive functioning in callosal agenesis. In: Lassonde M, Jeeves MA (eds) Callosal agenesis: a natural split brain?. Plenum, New York, pp 221–233
Saul RE, Sperry RW (1968) Absence of commissurotomy symptoms with agenesis of the corpus callosum. Neurology 18:307
Schmitz C, Hof PR (2005) Design-based stereology in neuroscience. Neuroscience 130:813–831. doi:10.1016/j.neuroscience.2004.08.050
Seeley WW, Carlin DA, Allman JM, Macedo MN, Bush C, Miller BL et al (2006) Early frontotemporal dementia targets neurons unique to apes and humans. Ann Neurol 60:660–667. doi:10.1002/ana.21055
Seeley WW, Allman JM, Carlin DA et al (2007) Divergent social functioning in behavioral variant frontotemporal dementia and Alzheimer disease: reciprocal networks and neuronal evolution. Alzheimer Dis Assoc Disord 21:S50–S57
Shin LM, Dougherty DD, Orr SP, Pitman RK, Lasko M, Macklin ML et al (2000) Activation of anterior paralimbic structures during guilt-related script-driven imagery. Biol Psychiatry 48:43–50. doi:10.1016/S0006-3223(00)00251-1
Singer T, Seymour B, O’Doherty J, Kaube H, Dolan RJ, Frith CD (2004) Empathy for pain involves the affective but not sensory components of pain. Science 303:1157–1162. doi:10.1126/science.1093535
Singer T, Seymour B, O’Doherty JP, Stephan KE, Dolan RJ, Frith CD (2006) Empathic neural responses are modulated by the perceived fairness of others. Nature 439:466–469. doi:10.1038/nature04271
Snowden JS, Gibbons ZC, Blackshaw A, Doubleday E, Thompson J, Craufurd D et al (2003) Social cognition in frontotemporal dementia and Huntington’s disease. Neuropsychologia 41:688–701. doi:10.1016/S0028-3932(02)00221-X
Sokal RR, Rohlf FJ (1995) Biometry: the priciples and practice of statistics in biological research. W.H. Freeman, New York
Sturm VE, Rosen HJ, Allison S, Miller BL, Levenson RW (2006) Self-conscious emotion deficits in frontotemporal lobar degeneration. Brain 129:2508–2516. doi:10.1093/brain/awl145
Taylor M, David AS (1998) Agenesis of the corpus callosum: a United Kingdom series of 56 cases. J Neurol Neurosurg Psychiatry 64:131–134
Temple CM, Ilsley J (1993) Phonemic discrimination in callosal agenesis. Cortex 29:341–348
Temple CM, Jeeves MA, Vilarroya O (1989) Ten pen men: rhyming skills in two children with callosal agenesis. Brain Lang 37:548–564. doi:10.1016/0093-934X(89)90111-9
Temple CM, Jeeves MA, Vilarroya OO (1990) Reading in callosal agenesis. Brain Lang 39:235–253. doi:10.1016/0093-934X(90)90013-7
TenHouten WD, Hoppe KD, Bogen JE, Walter DO (1986) Alexithymia: an experimental study of cerebral commissurotomy patients and normal control subjects. Am J Psychiatry 143:312–316
Tomasch J (1954) Size, distribution, and number of fibers in the human corpus callosum. Anat Rec 119:119–135. doi:10.1002/ar.1091190109
Von Economo C, Koskinas G (1925) Die Cytoarchitectonik der Hirnrinde des erwachsenen Menschen. Springer
Watson KK, Jones TK, Allman JM (2006) Dendritic architecture of the von Economo neurons. Neuroscience 141:1107–1112. doi:10.1016/j.neuroscience.2006.04.084
Watson KK, Matthews BJ, Allman JM (2006) Brain activation during sight gags and language-dependent humor. Cereb Cortex (in press)
West MJ, Slomianka L, Gundersen HJG (1991) Unbiased stereological estimation of the total number of neurons in the subdivisions of the rat hippocampus using the optical fractionator. Anat Rec 231:482–497. doi:10.1002/ar.1092310411
Zaidel DW (1995) A view of the world from a split-brain perspective. In: Critchley EMR (ed) The neurological boundaries of reality. Jason Aronson, Northvale, pp 161–174
Acknowledgments
We thank Archibald Fobbs, Curator of the Yakovlev Brain Collection, National Museum of Health and Medicine for his generous assistance in providing access to the acallosal and control specimens, and to Dr. D. Wolfe of the Mount Sinai School of Medicine for generously providing access to the cerebral infarction specimen. We also wish to thank Ralph Adolphs, Warren Brown and J. Michael Tyszka for their valuable discussions of this project. This research was generously funded by grants from the James S. McDonnell Foundation, the Gordon and Betty Moore Foundation, the David and Lucile Packard Foundation, and the Gustavus and Louise Pfeiffer Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaufman, J.A., Paul, L.K., Manaye, K.F. et al. Selective reduction of Von Economo neuron number in agenesis of the corpus callosum. Acta Neuropathol 116, 479–489 (2008). https://doi.org/10.1007/s00401-008-0434-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-008-0434-7