[2+2]
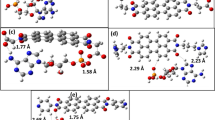
cycloaddition reaction between \chem{C_{60}} molecules is studied theoretically using the semi-empirical quantum chemical AM1 method. The studies are aimed at understanding of photo- and pressure polymerization of pristine \chem{C_{60}} and polymerization of the alkali metal (\chem{\chemvar{A}})/\chem{C_{60}} complex, \chem{\chemvar{A}_{1}C_{60}}. Full geometry optimizations of the [2+2] cycloaddition reaction, have been performed for neutral as well as anionic \chem{C_{60}} dimers. Based on these geometries, th e reaction barriers were calculated. Vanishing barriers were found for the anionic dimer and in the first singlet (\state{}{S}{1}) and triplet (\state{}{T}{1}) excited states of the neutral dimer, results that indicate spontaneous polymerisation for these systems in full agreement with experimental observations. The occurrence of the [2+2] cycloaddition reaction is fully understood from a detailed analysis of the molecular orbitals and how these correlate with the change in the interfullerene distance. We have located a critical intermolecular distance, R_{\text{C}}=\valunit{2.12}{\Angstrom}, for which the [2+2] cycloaddition occurs as a result of an interchange in the position in the energy spectrum between the highest occupied and lowest unoccupied molecular orbitals.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 2 October 1996/Accepted: 11 November 1996
Rights and permissions
About this article
Cite this article
Stafström, S., Fagerström, J. Electronic structure and stability of fullerene polymers. Appl Phys A 64, 307–314 (1997). https://doi.org/10.1007/s003390050483
Issue Date:
DOI: https://doi.org/10.1007/s003390050483