Abstract
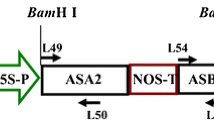
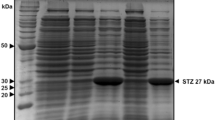
A transgenic cell suspension culture of Nicotiana tabacum L. `Petit Havana' SR1 was established expressing tryptophan decarboxylase and strictosidine synthase cDNA clones from Catharanthus roseus (L.) G. Don under the direction of cauliflower mosaic virus 35S promoter and nopaline synthase terminator sequences. During a growth cycle, the transgenic tobacco cells showed relatively constant tryptophan decarboxylase activity and an about two- to sixfold higher strictosidine synthase activity, enzyme activities not detectable in untransformed tobacco cells. The transgenic culture accumulated tryptamine and produced strictosidine upon feeding of secologanin, demonstrating the in vivo functionality of the two transgene-encoded enzymes. The accumulation of strictosidine, which occurred predominantly in the medium, could be enhanced by feeding both secologanin and tryptamine. No strictosidine synthase activity was detected in the medium, indicating the involvement of secologanin uptake and strictosidine release by the cells.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 25 February 1996 / Revision received: 16 August 1996 / Accepted: 30 September 1996
Rights and permissions
About this article
Cite this article
Hallard, D., van der Heijden, R., Verpoorte, R. et al. Suspension cultured transgenic cells of Nicotiana tabacum expressing tryptophan decarboxylase and strictosidine synthase cDNAs from Catharanthus roseus produce strictosidine upon secologanin feeding. Plant Cell Reports 17, 50–54 (1997). https://doi.org/10.1007/s002990050350
Issue Date:
DOI: https://doi.org/10.1007/s002990050350